1. Background
Currently, physical activity is recognized as a preventive approach to mitigate the side effects of medical conditions and mental disorders, as well as to support public and mental health. Pemphigus vulgaris (PV) is identified as a chronic immunobullous skin disease that initiates with the loss of adhesion within the epidermal keratinocyte layer, classifying it as a psychocutaneous disease. This condition is generally rare. Zahed et al., in their 2021 study, demonstrated that PV follows a long-term progression, leading to severe morbidity and significant mortality (1, 2). Although systemic corticosteroid therapy has reduced mortality rates, there remains no cure for the disease, which continues to be a major cause of death (1).
In a systematic review conducted by Zhao in 2023, the pooled incidence rate of pemphigus vulgaris was found to be 2.83 per million person-years (95% CI, 2.14 - 3.61). Southern Asia exhibited the highest incidence rate among subcontinents, with more than one study showing 4.94 per million person-years (95% CI, 2.55 - 8.10). The incidence of PV appears not to be influenced by economic levels. Despite its global presence, the distribution of PV is uneven across ethnic and geographical lines. Incidence rates vary, with 0.76 new cases per million per year in Finland, 3.5 in Japan, and up to 16.1 cases per million per year in Jerusalem. Notably, the incidence is highest among Ashkenazi Jews of Mediterranean origin. In central Europe, the estimated incidence rate is 2 new cases per million inhabitants per year, with a higher occurrence in women. PV can develop at any age, typically manifesting between 40 and 60 years. An increase in frequency has been noted among the elderly and children. Interestingly, in some Middle Eastern countries and Brazil, the onset of the disease occurs earlier, with a Brazilian study estimating that 17.7% of cases are present before the age of 30 (2).
The annual incidence of this disease in Iran is 1/100,000, which ranks among the highest globally. Typically, the disease initiates in the mouth before spreading to other body parts. The use of monoclonal antibodies and other systemic medications has enhanced clinical outcomes and patients' quality of life. However, patients often experience anxiety concerning the perceptions of others, and prolonged conditions can lead to depression (3). Several medical conditions, notably chronic diseases like diabetes mellitus, rheumatoid arthritis, and chronic renal failure, may exacerbate patient disabilities, thereby increasing the risk of anxiety and depression (4-7). Depression and anxiety are common mental health disorders, not only in other chronic conditions such as breast cancer but also significantly in patients with Pemphigus Vulgaris (8-10). The definition of quality of life is grounded in an individual's evaluation and satisfaction with their life, along with their perception of physical, mental, and social well-being. Numerous studies have identified a strong correlation between leisure-time physical activity and quality of life (6). Presently, physical activities are acknowledged as a remedy for various physical and mental challenges, contributing to the enhancement of community life quality and fostering moments (5, 11) of happiness and relaxation. Additional research has indicated that individuals across various age groups who lead healthy lifestyles exhibit similar benefits (12-16). Moreover, studies on animals have highlighted the antidepressant and anxiolytic effects of voluntary exercise through serotonergic activity in response to maternal separation stress in rats (17, 18). Jonsdottir et al., discovered that individuals who engaged in leisure-time physical activities reported lower levels of stress, depression, and anxiety compared to those with sedentary lifestyles (19).
2. Objectives
The aim of this study is to examine the role of leisure physical activity as a preventative measure and a tool for health promotion in influencing the quality of life, the severity of clinical symptoms, and the incidence of anxiety and depression in patients with Pemphigus Vulgaris.
3. Methods
The present study was conducted as an interventional study with pre- and post-intervention assessments on 76 patients with Pemphigus Vulgaris at the Razi Hospital of Tehran University of Medical Sciences in 2020. The sample size for this study was determined using G*Power software. The calculation aimed to compare the difference between the means of 2 groups, taking into account a significance level of 0.05, a power of 95%, and an average effect size (f = 0.23) for 2 measurements across the groups with a one-to-one ratio. The required sample size was calculated to be 64. To accommodate a potential 20% loss to follow-up, the final sample size was adjusted to 76 participants.
