1. Background
Alzheimer’s disease (AD) is a type of age-related illness that affects more and more people (1). It is a type of amnesiac disorder with dysfunction of the brain in which the patient’s mental ability is gradually dissipated. The most obvious type of dementia is a memory disorder (2). Memory disorder usually develops and progresses gradually (2). Studies have shown that this neurodevelopmental disease is one of the main causes of dementia (a condition where the ability to think, understand, and remember is impaired); it begins in old age with the loss of information retention power, especially temporary memory and gradually ends with the loss of the ability to recognize time, depression, loss of speech power, loneliness and eventually death due to respiratory disorders (2). Moreover, AD is currently the most common cause of death in western countries after heart disease, cancer, and stroke (3). In the living being’s brain, there is a special memory system, called the hippocampus, which is responsible for learning and storage of vital information and is a structure for learning and memories (4). This area in the right part of the human brain controls the ability of spatial orientations. There is some evidence that the hippocampus contains cognitive maps in humans. The memory of people whose hippocampus is damaged or surgically removed is severely impaired (5). Various factors such as oxidative stress, glutamate-dependent toxicity, decrease in acetylcholine and serotonin, decrease or disappearance of dopamine neurons, as well as inflammation of brain tissue, are involved in the development of AD (6). Serotonin (a type of monoamine biogenic acid neurotransmitter) is biochemically derived from tryptophan that has been found primarily in the gastrointestinal tract, platelets, and the central nervous system of animals as well as humans (7). Serotonin levels are related to the amount of tryptophan in the blood. Researchers have shown that 10% of tryptophan is free in the blood, and free tryptophan is able to enter the brain and the rest is bound to albumin (8). Serotonin also has cognitive functions, such as effects on memory and learning. Serotonin modulation in synapses has been recognized as a key function for several different classes of antidepressants (9). With growing old and aging of cells, the secretion of serotonin decreases, making it difficult to communicate between cells and transfer information (10). This problem is most evident in the segment of retention and memories so that memories are faded or forgotten (10). Several studies have reported that exercise can alter the release of neurotransmitters such as dopamine, glutamate, acetylcholine, serotonin, and endogenous opioids in the brain. In this regard, Arazi et al. found that combined training could advance levels of blood serotonin and dopamine, and improve health-related fitness factors in methamphetamine-addicted men and could be helpful as a non-pharmacological treatment during rehabilitation (11). Also, Yu et al. (12) reported that 3 sessions of moderate-intensity cycling aerobic training for 6 months could prevent memory loss. It has also been reported that aerobic training can reduce symptoms and disease, increase hippocampal neurogenesis, and stop memory loss in animal models (13). However, 8 weeks of aerobic training had no significant effect on serotonin concentration (14).
2. Objectives
Given the contradictory results of the research conducted and the possible effects of training on different slopes on preventing AD, as well as the possibility of influencing the type of training on AD disorders, and lack of research in comparing the effects of running on positive and negative slopes in AD, this study aims to investigate the effect of running on positive and negative slopes on serotonin gene expression in the hippocampus tissue of rats with AD.
