1. Background
Disinfection is the most important stage in water treatment to prevent waterborne diseases and ensure that the water is free of pathogenic microorganisms (1, 2).
Some waterborne diseases and health risks include intestinal worms, cholera, typhoid, trachoma, schistosomiasis, diarrhea and legionellosis (3). So far, various chemical disinfection methods such as chlorination are widely used (4). This method is inexpensive, has low operating cost in a wide range of pH and adequate residual effect (5-7). Nevertheless, the main disadvantages of chlorination are the reaction with natural organic matter (NOM) and formation of disinfection by products (DBPs). The importance of DBPs includes trihalomethanes (THMs), haloacetic acids (HAA5) and other dissolved organic halogens. If these products are formed after disinfection, they have health risks especially potential human carcinogens even at low concentrations (8, 9). Already more than 500 DBPs are recognized in finished drinking water. The type and quantity of DBP depends on the oxidizing agents used as disinfectants and reaction conditions (10). It was reported that the prolonged consumption of DBPs with low concentration such as trihalomethanes (80 ppb) and haloacetic acid (60 ppb) might make connate cardiac defects (11). For these reasons, scientists attempt to find disinfection technologies that DBPs do not form the least quantity (1).
Ozonation after chlorination greatly promoted the disinfection performance for all pathogens such as viruses, bacteria and parasites as well as removing toxicity and mutagenicity caused to enhance water quality. However, ozonation was restricted due to potentially microorganism regrowth in water distribution networks and high cost of production and operation of ozonation (8).
Consequently, selecting a suitable disinfectant depends on technical dependability, economic and environmental criteria. Many problems involved in water quality could be obviated or greatly improved using products and processes resulting from the progression of nanotechnology and engineering processes (12). In recent years, nanoparticles were focused on a wide range of research areas, especially environmental issues (13).
Nanosilver has antimicrobial properties. It is the most widely product in disinfection processes (14). Three possible disinfection mechanisms of nanosilver are as follows: Bonding of nanoparticles to the outer bacterial cell to alter the function of membrane properties. The nanoparticles have a small size and high specific surface area. These properties will result in a strong contact between nanoparticles and the microorganism surface; DNA damage of bacteria after penetration of nanosilver into the bacterial cell; Ag+ ions release due to dissolution of nanosilver which can interact with sulfur-containing proteins in the bacterial cell wall, which may cause to lose its functionality. This mechanism is often considered as the main mechanism of the antimicrobial activity of nanosilver (15). Also, UV irradiation improved nanosilver inactivation of bacteria, since silver ions are active in the presence of UV irradiation.
Although the antibacterial mechanisms of nanoparticles are somewhat known (16-22), there are still questions about their effectiveness in destroying pathogens and the possibility of their use for water disinfection.
2. Objectives
The current study aimed to determine the effectiveness of a new technology such as combined process of silver nanoparticles and UV light on E. coli inactivation as a water microbial pollution index and effects of some parameters on its efficiency.
3. Methods
3.1. Materials
The catalyst employed in this work was silver nanoparticles (metal basis with 20 nm primary particle size, purchased from USNano Inc.). Escherichia coli ATCC 25922 (PTCC 1399) were purchased from Persian type culture collection company. All reagents were of analytical grade and were used without further purification. Tryptic soy agar (TSA), tryptic soy broth (TSB), barium chloride and sulfuric acid were supplied by MERC Germany. A 6 W UV-C lamp was provided from ARDA Inc. (France).
3.2. Cell culture, Medium Preparation and Bacterial Counting
Escherichia coli species were cultured according to the manufacturer guidelines. Briefly, a single colony of E. coli was taken from refrigerated stock and precultured in 20 mL TSB by incubation at 37°C for 24 hours. Then it was transferred into TSA and incubated for 24 hours at 37°C. The top of each colony was touched with a sterile loop and the growth was transferred into a tube containing 4 to 5 mL of distilled water (23). A McFarland standard 0.5 was used to determine the cell concentrations. The cell density was compared to that of 0.5 McFarland standards using UV/VIS spectrophotometer; an equivalent optical density of 0.1 at 620 nm with regard to the calibrated standard cell suspensions in distilled water (24). Barium sulfate turbidity standard was used to standardize the inoculums density for a susceptibility test, its turbidity was equivalent to that of a 0.5 McFarland standards. A 0.5 McFarland standard was made according to Garcia (25).