Inclusion criteria for the study were as follows: Diagnosis of Pemphigus Vulgaris at least 3 months prior, no engagement in professional or regular physical activity in the last three months, capability to participate in physical exercises without any physical, motor, or severe mental impairments, absence of any severe mental disorders or cardiovascular diseases, no history of coronary artery disease, ability to read and write, receiving a maximum of 25 mg of corticosteroids per day, and provision of written informed consent. The Hospital Anxiety and Depression Inventory (HADS) is a self-report screening instrument developed by Zigmond and Snaith (20) for patients referred to hospitals, and it was later standardized by Kaviani et al. (21) for the Iranian population. This tool comprises 14 questions, divided equally between depression and anxiety symptoms. The scoring for depression is categorized as follows: 0 - 6 indicates normal levels, 7 - 8 mild, 9 - 10 moderate, and 11 or higher signifies severe depression. For anxiety, the scoring ranges are 0 - 6 for normal levels, 7 - 9 for mild anxiety, 10 - 13 for moderate anxiety, and 14 or higher for severe anxiety. Based on the studies by Zigmond and Snaith in 1983 (20) and Kaviani et al. (21), the validity of the questionnaire's depression subscale is 0.59 (P > 0.001), and for the anxiety subscale, it is 0.53 (P > 0.001). The questionnaire's reliability is confirmed with a Cronbach's alpha value of 0.85.
Regarding the Dermatology Life Quality Index (DLQI), reliability analysis yielded satisfactory results, with a Cronbach's alpha coefficient of 0.77. Validity assessments included convergent validity, which measured the extent to which the DLQI correlated with global quality of life scores, and construct validity, which was evaluated through factor analysis. The analysis demonstrated a significant number of correlations between items and the hypothesized scale (corrected for overlap) of ≥ 0.4 out of the total number of convergent validity tests (22, 23).
Before the intervention, demographic information of the patients was collected (refer to Table 1). Subsequently, the participants were randomly assigned into 2 groups using the permuted block randomization method. This method was implemented in 2 variations: One with equal block sizes and the other with randomly chosen block sizes, as was the case in our study. All patients completed the HADS and the DLQI questionnaires to assess their quality of life (24, 25). Additionally, the Pemphigus Disease Area Index (PDAI), a checklist used by dermatologists to evaluate the severity of Pemphigus Vulgaris symptoms, was filled out for all participants (26, 27).
| Variables; Subscales | Groups | No. (%) | Mean ± SD | P-Value |
|---|---|---|---|---|
| Gender | - | 0.646 | ||
| Women | Case | 21 (52.5) | ||
| Control | 19 (47.5) | |||
| Men | Case | 17 (47.2) | ||
| Control | 19 (52.8) | |||
| Age (y) | Case | 38 (50) | 43.53 ± 7.36 | 0.376 |
| Control | 38 (50) | 42 ± 7.56 | ||
| Body Mass Index (BMI) | Case | - | 27.45 ± 5.9 | 0.875 |
| Control | 27.25 ± 5.3 | |||
| Education | Case | - | 38.6 | 0.969 |
| Control | 384 | |||
| Marital status | 0.744 | |||
| Single/divorce | Case | 6 (54.5) | - | |
| Control | 5 (45.5) | |||
| Married | Case | 32 (49.2) | ||
| Control | 33 (50.8) | |||
| Number of children | Case | - | 1.68 ± 1.2 | 0.346 |
| Control | 1.45 ± 0.95 | |||
| Duration of disease | Case | - | 55.7 ± 74.7 | 0.487 |
| Control | 46.2 ± 38.6 | |||
| Onset of disease | Case | - | 69.84 ± 81.5 | 0.253 |
| Control | 52.89 ± 39.8 | |||
| Mucosal/cutaneousManifestations | 0.912 | |||
| None | Case | 10 (55.6) | - | |
| Control | 8 (44.4) | |||
| Cutaneous | Case | 12 (52.2) | ||
| Control | 11 (47.8) | |||
| Mucosal | Case | 7 (46.7) | ||
| Control | 8 (53.3) | |||
