1. Background
Type 2 diabetes mellitus (T2DM) is one of the most significant threats to human health in the 21st century (1). T2DM accounts for more than 90% of diabetic cases worldwide. Approximately one out of every 10 people suffers from T2DM or is to develop it and the overall number of T2DM patients is anticipated to double by the year 2025 (2).
Insulin resistance is defined as a reduced response of target tissues, such as the skeletal muscle, liver, and adipocytes, to insulin (3). In recent years, Genome-Wide Association Studies (GWAS) have recognized new Single Nucleotide Polymorphisms (SNPs) to be linked to T2DM (4) although SNPs in the resistin gene are still unclear.
Resistin, referred to as FIZZ-3, is found in inflammatory zone 3, and is a 12.5 kDa peptide hormone consisting of 108 amino acids. It belongs to the family of cysteine-rich secretory proteins. It has been elucidated to make a link between obesity and diabetes in mouse models (5). Several studies (6-8) have revealed that the serum resistin level is closely associated with metabolic risk factors and resistance to insulin, indicating that resistin may play an important role in the pathophysiology of T2DM.
In humans, resistin could suppress adipocyte differentiation by 80% in cultured 3T3-L1 cells (9). T2DM is now generally accepted to be due to a failure in adipocyte differentiation, probably with an ectopic overload of fatty acids and lipotoxicity of non-adipose tissues, such as muscles and liver (10, 11). Overall, the role of resistin is inconsistent in inflammation, glucose homeostasis, and insulin resistance in T2D patients.
The human resistin gene is localized to chromosome 19p13.3. It is predicted that up to 70% of the variation in serum resistin levels can be explained by genetic factors (12). The findings of studies exploring genetic variations of resistin and SNPs are controversial. The resistin gene promoter SNP at -420 C/G (rs1862513), as one of the most frequently studied SNPs, was revealed to be correlated with serum resistin levels (13).
Osawa et al. (14) demonstrated that the resistin-420 GG genotype was correlated with T2DM in the Japanese population. However, several subsequent investigations did not find a correlation. Lately, a study revealed that the resistin-420 CC genotype was correlated with the increased risk of T2DM (15). However, the correlation between resistin gene promoter -420 (C>G) polymorphism and T2DM risk remains unclear.
2. Objectives
The present study was conducted to investigate the difference of one promoter variant (SNP) in the resistin gene at position -420 (C>G) between unrelated T2DM patients and non-diabetic control subjects in Khuzestan province.
3. Methods
3.1. Subjects
In the present case-control study, we examined 200 unrelated subjects with T2DM and 200 non-diabetic control subjects. Diabetes was diagnosed based on the World Health Organization (WHO) criteria (fasting blood glucose ≥ 126 mg/dL and/or 2-h glucose level ≥ 200 mg/dL in the oral glucose tolerance test) (15). The T2DM patients were selected from among subjects referring to the Golestan Hospital of Ahwaz, southwest of Iran. The sample size was calculated using the G Power 2.9.1.3 software. The inclusion criteria for the non-diabetic control subjects were the lack of diabetes after clinical assessment and no family history of diabetes in first-degree relatives. The healthy control subjects and diabetic patients were matched for age, gender, and geographic area. The protocol was approved by the Ethics Committee of Ahvaz Jundishapour University Medical Sciences and all participants in the study agreed to participate by signing informed consent forms.
3.2. DNA Extraction and Genotyping
Approximately, 5 mL of peripheral blood was obtained from both case and healthy control groups in falcon tubes containing EDTA. Genomic DNA was obtained from blood samples following the salting-out method (16) and stored at -20ºC or amplified immediately. Then, the concentration of DNA samples was determined using a NanoDrop 1000 spectrophotometer.