To obtain the required cell suspensions, the stock was serially diluted in distilled water. This resulted in a suspension containing approximately 103, 104 and 105 CFU/mL. The standard plating method was applied to confirm the bacterial concentrations. This test was done in triplicates on tryptic soy agar. Samples were plated in triplicate. The colonies were visually identified and counted after incubation at 37°C overnight.
3.3. Experimental Procedure
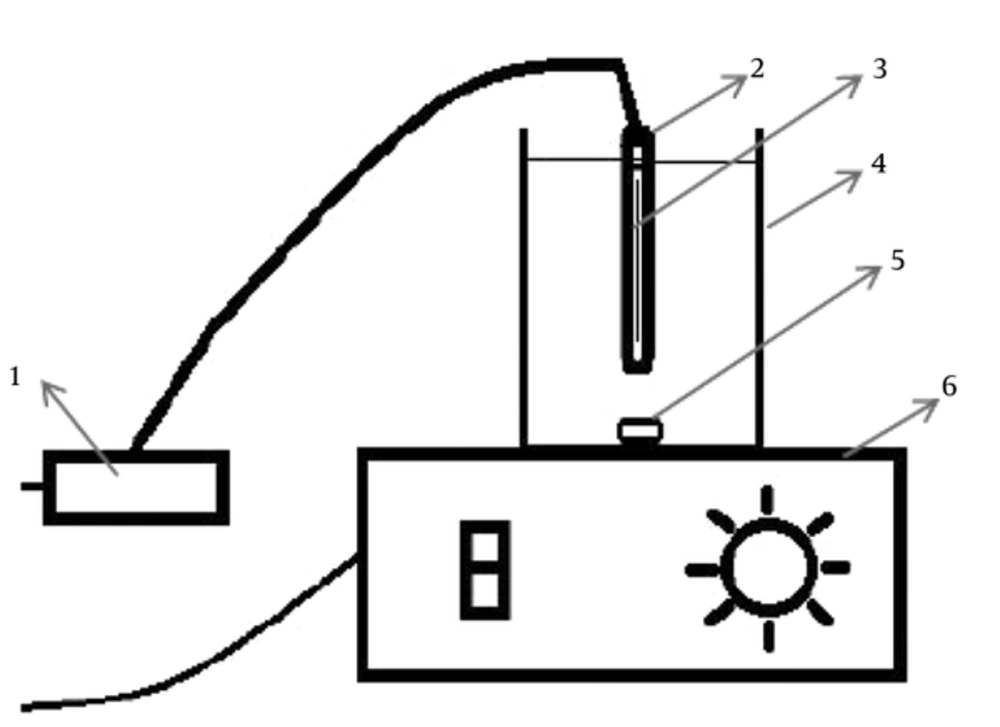
Disinfection experiments were carried out in distilled water. In all instances, glassware was cleaned with distilled water and finally autoclaved. To disperse the AgNPs uniformly in solution, it was suspended in water by pre-sonication for 30 minutes. The characteristics of silver nanoparticles were determined by scanning electron microscope (SEM), transmission electron microscopy (TEM) and X-ray powder diffraction (XRD) analysis. A 6 W UV-C lamp was installed 5 cm above the samples surface. The length of the lamp was 30 cm. The maximum emission of lamp was 254 nm. The light intensity was measured by a radiometer at 200 to 400 nm (Hagner ECL-X) and the intensity was 1.8 w/m2. All samples were analyzed according to the standard methods for water and wastewater examinations (26).
The sterile 250 mL beakers were used as the photochemical cells. The magnetic stirrers were used to stir the samples. Schematic of the used reactors are illustrated in Figure 1. All experiments were done at 20°C. In the first stage, the beakers containing 200 mL of polluted water were contacted with UV light, nanosilver and their combination. In the examinations studied various parameters such as E. coli concentration (103,104,105 CFU/mL), amount of AgNPs (0.05, 0.1, 0.2 and 0.4 mg/L) and time (10, 20, 40 and 60 minutes).
