1. Background
In December 2019, the rapid outbreak of “pneumonia of unknown origin” in Wuhan (China), caused by a new strain of coronavirus, was reported by the Chinese health authorities to World Health Organization (WHO) (1). This novel coronavirus disease was subsequently named “coronavirus disease 2019 (COVID-19)”, which was associated with “severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)”. Then, the rapidly spreading disease was declared a pandemic (2). As of June 11, 2020, among a total of 72 73958 laboratory confirmed COVID-19 cases, 4 13372 death were reported worldwide. In Bangladesh, as of June 11, 2020, 74 865 confirmed COVID-19 cases, and 1 012 deaths incidences have been recorded (3).
The heterogeneous clinical manifestations of COVID-19 with its fast human-to-human spreading across the world resulted in high rates of mortality rate in a short time (1, 2). According to the recent data, patients with COVID-19 having one or more comorbidities are at increased risk of mortality. Chronic comorbid conditions such as diabetes mellitus (DM), hypertension (HTN), endocrine disorders, and cardiovascular and pulmonary diseases have been identified as potential factors that aggravate the conditions of COVID-19 patients, resulting in more severe clinical outcomes (4). A nationwide analysis on 1590 hospitalized COVID-19 positive patients in China reported that hypertension (16.9%) and diabetes (8.2%) were the most prevalent comorbidities. This report highlighted that for 131 patients who reached in composite end-point of the disease, chronic obstructive pulmonary diseases (COPD), DM, HTN, and malignancy were the major risk factors (5). Studies found that having one or more comorbidity significantly increases the severity of the COVID-19, mostly in aged patients, and is associated with increased need for Intensive Care Unit (ICU) support in high-risk patients (4, 6). In Bangladesh, the prevalence of hypertension and diabetes, as the most common comorbidities, is 14 and 6%, respectively. In other words, Bangladesh is one of the 10 countries with the highest prevalence of diabetes. However, its diagnosis rate is low (7). The first case of COVID-19 in Bangladesh was identified on March 8 2020, and since then, the disease has spread rapidly (8). It’s well-documented that comorbidities are associated with increased risk of ICU hospitalization and need for mechanical ventilation, and declined survival rate (1, 4, 5). Bangladesh is in a big challenge to pull-down the number of positive cases along with the mortality rate.
2. Objectives
The current study aimed to, firstly, assess the impact of predisposed comorbidities on the severity of COVID-19, and secondly, investigating the associated clinical outcome of patients with COVID-19 infection in Bangladesh.
3. Methods
3.1. Study Design
This single-center observational retrospective study was conducted in Square hospitals ltd., which are tertiary level private hospitals, in Dhaka (Bangladesh) from April 30, 2020, to June 15, 2020. The study population consisted solely Bangladeshi citizens (South Asian) with moderate-to-critical COVID-19 symptoms with at least one comorbidity aged 35 to 74 years who were admitted in either the specialized ward or the ICU of the “COVID-19 unit” of the hospital. On admission, all patients were tested for COVID-19 using RT-PCR (real-time reverse-transcriptase polymerase chain reaction) cycler (by Rotor-Gene-Q/Cobas z480, and QIAGEN kits for real-time PCR, QIAGEN GmbH, Germany) for human coronavirus (SARS-CoV-2). The hospital has a two-step triage system in the emergency department for all admitted patients at the time of hospital-entry. The clinical management and treatment guidelines for patients with suspected/known COVID-19 are developed under the supervision of the “COVID-19 Clinical Management Team” (CCMT) of the hospital.
Data were extracted using “Electronic Medical Records” of the hospital, which filled by the physician members of the CCMT. The Modified Early Warning Score (MEWS) system was used to determine the severity of illness at the time of admission. All necessary laboratory tests were performed within 4 hours of hospitalization. The diagnostic and therapeutic interventions, patients, transfer from COVID-19 ward to COVID-19 ICU (with or without need for mechanical ventilation support), and discharge to general ward/home have been recorded by the hospital’s “COVID-19 clinical management protocol (CCMP)”. History of chronic diseases, provided therapeutic interventions, length of hospital stay (LHS), and survival/mortality records were assessed in different points of care to accurately estimate the impact of comorbidities on disease severity and clinical outcomes of patients.