| Mucocutaneous | Case | 9 (45) | ||
| Control | 11 (55) | |||
| Underlying disease | 0.773 | |||
| Yes | Case | 7 (46.7) | - | |
| Control | 8 (53.3) | |||
| No | Case | 31 (50.8) | ||
| Control | 30 (49.2) | |||
| History of depression | 0.723 | |||
| Yes | Case | 5 (55.6) | - | |
| Control | 4 (44.4) | |||
| No | Case | 33 (49.3) | ||
| Control | 34 (50.7) | |||
| FavoriteActivity in Leisure Time | 0.389 | |||
| None | Case | 3 (33.3) | - | |
| Control | 6 (66.7) | |||
| Active | Case | 29 (49.2) | ||
| Control | 30 (50.8) | |||
| Both | Case | 3 (75) | ||
| Control | 1 (25) | |||
| Passive | Case | 3 (75) | ||
| Control | 1 (25) | |||
| RegularExercise | 0.26 | |||
| Yes | Case | 6 (37.5) | - | |
| Control | 10 (62.5) | |||
| No | Case | 32 (53.3) | ||
| Control | 28 (46.7) | |||
| IndoorandOutdoorExercise | 0.512 | |||
| Out | Case | 17 (50) | - | |
| Control | 17 (50) | |||
| In | Case | 14 (58.3) | ||
| Control | 10(10) | |||
| Both | Case | 6 (46.2) | ||
| Control | 7 (53.8) | |||
| Nothing | Case | 1 (20) | ||
| Control | 4 (80) | |||
| Sleep quality | 0.852 | |||
| Bad | Case | 10 (55.6) | - | |
| Control | 8 (44.4) | |||
| Almost | Case | 6 (46.2) | ||
| Control | 7 (53.8) | |||
| Good | Case | 22 (48.9) | ||
| Control | 23 (51.1) | |||
| Life satisfaction | 1.00 | |||
| Yes | Case | 27 (49.1) | - | |
| Control | 28 (50.9) | |||
| Almost | Case | 4 (50) | ||
| Control | 4 (50) | |||
| No | Case | 7 (53.8) | ||
| Control | 6 (46.2) | |||
| Family loss | 1.00 | |||
| Yes | Case | 8 (50) | - | |
| Control | 8 (50) | |||
| No | Case | 30 (50) | ||
| Control | 30 (50) | |||
| Covid-19 general effect | 0.93 | |||
| No | Case | 27 (51.9) | - | |
| Control | 25 (48.1) | |||
| Approximately | Case | 3 (50) | ||
| Control | 3 (50) | |||
| Yes | Case | 8 (44.4) | ||
| Control | 10 (55.6) | |||
| Willingness of practice | 0.628 | |||
| At all | Case | 1 (25) | - | |
| Control | 3 (75) | |||
| Very Little | Case | 4 (33.3) | ||
| Control | 8 (66.7) | |||
| Little | Case | 5(45.5) | ||
| Control | 6 (54.5) | |||
| Moderate | Case | 19 (59.4) | ||
| Control | 13(40.6) | |||
| Lot | Case | 6 (54.5) | ||
| Control | 5 (45.5) | |||
| Very lot | Case | 3 (50) | ||
| Control | 3 (50) | |||
| Smoking | 0.497 | |||
| Yes | Case | 6 (60) | - | |
| Control | 4 (40) | |||
| No | Case | 32 (48.5) | ||
| Control | 34 (51.5) |
Demographic Characteristics of Patients in the 2 Personal and Medical Groups
After 8 weeks, the HADS, DLQI questionnaire, and PDAI were administered once again. The intervention group participated in walking sessions for 60 minutes three times a week for 8 weeks, either in a park or another outdoor setting. Each session included a 15-minute warm-up before starting the walk and a cool-down period afterward, during which participants performed stretching exercises independently. This adaptation was made due to COVID-19 restrictions (28). To ensure proper adherence to the intervention, participants were regularly instructed on the correct execution of leisure physical activities, including the duration, moderate intensity (ensuring they could comfortably speak while walking), and time recording. Participants were encouraged to engage in their preferred physical activity, reflecting the principle of free choice. This was particularly relevant given the social restrictions and closures of sports clubs and swimming pools during the coronavirus pandemic, leading participants to opt for walking in the park as their chosen activity.
Exclusion criteria for the study encompassed suffering from severe psychiatric illness, coronary heart disease, debilitating illness, unwillingness to continue participation in the research, discontinuation of walking for more than 2 sessions per month, and hospitalization due to Pemphigus Vulgaris. All statistical analyses were conducted using SPSS version 25.