3.4. Statistical Tests
The normality of the data was assessed by Kolmogorov-Smirnov test. Amounts greater than 0.05 were considered as normal data. Then, T-test and ANOVA parametric tests were used for statistical analysis.
4. Results
4.1. Nanoparticles Characteristics
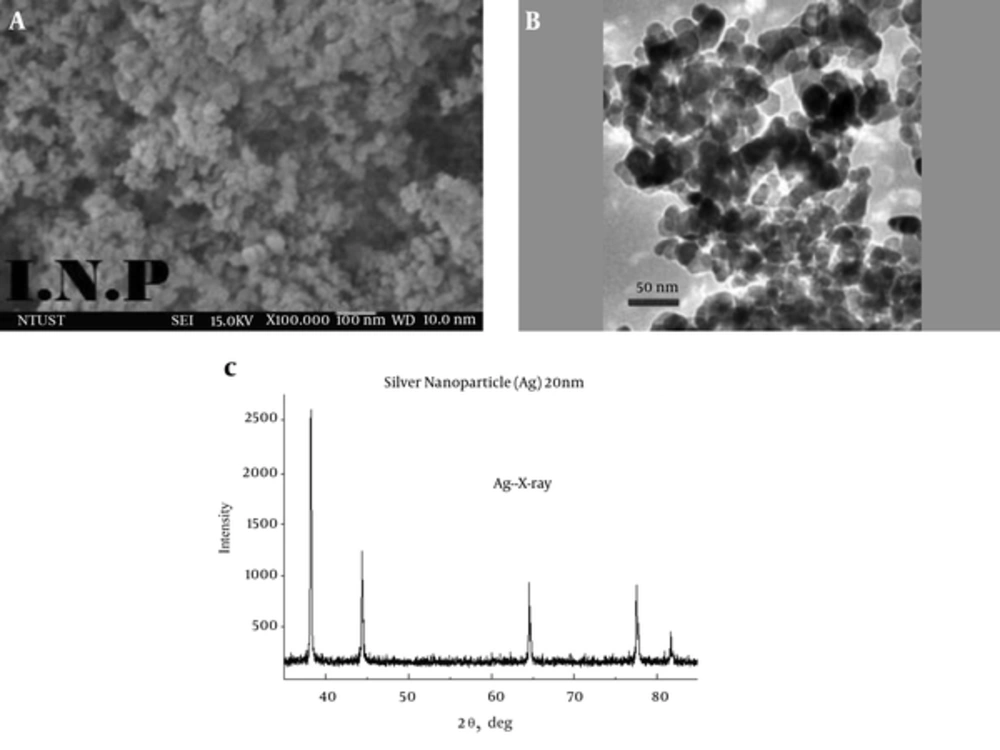
Silver nanoparticles were characterized by SEM, TEM and XRD analysis (Figure 2). Silver nanoparticles had a purity of 99.99%, particle size of 20 nm, the surface area of 18 - 22 m2/g, density of 10.5 g/cm3 and black color.
As can be observed in XRD analysis, the reflection angle was 2θ that confirmed the presence of silver nanoparticles. Also, the TEM images showed that the particles were well dispersed and spherical in shape. The particle size was 20 nm.
4.2. The Kolmogorov-Smirnov Test
To assess the normality of the data, Kolmogorov-Smirnov test was used. The results of the test showed that the data were normal (P > 0.05). Therefore, parametric tests (T-test and ANOVA) were used. Results of the normality test showed P > 0.05, indicating data were normal. Hence, ANOVA was used.