The study design, including the selection of samples and data collection processes, is depicted in Figure 1.
3.2. Inclusion and Exclusion Criteria
The participants were evaluated against the following inclusion criteria:
1) A period of 14 days between the onset of symptoms and hospital admission;
2) Having at least one comorbidity added to COVID-19;
3) No history of hospitalization within the last 4-weeks of admission;
4) Positive COVID-19 test with one or more symptoms of moderate-to-critical COVID-19;
5) No current pregnancy;
The exclusion criteria were as follows:
1) Patient with newly diagnosed comorbidity (i.e. after hospital admission);
2) Any history of trauma or surgical procedure within the last 3 months of admission;
History of receiving immunosuppressants.
3.3. Definitions and Ethical Approval
Patients with asymptomatic (no significant symptoms of COVID-19) and mild COVID-19 (may have fever, loss of sense of smell, extreme tiredness, sore throat, etc.) were not hospitalized and excluded from the study. Only patients with moderate COVID-19 (persistent dry cough, the body temperature of 37.8°C or higher, diarrhea, headache, muscle aches, early signs of inflammation in lungs with difficulty in breathing, and oxygen saturation in arterial blood (SpO2) ≥ 90% on room air); severe COVID-19 (severe respiratory distress, breathlessness, respiratory rate > 30 breaths/min; SpO2 < 90% on room air, raised body temperature, tightness of chest, declined level of consciousness); and critical COVID-19 (bilateral opacities in chest imaging, respiratory failure, mild to severe acute respiratory distress syndrome (ARDS), an urgent requirement of intubation with a high fraction of inspired oxygen (FiO2) demand, and evidence of sepsis/septic shock) (9) were primarily considered as study samples.
Anemia, bronchial asthma (BA), chronic kidney disease (CKD), chronic obstructive pulmonary disease (COPD), dementia, dyslipidemia, DM, end-stage renal disease (ESRD), hypothyroidism (HT), hypertension (HTN), irritable bowel syndrome (IBS), ischemic heart disease (IHD), myocardial infarction (MI), malignancy (MN), non-alcoholic fatty liver disease (NAFLD), obstructive sleep apnea (OSA), Parkinson’s disease (PD), peptic ulcer disease (PUD), rheumatoid arthritis (RA), renal transplantation (RT), severe obesity (SO) (a condition while the calculated body mass index (weight in kilogram divided by the square meters of height) value is ≥ 40 in adults) (10), stroke, and ulcerative colitis (UC) were recorded as chronic comorbidities in patients of this study. The ethical approval (no. 2004SH-OR021 was obtained from the ‘Research Ethics Committee’ of the Square hospital on April 20, 2020. Informed consent was obtained from all patients, and the study protocol was developed following the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a prior approval by the institution’s human research ethics committee.
3.4. Statistical Analysis
Data were analyzed using SPSS version 22.0 (SPSS, Chicago, IL, USA). Descriptive statistics were presented through median with interquartile range (IQR). Categorical variables were compared using Pearson’s chi-square test, and continuous variables were compared using the one-sample t-test. A P-value of less than 0.05 was considered statistically significant.
4. Results
In total 173 COVID-19 patients with moderate-to-critical symptoms and one or more chronic diseases or comorbidities, were admitted in the “COVID-19 unit”. Among them, 157 (N) patients were finally included in the present study after evaluating against inclusion and exclusion criteria. The mean age of participants was 63 years (IQR: 57 - 69) and the number of male patients was more than 2-fold of females (66.8/33.1, n = 157). All patients were admitted with, on average, a 6-day (IQR: 5 - 8) long history of onset of symptoms of COVID-19. Fever, dry cough, shortness of breath, weakness, diarrhea, anosmia, and sore throat were commonly found as suspected COVID-19 symptoms on hospital admission (Table 1). The median arterial blood oxygen saturation level (SpO2) was 90 (IQR: 88 - 92). The median value of common infection markers, including C-reactive protein, procalcitonin, and white blood cells, was higher than the normal range. Other biomarkers used in COVID-19 infection, including plasma D-Dimer, ferritin, and lactic acid dehydrogenase (LDH) levels, had high median values [2.1 (IQR: 1.5 - 4.3), 581 (IQR: 437 - 732) and 469 (IQR: (398.5 - 589.5), respectively]. The median serum creatinine level was 1.7 (IQR: 0.6 - 2.3), and the median MEWS score was 3 (IQR: 2 - 3) (Table 1).