3. Methods
In this experimental study, 24 rats were purchased from animal breeding and proliferation center of Islamic Azad University of Marvdasht and transferred to the physiology laboratory of this university. To adapt, rats were kept for one week under standard conditions in transparent auto- cleavable polycarbonate cages with optimum temperature (20 to 24ºC), relative humidity (55 to 65%), 12-hour darkness-light cycle, and had free access to food and water. Then on the eighth day, 18 rats were intra-peritoneally injected with 8mg/kg trimethyltin chloride (TMT) (11); after 24 hours, being assured of complete effect of TMT on the hippocampus, AD symptoms were observed by a number of behavioral symptoms in rats. These clinical symptoms included (1) muscle tremors, (2) elevated body temperature, (3) nausea, (4) seizures, and (5) tail twists. Rats with AD were randomly divided into 3 groups of 6 rats, including (1) control, (2) training on positive slope and (3) training on negative slope. It is noteworthy that in order to investigate the effects of AD induction on serotonin levels, 6 rats were placed in the healthy control group. The training groups ran on the treadmill for one week, three sessions per week, at a speed of 5 to 10 m/min for 5 to 10 min. Then, the rats in the positive training group (at a speed of 16 m/min on positive upward slope) and negative training group (at a speed of 16 m/min on negative downhill slope) performed endurance training for eight weeks, five sessions per week and 60 minutes per session (15). To perform training, rats initially ran on the treadmill with zero slope for 5 min at a speed of 8 m/min for warm-up; afterward, the training groups performed training at a speed of 15 cm/s in the first week at +15% and -15% slopes, adding 5 cm/s to the treadmill speed each week. At the end of each training session, the rats cooled down again for 5 min at 8 m/min on the treadmill with zero slopes. The training was performed such that rats trained for one hour as the sum of warm-up and cool-down (15). It should be noted that all training sessions performed between 9 a.m. to 12 a.m. Moreover, the rats in the control group were the rats with AD but in the healthy control group were intact rats (healthy