Descriptive statistics, including means, medians, and standard deviations for quantitative variables and frequencies, along with percentages for qualitative variables, were reported. The Kolmogorov-Smirnov test was utilized to assess the normality of data distribution. For comparing 2 quantitative groups, the independent t-test was applied, while the paired t-test was employed for comparing 2 dependent quantitative groups. In cases of non-normal distribution, non-parametric equivalents were used instead. The relationship between 2 qualitative variables was examined using the chi-square test. The last observation carried forward method was employed to handle missing data. All statistical analyses were performed using SPSS software version 25. The level of significance for this study was set at 0.05.
According to the data presented in Table 1, there was no significant difference observed in the demographic characteristics between the control and intervention groups. This indicates that the randomization process was effective in mitigating the impact of confounding variables.
4. Results
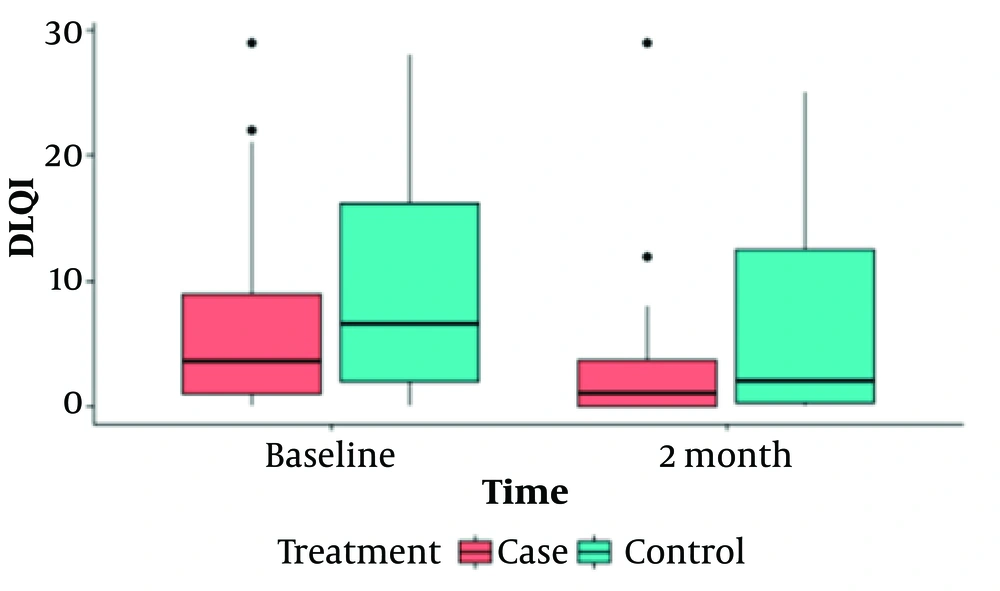
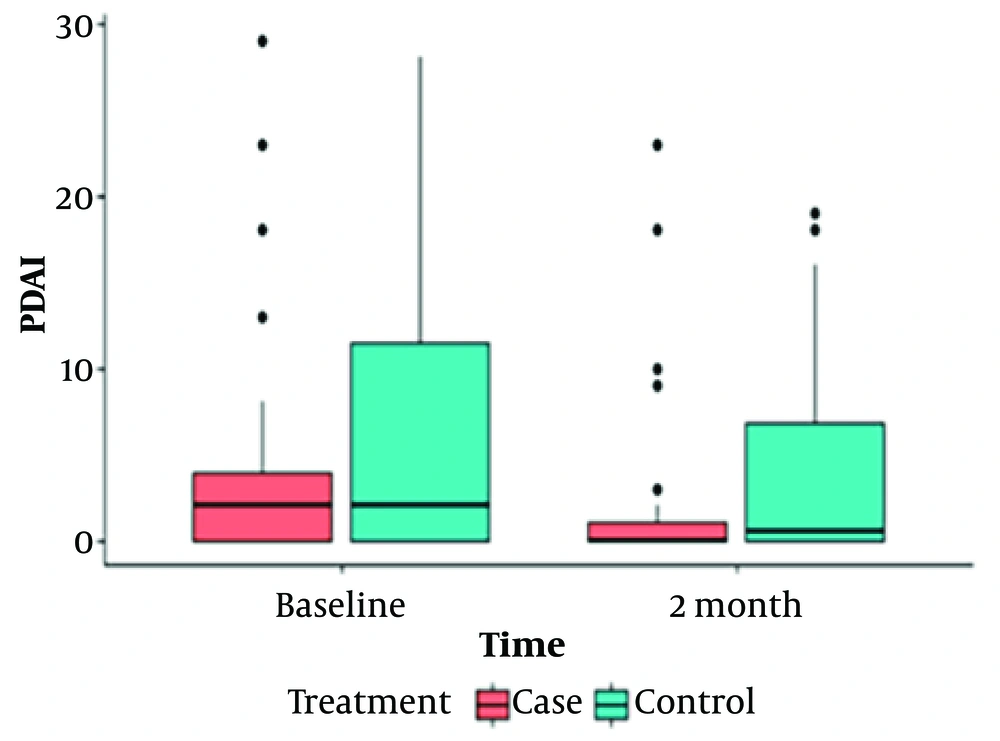
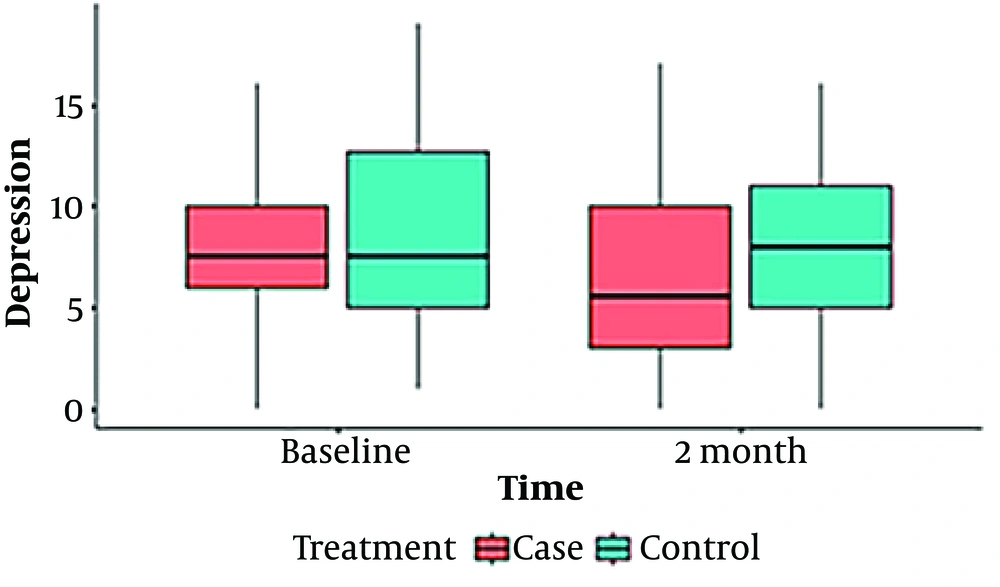
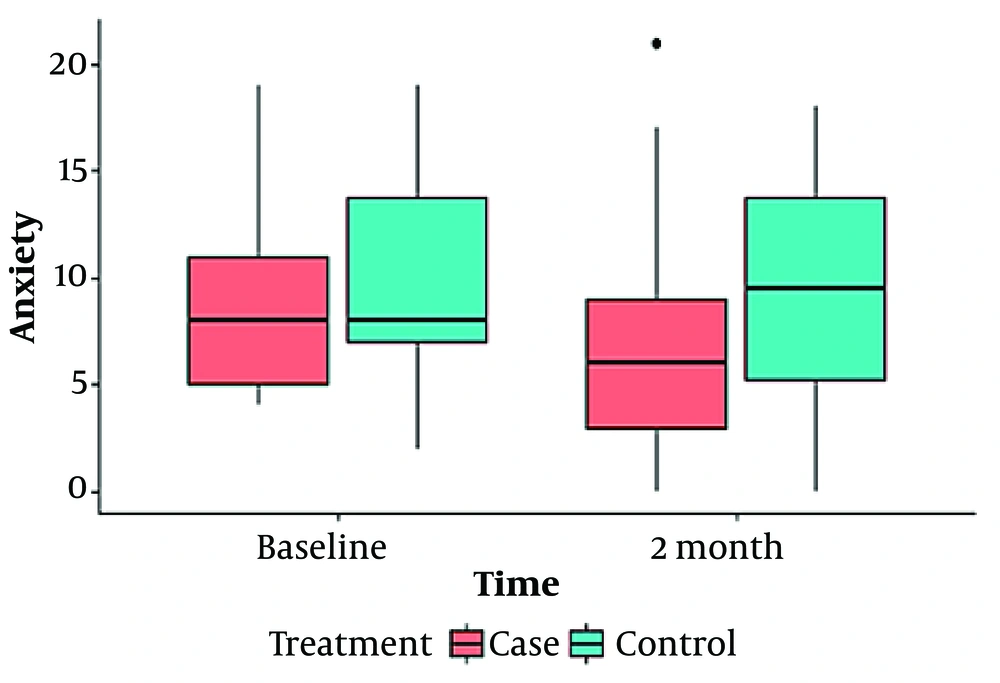
Our findings are shown in 2 tables (Tables 1 and 2) and 4 box plot diagrams (Figures 1 , 2, 3, and 4), as are seen below:
| Responses; Times | Group | Mean ± SD | Median (IQR) | Min-Max | P-Value a | P-Value b | P-Value c |
|---|---|---|---|---|---|---|---|
| Dermatology Life Quality Index (DlQI) | < 0.001 | 0.004 | |||||
| Baseline | Case | 6.42 ± 7.5 | 3.5 (8) | 0 - 29 | 0.163 | ||
| Control | 9.47 ± 9.3 | 6.5 (15) | 0 - 28 | ||||