| Characteristics | Variable |
|---|---|
| Age, y | 63 (57 - 69) |
| Gender, | |
| Male | 105 (66.8) |
| Female | 52 (33.1) |
| Days from onset of symptoms-to-hospitalization | 6 (5 - 8) |
| Fever, °F | 114 (72.6) |
| Dry cough | 157 (100) |
| Shortness of breath | 96 (61.1) |
| Weakness | 157 (100) |
| Diarrhea | 82 (52.2) |
| Anosmia | 94 (59.8) |
| Sore throat | 147 (93.6) |
| SpO2, % | 90 (88 - 92) |
| Respiratory rate, breaths/min | 22 (20 - 25) |
| PaO2/FiO2, mmHg | 238 (195 - 263.5) |
| Heart rate, beat/min | 96 (84.5 - 102.5) |
| CRP, mg/L | 168.2 (42.1-239.6) |
| Procalcitonin, ng/mL | 1 (0.07 - 2.4) |
| WBC, K/µL | 10.3 (7.6 - 15.1) |
| Neutrophils, % | 89.2 (80.1 - 93.4) |
| Lymphocytes, % | 14.3 (11.1 - 16.5) |
| Platelet, k/µL | 219 (159 - 311.5) |
| D-Dimer, mg /L FEU | 2.1 (1.5 - 4.3) |
| Serum Ferritin, ng/mL | 581 (437 - 732) |
| LDH, U/L | 469 (398.5 - 589.5) |
| Creatinine, mg/dL | 1.7 (0.6 - 2.3) |
| ALT, U/L | 72 (56.5 - 94) |
| AST, U/L | 36 (25.9 - 49.8) |
| MEWS | 3 (2 - 3) |
Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase; CRP, C-reactive protein; dL, deciliter; FEU, fibrinogen equivalent units; IQR, interquartile range; K/µL, thousand cells per micro liter; L, liter; LDH, lactate dehydrogenase; MEWS, Modified Early Warning score; mg, milligram; min, minute; mmHg, millimetre of mercury; No., number; ng, nanogram; PaO2/FiO2, ratio of arterial oxygen partial pressure to fractional inspired oxygen; SpO2, peripheral capillary oxygen saturation; U/L, units per liter; WBC, white blood cells; %, percentage, °F, grade Fahrenheit;.
aValues are expressed as No. (%) or median (IQR).