rats). Indeed, during the research period, the control and healthy control groups did not perform positive and negative slope training. The healthy control group was used to review the effects of AD induction on the research variables; so TMT was injected into the control group and healthy control group did not receive TMT. Forty-eight hours after the last training session, the rats were anesthetized with ketamine 10% (50 mg/kg dose) and xylazine 2% (10 mg/kg dose) after approximately 5 minutes. The hippocampus tissue was extracted by specialists and after setting in cryotube was placed in liquid nitrogen and stored at -70ºC for further investigation. For molecular analysis at the gene expression level, firstly, extraction of RNA from the hippocampus tissue was carried out according to the manufacturer’s protocol (Sinagen, Iran); secondly, using light absorbance at a wavelength of 260 nm, the concentration and degree of purity of the RNA sample were quantitatively obtained using the following equation:
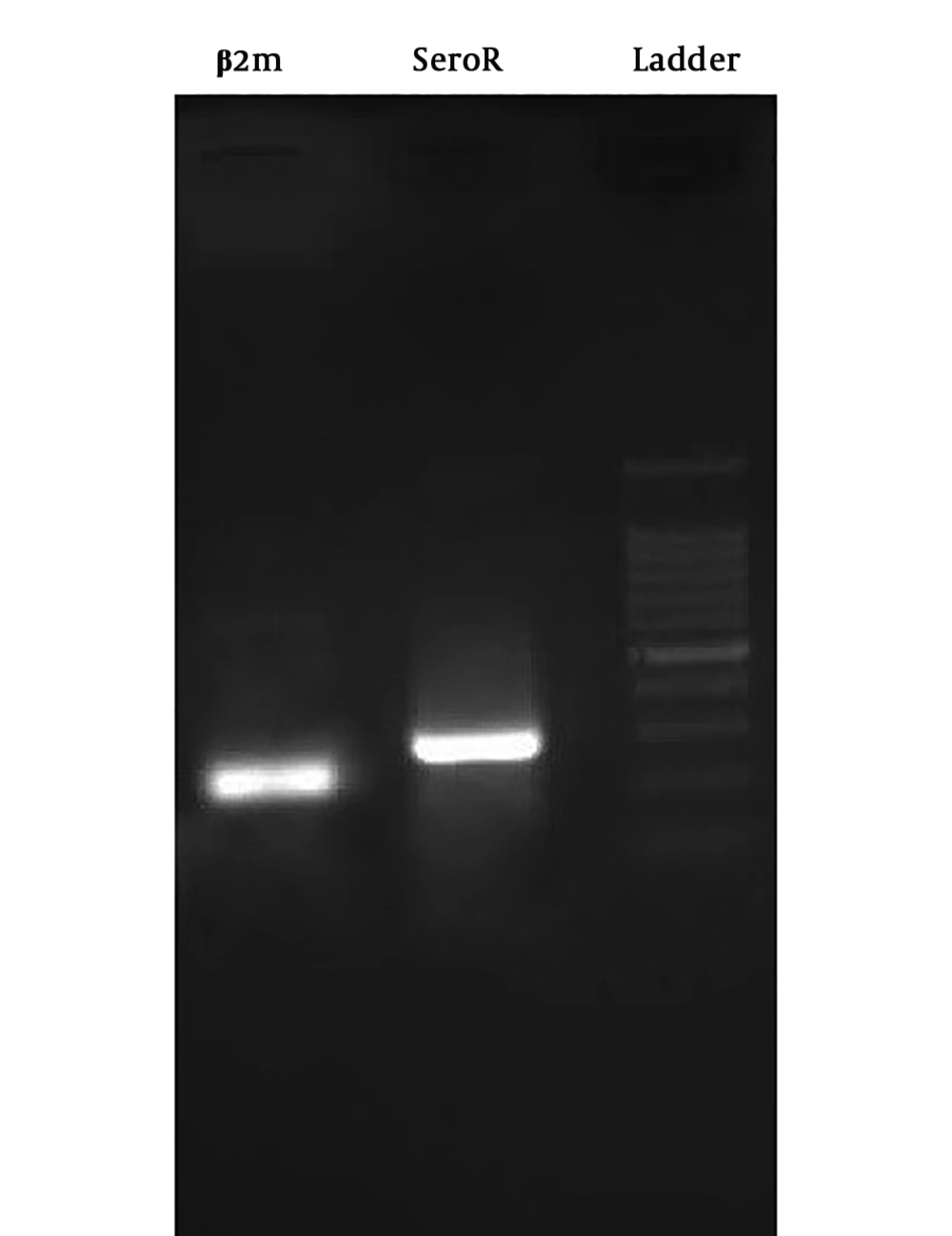
After extracting RNA with high purity and high concentration from all of the samples, cDNA synthesis steps were taken according to the manufacturer’s protocol, and then the synthesized cDNA was used for reverse transcription reaction. Initially, the designed primers for genes were examined, and then gene expressions were examined by quantitative q-RT PCR method. It should be noted that B2m was selected as the housekeeping gene. The sequence of primers used is shown in Table 1. Shapiro-Wilk, One-way ANOVA with Tukey’s post hoc tests were used for statistical analysis of data (P ≤ 0.05).
| Genes | Primer Sequences | Product Size (bp) |
|---|---|---|
| B2m | 244 | |
| Forward | 5’- CGTGCTTGCCATTCAGAAA -3’ | |
| Reverse | 5’-ATATACATCGGTCTCGGTGG -3’ | |
| Serotonin receptor | 283 | |
| Forward | 5’- TTAGGAACTTCGTCGGCACC -3’ | |
| Reverse | 5’- CCATCTTGCGCTTTGCTTCA-3’ |
4. Results
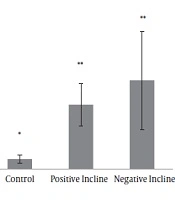
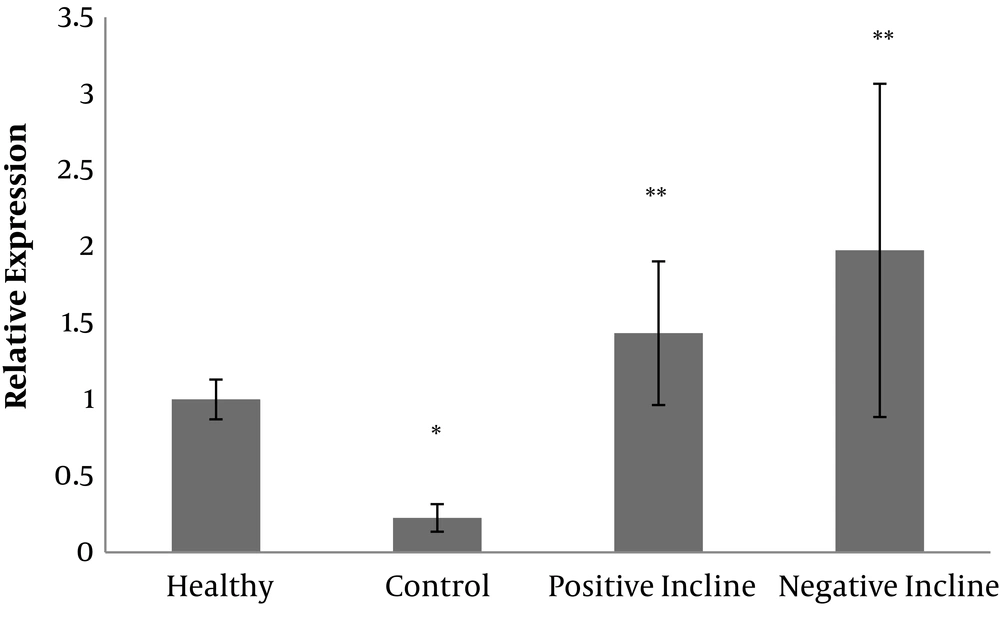
Serotonin gene expression levels are presented in Figure 1. The results of one-way ANOVA test showed that there were significant differences in serotonin gene expression levels in the four groups of study (F = 9.01, P = 0.001).