4.3. Effect of UV Irradiation on the Efficiency of AgNPs
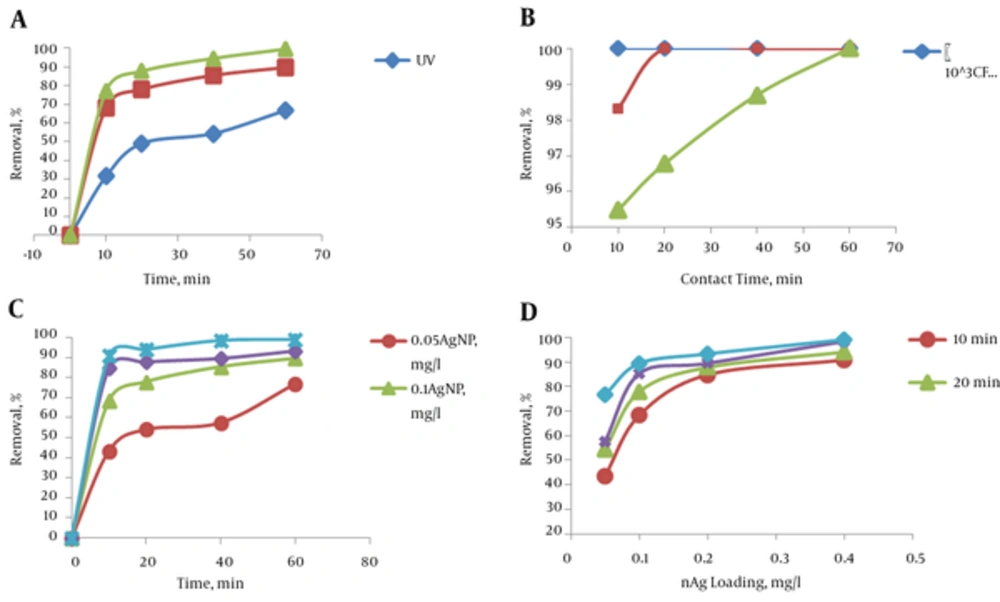
Three series of processes were carried out to perform this experiment. In all of the three experiments a fixed number of colonies per milliliter (105) was used. In the first experiment, the bacteria were exposed to UV light without AgNPs. In the second experiment, the samples were exposed to the AgNPs without UV light. In the third experiment, samples were exposed both to the UV light and AgNPs. A silver nanoparticles concentration of 0.1 mg/L was used. The efficiency of disinfection in all experiments was determined in 10, 20, 40 and 60 minutes. The pH was adjusted at neutral range (7.5 ± 0.5) in all experiments. The efficiencies of bacterial inactivation at different times are illustrated in Figure 3A. As shown in this illustration, the efficiency of UV light was lower than that of AgNPs. Also the combined process (UV + AgNPs) was more effective than only UV light and only AgNP.
a, The effect of AgNPs and UV light on the E. coli inactivation efficiency (initial bacterial population: 105 CFU/mL); b, The effect of initial bacterial population on the combined process (UV + AgNPs), inactivation efficiency of E. coli (AgNPs dosage: 0.2 mg/L); c, The effect of AgNPs dosage on the E. coli inactivation efficiency by the UV light and silver nanoparticles (UV + AgNPs) process (initial bacterial population: 105 CFU/mL) and; d, The effect of contact time on the E. coli inactivation efficiency by the UV light and silver nanoparticles (UV + AgNPs) process (initial bacterial population: 105 CFU/mL)
To ensure that the bacteria population without the presence of light and nanoparticles was changing over time or not, blank sample similar to the real sample was investigated and no changes were observed in the number of bacteria. Due to the lack of inactivation of bacteria, blank sample was not presented in the chart. As shown in Figure 3A, inactivation of E. coli by AgNPs increased in the presence of UV light. This finding indicated that maximum efficiencies in 60 minutes of UV, AgNPs and UV + nAg were 66%, 89% and 99%, respectively.
4.4. Effect of Bacterial Population
The UV light and silver nanoparticles combined process (UV + AgNPs) was performed with different bacterial concentrations (103, 104 and 105 CFU/mL) to study the effect of bacterial population on inactivation efficiency. The pH was adjusted at neutral range (7.5 ± 0.5) in all experiments. The contact time was 10, 20, 40 and 60 minutes. Figure 3B shows the effect of initial bacterial population on the inactivation efficiency of E. coli under optimum conditions. The silver nanoparticles dosage was 0.2 mg/L in these experiments. As shown in Figure 3B, inactivation efficiency was 100% when bacterial populations were lower than 105 CFU/mL.