To determined genotype frequencies of -420 C/G resistin gene polymorphism, we used the polymerase chain reaction (PCR)-amplified DNA based on the restriction fragment length polymorphism (RFLP) method. The primer pair sequence used for the amplification of each DNA template was previously reported (15). The PCR cycling conditions included an initial denaturation at 95ºC for 5 minutes, 35 cycles of denaturation at 95ºC for 30 seconds, annealing at 60ºC for 30 seconds, extension at 72ºC for 30 seconds, and a final extension at 72ºC for 10 minutes in a Biorad thermocycler (Bio-Rad Laboratories, Hercules, CA, USA).
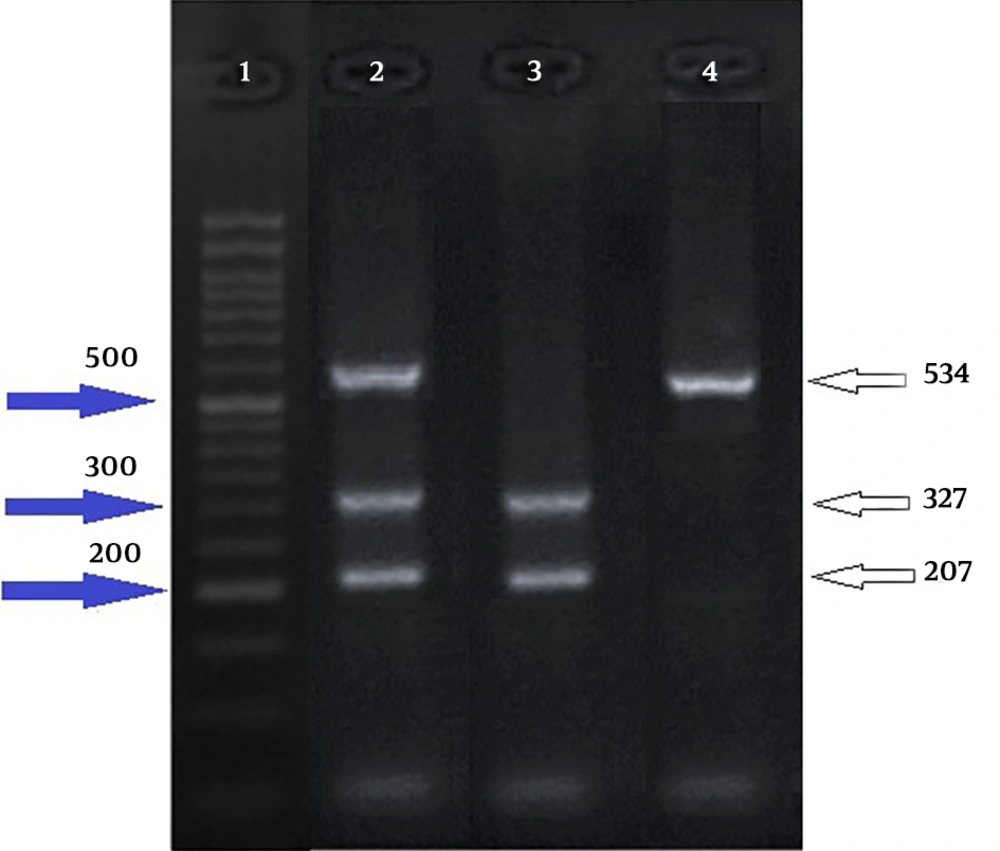
After amplification with PCR, all amplified fragments were subjected to digestion using a restriction enzyme. The selected restriction enzyme was BbsI for polymorphic site-420C/G; the mentioned restriction enzyme was not sensitive to methylated CpGs. The PCR products were run on a 2.5% agarose gel electrophoresis and illuminated by the safe stain. The digested DNA fragments were obtained for the CC genotype (207 bp and 327 bp), CG genotype (207 bp, 327 bp, and 534 bp), and GG genotype (534 bp) (Figure 1).
3.3. Statistical Analysis
Genotype distribution and allelic frequency were compared between T2DM case and control groups using the chi-square test. The results were analyzed using SPSS version 25 statistical software. Two-sided P values of < 0.05 were considered significant.