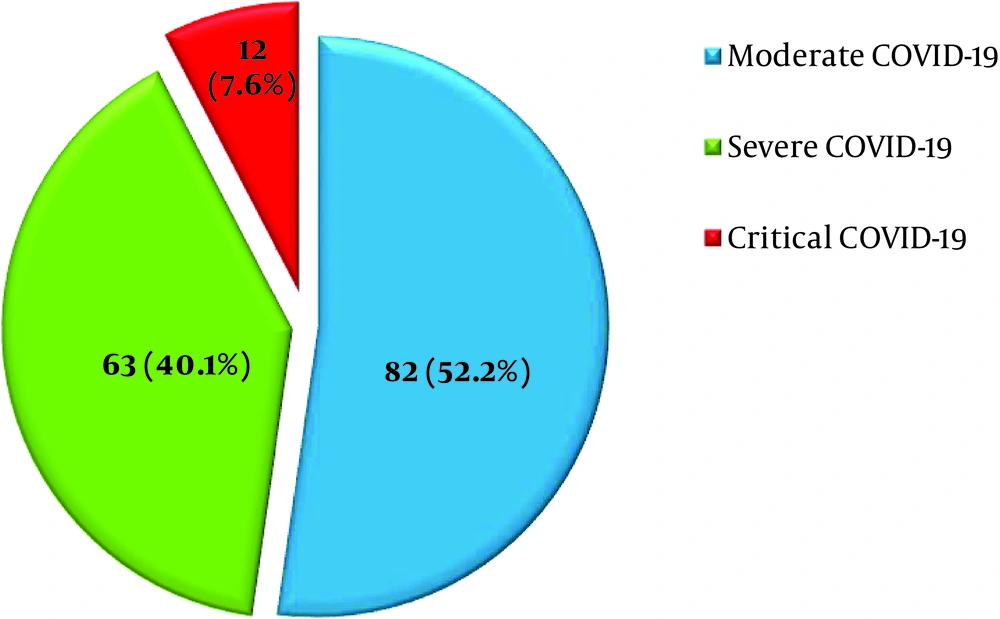
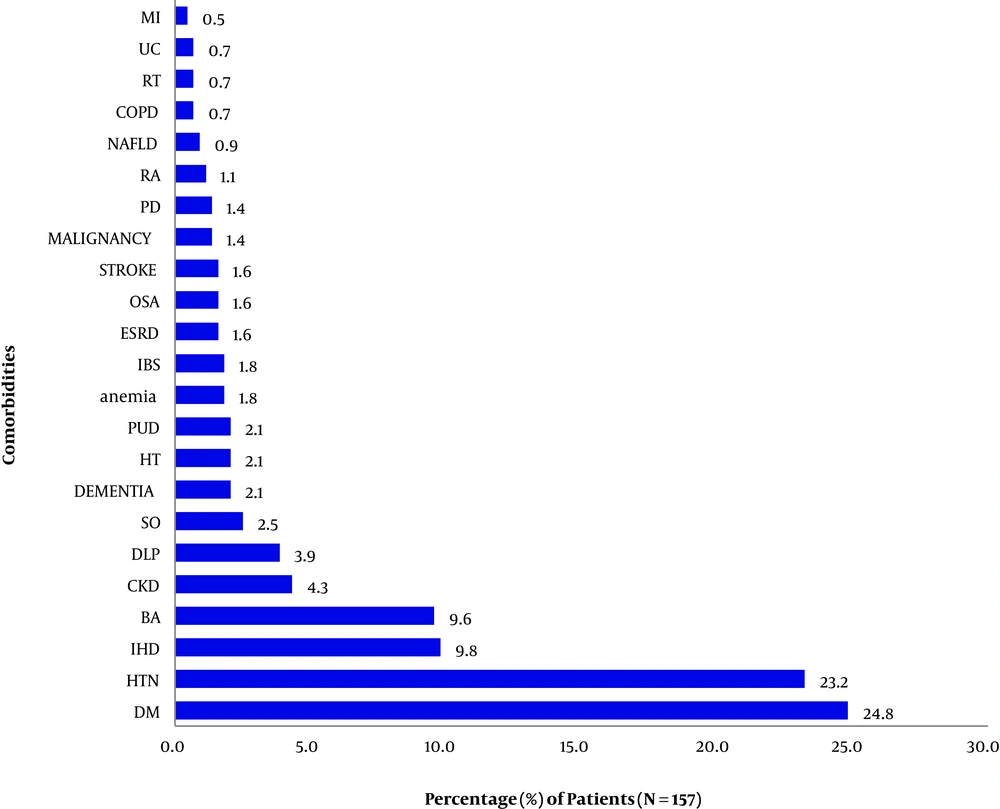
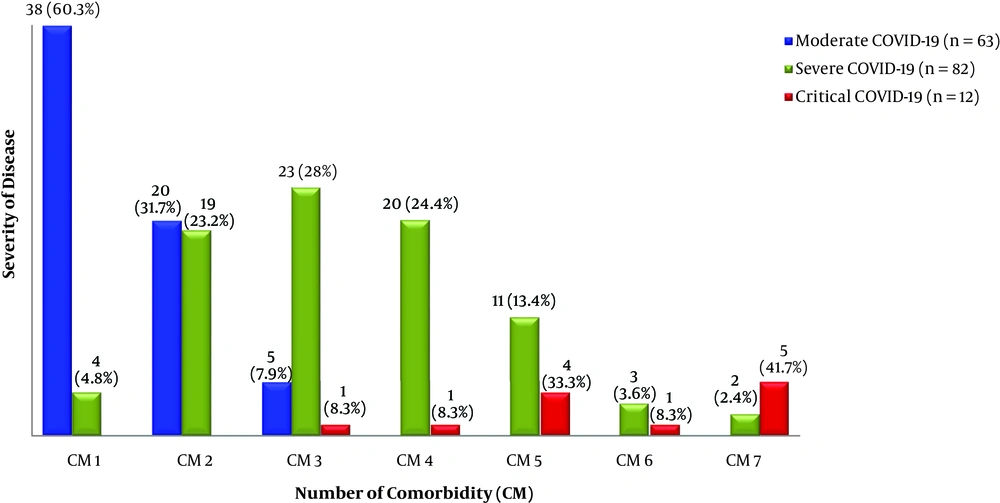
Of 157 patients, more than 50% of participants with one or more comorbidities were admitted with moderate COVID-19 symptoms, while 40.1 and 7.6% of patients had severe and critical symptoms, respectively (P = 0.001) (Figure 2). DM (24.8%), HTN (23.2%), IHD (9.8%), BA (9.6%), and CKD (4.3%) were the most common comorbidities in patients with moderate-to-critical COVID-19. In patients with moderate/severe/critical COVID-19 infection, all patients had at least one (up to 7) preexisting comorbidity. In the present study, patients with 1 and 2 comorbidities developed more moderate COVID-19 symptoms (60.3%/31.7%, n = 82) compared to patients with severe symptoms (4.8%/23.2%, n = 63) (P < 0.05). Also, no patient presented critical symptoms of COVID-19. DM (24.8%), HTN (23.2%), IHD (9.8%), bronchial asthma (9.6%), and CKD (4.3%) were the five top-most prevalent comorbidities, among the 23 chronic diseases found in participating patients (n = 157) during their hospital admission (Figure 3). Also, 8.3% (n = 12) of patients with 3 and 4 comorbidities developed critical COVID-19 symptoms, while 33.3% of patients with 5 comorbidities developed critical COVID-19 symptoms (P = 0.038). Similarly, patients with 7 comorbidities had more critical COVID-19 infection compared to those with 6 comorbidities (41.7 and 8.3%, respectively) (P = 0.001) (Figure 4).