The results indicated that the inactivation efficiency decreased with increasing the number of colonies. An increase in the bacterial density decreases the chance of exposure to light and AgNPs. Therefore, researchers preferred to continue the experiments with the high number of colonies in such an order that methods could be comparable.
4.5. Effect of Nanosliver Dosage
Experiments were carried out with various silver nanoparticles dosage (0.05, 0.1, 0.2 and 0.4 mg/L) to investigate the effect of AgNPs dosage on the inactivation efficiency of the UV light and silver nanoparticles (UV + AgNPs) combined process. The number of colonies was 104 CFU/mL in these experiments. The results of the experiments are presented in Table 1. According to this tabulation, the inactivation efficiency of processes was 100%, when the AgNPs dosage was more than 0.05 mg/L. Even after 40 minutes, the inactivation efficiency of bacterial was 100% with 0.05 mg/L AgNPs dosage. Therefore, other experiments were performed with more number of colonies.
| Number of Test | Initial Bacterial Population, × CFU/mL | Silver | Time, Mina | |||
|---|---|---|---|---|---|---|
| Nanoparticles Dosage, mg/L | 10 | 20 | 40 | 60 | ||
| 1 | 18 | 0.05 | 50 | 83.33 | 94.4 | 100 |
| 2 | 18 | 0.1 | 89.9 | 100 | 100 | 100 |
| 3 | 18 | 0.2 | 96.67 | 100 | 100 | 100 |
| 4 | 18 | 0.4 | 100 | 100 | 100 | 100 |
The Effect of Nanosilver Dosage on the Efficiency of Ultraviolet Light and Silver Nanoparticles process
The comparison of the effect of different dosages of silver nanoparticles on disinfection efficiency was shown in Figure 3C. The minimum, average, and maximum efficiency of the combined processes with different dosages of AgNPs were presented in Table 2. According to this tabulation, the minimum and maximum of efficiency with 0.05 mg/L of AgNPs dosage were 42% and 82%, respectively. But its comparison with UV light process without catalyst was insignificant (P > 0.05). By increasing the contact time and AgNPs dosage, minimum, average and maximum of efficiency ascends. Efficiency was 100%, when the AgNPs dosage and time were 0.4 mg/L and 20 minutes, respectively.
| Number of Test | Silver Nanoparticles Dosage, mg/L | Contact Time, Min | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 10 | 20 | 40 | 60 | ||||||||||
| Min | Mean ± SD | Max | Min | Mean ± SD | Max | Min | Mean ± SD | Max | Min | Mean ± SD | Max | ||
| 1 | 0.05 | 42.4 | 43.2 ± 0.9 | 44.2 | 52.4 | 54.2 ± 1.8 | 56 | 55.2 | 57.5 ± 2.4 | 60 | 64.8 | 76.8 ± 11 | 86.2 |
| 2 | 0.1 | 52 | 68.2 ± 14 | 77.2 | 66 | 77.9 ± 11 | 87.6 | 68 | 85.3 ± 15 | 94.4 | 70.4 | 89.5 ± 16.5 | 99.3 |
| 3 | 0.2 | 60.8 | 84.7 ± 21 | 97.9 | 68 | 87.8 ± 11 | 98.6 | 70.8 | 89.6 ± 16.3 | 99.3 | 80 | 93.3 ± 11.5 | 100 |
| 4 | 0.4 | 76.8 | 90.7 ± 12.1 | 98.6 | 82.8 | 94.1 ± 9.7 | 100 | 95.6 | 98.5 ± 2.5 | 100 | 97.2 | 99.1 ± 1.6 | 100 |
The Effect of Silver Nanoparticles Dosages on the E. coli Inactivation Efficiency by the Ultraviolet Light and Silver Nanoparticles Processa
4.6. Effect of Time
The effect of contact time (10, 20, 40 and 60 minutes) on UV + AgNPs process efficiency is shown in Figure 3D. According to this tabulation, inactivation efficiency increased by increasing the contact time.