4. Results
This study sample consisted of 400 subjects including 202 (51%) and 198 (49%) males and females, respectively. Type 2 diabetes was present in 50% (n = 200) of all participants. The clinical features of the subjects are shown in Table 1. There were no significant differences in terms of age distribution, body mass index (BMI), sex, and hsCRP between T2DM and control groups, but the T2DM group had significantly higher FBG, HbA1C, triglyceride, and cholesterol levels than control subjects.
| Variables | T2DM Group (N = 200) | Control Group (N = 200) |
|---|---|---|
| Age, y | 61 ± 6 | 62 ± 5 |
| Sex, Male/Female, No. (%) | 96 (48)/104 (52) | 106 (53)/94 (47) |
| Body Mass Index, kg/m2 | 27.3 ± 5 | 26.2 ± 6.2 |
| Fasting blood glucose, mg/dL | 136.7 ± 59 | 88.5 ± 12.3 |
| Total cholesterol, mg/dL | 206.1 ± 53.3 | 180.3 ± 37.8 |
| Tri glyceride, mg/dL | 163 ± 67 | 141 ± 52 |
| hsCRP, mg/dL | 2.3 ± 1.9 | 2 ± 1.3 |
| HbA1C, % | 5.7 ± 1.8 | 4.1 ± 0.9 |
| HDL, mg/dL | 41 ± 16 | 46 ± 9 |
Table 2 shows the genotype distribution and allele frequency of the resistin gene polymorphism at -420 (C>G). Genotype distribution was different for the -420 (C>G) polymorphism between the T2DM and healthy groups (P ≤ 0.05). Moreover, CC and CG genotype carriers (heterozygous combined with homozygous mutant types) had nearly three-fold and two-fold higher risks, respectively, for developing T2DM than the GG genotype carriers (wild type). The frequency of the C allele was higher in the T2DM patients than in healthy control groups, which was statistically significant (P ≤ 0.05).
| Genotypes of -420 Resistin Gene Polymorphism | T2DM Group (N = 200) | Control Group (N = 200) | P Valueb |
|---|---|---|---|
| CC | 102 (51) | 42 (21) | < 0.001 |
| CG | 66 (33) | 135 (67) | < 0.001 |
| GG | 32 (16) | 33 (16.5) | 0.89 |
| C-allele | 270 (67.5) | 219 (54) | < 0.001 |
Frequency of Genotypes and Alleles of -420 Resistin Gene Polymorphism in Type 2 Diabetic Patients and Healthy Subjectsa
Table 3 shows the age of onset, FBG, Hb1AC, and resistin concentration based on resistin -420 (C>G) polymorphism genotypes in T2DM patients. Because of the lower frequency of T2DM patients with the GG genotype and different distributions of the CC genotype in T2DM patients, G allele carriers (CG and GG) were considered a single group. Our results revealed that the age of onset was lower in patients with the CC genotype than in G allele carriers. Also, CC homozygotes had higher FBG levels than carriers of the G allele, but G allele carriers had higher Hb1AC levels than CC homozygotes. In addition, resistin concentration was higher in the CC genotype than in the CG and GG genotypes. No statistically significant correlation was found between resistin -420 (C>G) polymorphism and the mentioned parameters.