| After 2 Months | Case | 2.97 ± 5.27 | 1 (4) | 0 - 29 | 0.045 | ||
| Control | 6.45 ± 7.5 | 2 (13) | 0 - 25 | ||||
| Anxiety | 0.03 | 0.704 | |||||
| Baseline | Case | 8.66 ± 4 | 8 (6) | 4 - 19 | 0.345 | ||
| Control | 9.63 ± 4.5 | 8 (7) | 2 - 19 | ||||
| After 2 Months | Case | 6.92 ± 4.7 | 6 (6.25) | 0 - 21 | 0.03 | ||
| Control | 9.31 ± 4.9 | 9.5 (9) | 0 - 18 | ||||
| Depression | 0.004 | 0.36 | |||||
| Baseline | Case | 7.89 ± 3.4 | 7.5 (4) | 0 - 16 | 0.975 | ||
| Control | 8.29 ± 4.5 | 7.5 (8) | 1 - 19 | ||||
| After 2 months | Case | 6.03 ± 4.6 | 5.5 (7.25) | 0 - 17 | 0.067 | ||
| Control | 7.78 ± 4.3 | 8 (6.25) | 0 - 16 | ||||
| Pemphigus Disease Area Index (PDAI) | < 0.001 | 0.004 | |||||
| Baseline | Case | 3.89 ± 6.6 | 2(4) | 0 - 29 | 0.163 | ||
| Control | 6.76 ± 7.9 | 2(13) | 0 - 28 | ||||
| After 2 months | Case | 1.89 ± 4.97 | 0 (1) | 0 - 23 | 0.066 | ||
| Control | 4 ± 6.1 | 0.5 (8) | 0 - 19 |
Comparing the Patient's Response over Time in 2 Groups.
4.1. Demographic Data
In this study, a total of 76 patients, comprising 40 women and 36 men, were randomly allocated into 2 groups. Each group consisted of 38 participants, ensuring equal distribution between the experimental and control groups. The average age of the participants was 42 years, with an age range of 23 to 58 years. Their educational levels varied from high school diplomas to Ph.D. degrees. The number of children among the participants ranged from 0 to 4, and the average duration of the disease was 50 months, with a span from 3 to 324 months. The average time since disease diagnosis among the respondents was 61 months, within a range of 6 to 324 months. Among the participants, 16 had engaged in professional sports activities, with at