5. Discussion
5.1. Effect of UV Irradiation on the Efficiency of AgNPs
The current study showed that the combined process (UV + AgNPs) was more effective than only UV light and only AgNP. The result of the study corresponded with the data given by Noroozi et al. (27) and Zazouli et al. (28); although they used ZnO and TiO2 nanoparticles, respectively.
Inactivation of E. coli with AgNPs increased in the presence of UV light. The nanoscale particles had a high specific relative surface area; therefore, their contact with bacteria increased and oxidation efficiency immensely improved (13, 29).
In the current study, by increasing the contact time from 10 to 60 minutes, the inactivation of bacteria increased. Therefore, in the presence of UV light and AgNPs, inactivation efficiency increased from 77% in 10 minutes to 99.31% in 60 minutes. Although inactivation efficiency increased by increasing the time, this trend was not very high after 20 minutes. Miranzadeh et al. (19) investigated the effect of silver nanoparticles on the coliforms removal and water disinfection and showed that while increasing the time in all concentrations, the removal of probable coliforms ascended. Their results were in accordance with the findings of the current study.
Miranzadeh et al. (19) showed that after 100 minutes contact in the 0.06 mg/L silver concentration, the maximum removal efficiency was observed (92.41%). Also, ANOVA test showed that a significant relationship between time and E. coli removal. Noroozi et al. (27) studied the photocatalytic removal of E. coli by ZnO. They reported that the efficiency of UV light, ZnO and UV/ZnO processes to inactivate 1000 CFU/mL of E. coli was 70%, 90% and 100%, respectively. Whereas in the current study, the maximum efficiency of UV light process to inactivate E. coli was 66%. The reason for this difference was the number of bacteria. The number of bacteria in the current study was105/mL, but in that of Noroozi et al. (27) was 103/mL.
The comparison of the effect of UV light process, AgNPs, and UV light and silver nanoparticles combined process (UV + AgNPs) showed that the removal efficiency of UV light process was lower than that of AgNPs; and those of the two of them individually were lower than that of combined process. The study on the effect of three processes by LSD test (95% Confidence intervals) showed that the effects of UV light as compared with those of AgNPs and UV + AgNPs were significant (P < 0.05), but the comparison of AgNPs process with UV + AgNPs process was insignificant (P > 0.05). However, disinfection efficiency of combined process (UV + AgNPs) was more than that of only the catalyst (AgNPs). This efficiency difference was about 9% - 10% in all times.
5.2. Effect of Bacterial Population
The results indicated that the inactivation efficiency decreased with increasing the number of colonies. But statistical LSD test showed that their effect was insignificant in two consecutive concentrations (P > 0.05). However, disinfection efficiency with 105 CFU/mL E. coli was lower than 104 CFU/mL and 103 CFU/mL. Disinfection efficiency was 100% when bacterial population was 104 CFU/mL and 103 CFU/mL. The effect of bacterial population was significant only between 103 CFU/mL and 105 CFU/mL (P < 0.05).
The antimicrobial properties of silver compounds and ions were identified many years ago, and were applied to disinfect medical devices, home appliances, and water treatment. But the mechanism of toxicity is still only partially understood. Interaction of silver ions with thiol groups present in proteins, result in inactivation of respiratory enzymes and lead to the creation of reactive oxygen species (ROS). Also, Ag ions inhibit DNA replication and affect the structure of cell membrane and alter its permeability. Moreover, the use of silver ions and UV irradiation together promote inactivation of bacteria and viruses. It is presumed that a combination of Ag and cysteine promotes photodimerization of viral genome contributing to a synergistic effect observed in inactivation of Haemophilus influenzae and MS2 phages (30).
Already, several mechanisms are assumed for the antimicrobial properties of nanosilver: Bonding to the cell surface that leads to altering the functions of the cell membrane properties. It is reported that silver nanoparticles can degrade lipopolysaccharide molecules, accumulate inside the membrane by forming pits, and increase membrane permeability; silver nanoparticles can penetrate into the bacteria cell, and then damage DNA of bacteria; Ag ions are released by dissolution of AgNPs. The antimicrobial activity of AgNPs depends on their physicochemical properties. In other words, physicochemical properties have an important role in antimicrobial activity. Generally, particles that their size is less than 10 nm are more toxic to bacteria such as E. coli and Pseudomonas aeruginosa (30).