Different comorbidities in patients with moderate-to-critical COVID-19. Abbreviations: MI, myocardial infarction; UC, ulcerative colitis; RT, renal transplantation; COPD, chronic obstructive pulmonary diseases; NAFLD, non-alcoholic fatty liver disease; RA, rheumatoid arthritis; PD, Parkinson's disease; OSA, obstructive sleep apnea; ESRD, end-stage renal disease; IBS, irritable bowel syndrome; PUD, peptic ulcer disease; HT, hypothyroidism; SO, severe obesity; DLP, dyslipidemia; CKD, chronic kidney disease; BA, bronchial asthma; IHD, ischemic heart disease; HTN, hypertension; DM, diabetes mellitus.
Of 157 patients with one comorbidity, 26.7% did not require ICU hospitalization or mechanical ventilation support, and no mortality was observed. There was a positive association between the number of comorbidities and the severity of symptoms. More than 60% of patients with 5 (n = 15), 6 (n = 4), and 7 (n = 7) comorbidities had high requirement of ICU support (5 comorbidities: 73.3%, 6 comorbidities: 75%, and 7 comorbidities: 85.7%, respectively) (P = 0.025) and intubation or mechanical ventilation support (5 comorbidities: 63.6%, 6 comorbidities: 66.7%, and 7 comorbidities: 83.3%, respectively) (P = 0.038). Similarly, the 30-day mortality rate was increased following the increasing number of comorbidities in patients (patients with 4, 5, 6, and 7 comorbidities: 30-day mortality rate was 4.7%, 6.7%, 25%, and 28.6%, respectively) (P = 0.002). In contrast, no mortality was observed among patients with ≤ 3 comorbidities and moderate-to-critical COVID-19 infection within 30-day of onset of infection (Table 2).