least 3 months having elapsed since their last activity. The average duration of sports activity reported by the respondents was approximately 48 months, ranging from 3 to 240 months. Further demographic information and data are detailed in Table 1, as presented below.
According to the results presented in Table 2, the anxiety scores in the intervention group showed a significant decrease over time (P = 0.03), whereas the scores within the control group did not exhibit any significant changes (P = 0.704). Prior to the intervention, there was no significant difference in anxiety scores between the 2 groups (P = 0.345). However, a significant difference emerged between them after the intervention (P = 0.03). In terms of depression scores assessed by the HADS questionnaire, there was a noticeable reduction in the intervention group before and after the intervention (P = 0.004), while the control group saw no significant change in this index over time (P = 0.36). Before and after the intervention, the difference between the 2 groups' depression scores was not significant (P = 0.975), followed by a non-significant trend (P = 0.067). Yet, within the intervention group, the depression score significantly decreased during the period. Quality of life, evaluated using the DLQI questionnaire for both groups before and after the intervention, revealed that the intervention group experienced a significantly improved quality of life compared to the control group (P = 0.045), indicating an enhancement in the intervention group's quality of life after 2 months. Both groups witnessed significant changes over time (P = 0.004 for the control group and P = 0.001 for the intervention group), as shown in Table 2.