5.3. Effect of Nanosliver Dosage
Statistical analysis showed that the efficiency of combined process with 0.05 mg/L of catalyst was not significant, as compared with only UV light process (P > 0.05), but was significant when more catalyst dosage was used (P < 0.05). The contact with bacteria increased; hence, the efficiency increased. The differences between efficiency of combined process with 0.1 and 0.2 mg/L of catalyst were not significant. However, by increasing AgNPs dosage, the inactivation rate of bacteria increased. By increasing the AgNPs dosage, the probability of attachment of nanoparticles to the surface of bacteria, changing its membrane properties, its penetration into the cell and DNA damage and inactivation increase.
Rabbani et al. (31) investigated the disinfection of synthetic polluted water with 30 to 180 mg/L of nanosliver without UV light. They showed that by disinfection of water with 30 to 180 mg/L of nanosilver without UV irradiation, the further coliforms were removed when the concentration of AgNPs was raised, which corresponded with the results of the current study. But they reported no significant correlation between the AgNPs dosage and coliform removal (P = 0.6). However, statistical analysis showed that this relation was significant in the current study.
The effect of nanoparticles on bacteria removal was studied by Gao et al. (18). They reported that regardless of the time, LC50 of nanosilver against the E. coli and Ceriodaphnia dubia was lower than 112.14 and 6.18, respectively. However, in this study, 0.1 mg/L of nanosilver with 10 minutes contact time caused the inactivation of 52% of E. coli species.
5.4. Effect of Time
The current study showed that the inactivation efficiency increased by increasing the contact time. Rabbani et al. (31) showed that by increasing the contact time, the inactivation efficiency increased in all concentrations of AgNPs. Statistical analysis showed that the efficiency of combined process with 0.05 mg/L of silver nanoparticles was significant (P < 0.05) in different times, as compared with those of UV light process. Only between 20 and 40 minutes was not significant (P > 0.05). Also it was not significant at more concentration of AgNPs (P > 0.05). The maximum activity of nanoparticles occurred at 0.05 mg/L concentration and 60 minutes contact time, but based on statistical tests, it was not significant in more dosages. However, it had a rising trend. While the contact time increases, the contact of nanoparticles and bacteria, its penetration into cells, the rate of silver ions production and then the rate of bacterial inactivation increase (15).
5.5. Conclusion
The current study aimed to evaluate the efficiency of the UV light and silver nanoparticles (UV + AgNPs) process in water disinfection. The final results of this study were summarized as follows:
1, The efficiency of the UV + AgNPs process was more than that of only AgNPs catalyst. In fact UV irradiation had a synergistic effect on efficiency of AgNPs. The difference of efficiencies was about 9% - 10% at various times; 2, The minimum, median and maximum inactivation efficiency increased by increasing AgNPs dosage and contact time. As the minimum, median and maximum efficiency at dosage of 0.4 mg/L of AgNPs and contact time of 20 minutes were 82.8, 94.1 ± 9.7 and 100%, respectively; 3, The statistical test showed that the efficiency of the UV + AgNPs process at 0.05 mg/L dosage of AgNPs compared with that of UV light only was insignificantly different (P > 0.05). But it was significant (P < 0.05), when more AgNPs dosage was used. The increase of dosage increased the rate of photoexcitition and radical production. Furthermore, its contact with bacteria and also efficiency increased; 4, While bacterial population increased, the photocatalytic process efficiency decreased. When the bacterial population increased, the contact with light and catalyst and also photocatalytic products decreased.
Finally, it is suggested to evaluate the disinfection efficiency of nanosilver in form of composite or coated on specific materials and zeolites, the comparison between combined process with silver nanoparticles and silver ions, and disinfection process with nanocatalyst compared with non-nanocatalyst. Also there is a need for similar studies on inactivation of microorganisms such as heterotrophic plate count (HPC) bacteria, total coliforms and fecal coliforms and fecal streptococci.