| Number of Comorbidity | Patient (N = 157) | Patients Received ICU Support | Patients Required Intubation on MV in ICU | 30-day Mortality Rate |
|---|---|---|---|---|
| 1 | 42 (26.7) | - | - | - |
| 2 | 39 (24.8) | 6 (15.4) | 1 (16.7) | - |
| 3 | 29 (18.5) | 11 (37.9) | 3 (27.3) | - |
| 4 | 21 (13.4) | 17 (80.9) | 6 (35.3) | 1 (4.7) |
| 5 | 15 (9.6) | 11 (73.3) | 7 (63.6) | 1 (6.7) |
| 6 | 4 (2.5) | 3 (75) | 2 (66.7) | 1 (25.0) |
| 7 | 7 (4.5) | 6 (85.7) | 5 (83.3) | 2 (28.6) |
| P-value | 0.001 | 0.025 | 0.038 | 0.002 |
Abbreviations: ICU, Intensive Care Unit; MV, mechanical ventilation; N, total number of patient; No., number of patient; %, percentage.
aValues are expressed as No. (%).
5. Discussion
In this study, moderate COVID-19 (52.2%) was more prevalent than severe (40.1%) and critical COVID-19 (7.6%) in patients with one or more comorbidities. Besides, there was a positive association between the severity of COVID-19 and the number of preexisting comorbidities. For patients with 1 or 2 comorbidities, no critical case was observed, and the prevalence of severe symptoms was higher (up to 28%) in patients with 3 or 4 comorbidities. More than 40% of patients with 5 or more comorbidities (up to 7) developed critical COVID-19 symptoms, while the occurrence of severe infection was comparatively lower (up to 13.4%) with no incidence of moderate COVID-19. Gao et al. (11) found that the presence of one or more comorbidities in patients with avian influenza A (H7N9) was associated with an increased risk of developing acute respiratory distress syndrome by 3.4 times. Similar to H7N9, SARS-CoV, and the Middle East Respiratory Syndrome coronavirus (MERS-CoV), SARS-CoV-2 rapidly causes respiratory failure and death, mostly in patients with preexisting comorbidities (5, 12-17). Huang et al. (18) firstly reported that 32% (n = 13) of patients (out of 41 confirmed COVID-19 cases) had common comorbidities, including HTN, DM, cardiac diseases, and COPD. In previous studies, HTN, CHD, and DM are reported as the most common comorbidities in COVID-19 patients (19-21). A previous study mentioned that the prevalence of respiratory diseases, including bronchial asthma (BA) and COPD, in COVID-19 patients was not significant compared to HTN and DM, and this may be due to under-diagnosis of the respiratory diseases or lack of awareness (5, 22). In patients with avian influenza, SARS-CoV, and MERS-CoV infection, HTN and DM were at the top of the list of common comorbidities in hospital admitted patients, followed by respiratory diseases, cardiac diseases, renal diseases, and malignancies (23-25). In this study, DM (24.8%) and HTN (23.2%) were the most common predisposed chronic diseases in the hospitalized patients with moderate-to-critical COVID-19, followed by IHD (9.8%), BA (9.6%), and CKD (4.3%).