The results of Tukey’s post hoc test showed that serotonin gene expression levels in the control group were significantly lower than the healthy group (P = 0.04). On the other hand, serotonin gene expression levels in the training on the positive slope group (P = 0.01) and training on negative slope group (P = 0.001) were significantly higher than the control group; however, there was no significant difference in serotonin gene expression levels in the training on positive slope and training on negative slope groups (P = 0.42) (Figure 1). A figure of PCR product is presented in Figure 2.
5. Discussion
The purpose of the present study was to investigate the effect of running on positive and negative slopes on serotonin gene expression levels in the hippocampus tissue of rats with AD. The results of this study showed that induction of AD with TMT leads to decreased serotonin gene expression in the hippocampus tissue of the rats. However, running on positive and negative slopes had a significant effect on increasing serotonin gene expression levels in rats with AD. Also, running on both positive and negative slopes had the same effects on increasing serotonin gene expression levels in rats with AD. Serotonin in the human body is concentrated in the enterochromaffin cells (8), which are scattered in the gastrointestinal system membrane, where it regulates bowel motility. Serotonin also has cognitive functions, such as effects on memory and learning (8). Increased serotonin has been reported to improve spatial memory and learning as well as happiness and delight in individuals; however, as cells grow up and become old, serotonin secretion decreases, thus intracellular communication and data transfer become difficult (16). This problem is most evident in the segments of retention and memory; hence, a decrease in serotonin secretion in the brain may cause memories to become faded or forgotten and finally, may result in dementia (16). In fact, the results of the present study indicate a significant effect of running on negative and positive slopes on the improvement of serotonin gene expression. Consistent with the findings of the present study, the results of the study by Tapia-Rojas et al. (10), Rammes et al. (13) and O’Dell et al. (17) revealed that AD induction can lead to a significant decrease in serotonin. Such a decrease in AD induction may result in impairment of dopaminergic and serotonergic probably due to disturbance in the serotonin receptors in the brain or their damage, and this may decrease the serotonin and dopamine transmitters (18, 19). Concerning the effects of exercise on serotonin, in line with the findings of the present study, Avila et al. (1), Garcia-Mesa et al. (2), and O’Dell et al. (17) reported that exercise activities had a significant effect on increased serotonin levels.
Exercise activity seems to specifically influence specific levels of serotonin and dopamine transmitters that increase fatty acid recruitment; thus, the level of free tryptophan in blood increase due to the fatty acid-tryptophan competition in binding to albumin (13). Therefore, the rate of serotonin synthesis in the brain increases and raises them (13, 20). Aerobic exercise activities seem to induce vascular endothelial growth factor and it may contribute to recover angiogenesis-induced injury and have a direct effect on neurotrophic growth factor, leading to the restoration and repair of monoaminergic damage to dopamine and serotonin (21). Also, exercise may specifically affect specific levels of serotonin and dopamine transmitters and may increase serotonin receptors such as 5-HT1B, which determine the final pathway and release of serotonin (21). Given that the intensity of exercise activity may play an important role in increasing serotonin levels, it is suggested that in future studies the effect of exercise with different intensities on negative and positive slopes on serotonin gene expression levels should be considered. Besides, lack of use of various methods of measuring serotonin such as ELISA and western blot as well as lack of control the possible differences in stress levels between animals were the limitations of the present study. Therefore, it is recommended that future studies measure serotonin levels at the levels of gene and protein expression and review the effect of training in different times of day on serotonin levels in the hippocampus tissue of rats with AD.
5.1. Conclusions
According to the findings of the present study, it seems that running on positive and negative slopes can improve serotonin gene expression levels in the hippocampus tissue of rats with AD.