| Variables | CC (N = 102) | CG+GG (N = 98) |
|---|---|---|
| Age of onset, y | 47 ± 7 | 51 ± 3 |
| Fasting blood glucose, mg/dL | 132 ± 43 | 113 ± 37 |
| HbA1C, % | 6.3 ± 3 | 6.8 ± 2 |
| Resistin, ng/mL | 2.7 ± 1.2 | 2.5 ± 1.3 |
5. Discussion
Several studies tried to clarify the role of resistin and revealed that resistin may impair insulin action and glucose tolerance and reduce glucose uptake by human skeletal muscle cells (17, 18). Moreover, the neutralization of resistin with an anti-resistin antibody developed insulin action in diet-induced obese mice (19). Resistin mRNA was expressed several times higher in human omental and subcutaneous white adipocyte cells than in adipocyte cells from the thigh, proposing that human resistin could play a key role in obesity-linked insulin resistance (20). It is recognized that most T2DM patients are involved with obesity and insulin resistance. Several authors showed that serum resistin levels raised in T2DM subjects (21, 22). Such results were confirmed in the present study. Also, in Thai subjects, the T2DM group had a higher waist-to-hip ratio (WHR) and resistin levels than the healthy control group and T2DM was correlated with raised central obesity. On the contrary, other studies showed no significant difference in plasma resistin levels between T2DM subjects and healthy controls (23). Studies have demonstrated no correlation between resistin levels and insulin resistance markers in T2DM patients (24, 25). Thus, the exact function of resistin requires further study. The -420 C/G polymorphism in the resistin gene promoter region is one of the most commonly studied polymorphisms. It seems that the distribution of genotype in this region is related to the regulation of gene expression of resistin (26, 27). Although, the findings of studies investigating -420C/G polymorphism in the resistin gene were debatable.
Our observations found a correlation between resistin gene polymorphism at -420 (C>G) and T2DM. In the present study, the presence of the CC genotype was two times more in subjects with T2DM than in healthy people. Also, diabetic patients with CC genotype had the highest fasting blood glucose and the lowest HbA1C levels among studied patients with other genotypes, but these differences were not statistically significant (P ≥ 0.05). Also, our observations showed no association between this polymorphism in T2DM and higher resistin concentration. In addition, the frequency of the C allele in the diabetic group was higher than that of the control group.
Of nine studies that analyzed the -420C/G polymorphism (15, 26, 28-35), four reported significant correlations with quantitative traits, all in T2DM subjects. In three studies, the C allele was correlated with reductions in weight-linked variables (15, 26, 28). In the fourth study, the C/C genotype of the -420C/G polymorphism was correlated with reduced SI in the interaction with BMI (29). In the study by Ukkola et al. on non-diabetic and hypertensive individuals, they found fasting blood glucose, HbA1 levels, and LDL levels were higher in subjects with CC genotype than in other genotype carriers, but these differences were not statically significant (33). In addition, in a study by Wang et al. on a Caucasian population, the CC genotype was related to lower insulin sensitivity compared to the CG genotype in the -420 region. These two studies indicate that C alleles in the -420 region are associated with obesity and diabetes phenotypes, in line with our study (29). Although several studies in China (34), Japan (7, 14), and Quebec (28) showed a relationship between the -420 G allele and high levels of glucose and type 2 diabetes, there was no significant association between -420 genotypes and type 2 diabetes and insulin resistance in the study by Norata et al. (6). In the present study, the age of diabetes was lower in individuals with CC genotype than in individuals with other genotypes, but this difference was not statistically significant; this result is in line with a previous study from Iran (15). In contrast, the results of a study by Ochi et al. showed that people with GG genotype in the -420 resistin gene were affected by type 2 diabetes at an earlier age (34). Overall, these results propose an intricate association between the resistin genotype and phenotypes linked to insulin resistance and obesity.
As previously noted, there is a contradiction between the findings of various studies about the involvement of the -420C/G polymorphism in the resistin gene promoter region in the pathogenesis of insulin resistance and diabetes. The contradictory results in the present study and other studies may be attributed to some factors including the design of the study, sample size, ethnicity, and the age distribution of cases and controls. Also, the interaction between resistin gene and other involved genes in diabetes etiology and environmental factors could clarify this discrepancy in the findings.
5.1. Conclusions
In summary, our findings demonstrated that the CC genotype, as compared to the CG and GG genotypes, was correlated with increased risk of T2DM. Further studies are warranted to fully explain the function of the resistin gene in T2DM etiology in other populations with large sample sizes and determine how the resistin polymorphism at -420 (C>G) can affect resistin gene expression.