Future studies are needed to extend our knowledge about the association between the predisposed comorbidities and the severity of COVID-19. In the same vein, the poor clinical outcomes of patients infected with H7N9, SARS-CoV, and MERS-CoV associated with cardiac diseases and endocrine disorders were not clearly determined (23-25). In our study, patients with 4 or more comorbidities admitted to the hospital within six-day (median) of onset of symptoms developed severe and critical COVID-19 symptoms more, along with declined clinical outcome and increased number of death, compared to patients with three or fewer comorbidities. Similarly, patients with 1 and 2 comorbidities had mostly moderate COVID-19 (60.3% and 31.7%, respectively), but no incidence of critical COVID-19 was observed. During the MERS-CoV pandemic, no preexisting comorbidities, except for DM, had a clear contributory role in the poor clinical outcomes of the disease (24). A case-control study showed that diabetes and CHD were associated with increased complications of seasonal influenza, and diabetes was identified as the independent risk factor for severe influenza (OR 3.63, 95% CI: 1.15 - 11.51, P = 0.02) (26). To find out the root cause, a diabetic mice-model study demonstrated that profuse inflammation in lungs, dysfunction of immune cells, and overexpression of the inflammatory mediators resulting in massive cytokine storm in MERS-CoV are the basic reasons for serious disease aggravation (27). Immune dysfunction, including damaged neutrophil functions, altered antioxidant system, and humoral immunity, due to chronic hyperglycemia in patients with DM makes the diabetic patients highly susceptible to viral infections, including SARS-CoV-2, which in turn leads to poor clinical outcomes (28). This is why, during the SARS-CoV pandemic in 2003, hyperglycemia was used as an independent biomarker for the assessment of the severity-state of the disease (29). A recent study on the hypertensive cohort found the severity of COVID-19 was increasingly associated with hypertension and diabetes, as the most prevalent comorbidities (6). Similarly, both DM and HTN were found as the most privileged comorbidities in patients of this study and may have a significant contribution in the progression of the severity of the disease. Further studies are highly required to find out the actual mechanism behind the comorbidity-COVID-19 interaction.
Wang et al. (20) reported that 46.4% (n = 64) of patients (out of 138) with COVID-19 had multiple comorbidities, and 72.2% of comorbid patients required ICU hospitalization. Another recent meta-analysis reported that severe COVID-19 was highly associated with the underlying diseases such as HTN (21.1%, 95% CI: 13.0% - 27.2%), DM (9.7%, 95% CI: 7.2% - 12.2%), cardiovascular diseases (8.4%, 95% CI: 3.8% - 13.8%), and respiratory system diseases (1.5%, 95% CI: 0.9% - 2.1%) (4). Here, moderate-to-critical COVID-19 patients admitted with 4 to 7 underlying chronic diseases had poorer clinical outcomes (i.e. increased need for ICU support) (80.9, 73.3, 75, and 85.7, respectively), mechanical ventilation support (35.3, 63.6, 66.7, and 83.3%, respectively), and increased 30-day mortality rate (4.7, 6.7, 25, and 28.6%, respectively) than the patients with ≤ 3 comorbidities (ICU requirement: < 40%; mechanical ventilation support: < 30%; and no mortality). HTN and DM are the most common comorbidities among Bangladeshi citizens (7, 28, 29). So patients with underlying chronic diseases, including DM and HTN, may be at increased risk of severe COVID-19 in Bangladesh. Similarly, in our study, DM and HTN were highly privileged in patients, and there was a positive association between the severity of COVID-19 infection and the number of comorbidities, with remarkable declination in the clinical outcome of patients with moderate-to-critical COVID-19 infection. Randomized controlled trials are highly required to analyze the impact of individual comorbidity in worsening the COVID-19 symptoms. The major limitations of the present study included the small sample size, single-center data, short duration of the study, and no specific impact of individual comorbidity on the severity of COVID-19. Community awareness programs and patient education activities need to be stronger in the communities of Bangladesh to enhance patients’ awareness regarding the exacerbating-risk of COVID-19 infection with multiple chronic diseases, mostly in the elderly population.
5.1. Conclusions
This study demonstrated that underlying chronic diseases in patients with COVID-19 may play a disease-worsening role. In this study, diabetes and hypertension were the most privileged comorbidities in patients with moderate-to-critical COVID-19 infection, and patients with 4 to 7 comorbidities developed more severe symptoms with poor clinical outcome, including increased need for ICU hospitalization and mechanical ventilation, as well as increased mortality rate compared to patients with 3 or fewer comorbidities. Hence, the more the number of comorbidities, the higher would be the risk of progression of the severity of COVID-19 infection.