Figures 1 to 4 offer a graphical summary of our data, covering the Dermatology Life Quality Index, Pemphigus Disease Area Index, depression scaling ranges, and anxiety scaling ranges, respectively, before and after the intervention for both case and control groups at different times.
The intensity of clinical symptoms, as measured by the PDAI checklist before and 2 months after the intervention, showed no significant difference between the 2 groups (P = 0.066). However, both the intervention and control groups experienced significant changes over time. In the intervention group, the average intensity of clinical symptoms decreased from 3.89 to 1.89 (P < 0.001). In the control group, it changed from 6.76 to 4.1 (P = 0.004), as detailed in Table 2.
5. Discussion
International studies conducted during the COVID-19 crisis have confirmed the positive impact of recreational exercise on physical and mental health, particularly when performed in the green spaces of parks (28). Green spaces play a crucial role in enhancing sports activity, as highlighted in the current research protocol, which emphasizes physical activity training in such environments (29). The high incidence of depression and anxiety among patients with Pemphigus Vulgaris has been well-documented in numerous studies (8-10). In 2009, Legrand and Mille investigated the effects of group walking on depressive symptoms in older women, with sessions lasting 60 minutes, 3 to 5 days a week. The results indicated that walking in a group effectively reduced depression, although these findings are not consistent with our own (30). This discrepancy could stem from the solitary nature of our walking method, leading to differing outcomes. The sense of sociability and belonging within a group is a crucial element of recreational sports that contributes to enjoyment. However, adherence to COVID-19 pandemic protocols, such as social distancing, has posed limitations to our study. Jonsdottir found that individuals who engaged in physical activity during their leisure time reported lower levels of depression, anxiety, and fewer psychological issues compared to those leading sedentary lifestyles (19). Hamer et al. demonstrated in 2009 that recreational exercise positively affects mental health, showing a negative correlation between physical activity and depression (31). However, those studies reporting a significant impact of exercise and physical activity on depression do not align with our findings (12-14, 32, 33). This discrepancy may arise from various factors, including the duration of the intervention, pandemic-related conditions and stressors, methodological differences, or variations in the tools used for the study. Our results also corroborate the significant influence of leisure sports activity on reducing anxiety, aligning with the findings of Aguiñaga et al., Finlay, and Hamer et al. (12, 25, 31) and are consistent with a large cross-sectionalstudy from 2018 involving 237,964 participants across 47 countries conducted by Stubbs (34). Although our study differs from Hanaf's qualitative research regarding the modeling of individual participation in leisure sports, we observed all four participation patterns described by Hanaf; some individuals were initially reluctant but chose to engage in leisure sports spontaneously based on perceived benefits and their own capabilities (34). Regarding Pemphigus Vulgaris, our findings indicate a negative correlation between quality of life and levels of anxiety and depression, echoing the observations made by Stubbs et al. (35). Another study has indicated that the quality of life in patients improved through the reduction of anxiety and depression. In 2010, Timóteo et al. conducted a study in Brazil assessing the impact of exercise activities on seven patients with Pemphigus over four months, using the SF-36 quality of life questionnaire in a case-control intervention (36). The study found that physical activity over the months enhanced the quality-of-life components for the patients. Our findings do not align with Timóteo's results regarding the improvement in depression, likely due to differences in intervention methods and durations. However, our results do concur with the positive impact on anxiety. Additionally, Timóteo, in a separate 2019 study, demonstrated that physical activity reduces pro-inflammatory physical symptoms, which are a primary concern for patients (33). Despite this, our study observed a significant change in clinical symptoms when comparing the 2 groups, diverging from Timóteo's second study in 2019 (36). The three-month duration of his study might have offered more opportunity to improve Pemphigus inflammatory markers than our two-month study period.
In an Italian descriptive study assessing the quality of life, depression, and anxiety in 58 Pemphigus patients using the SF-36 questionnaire, it was reported that patients with less severe symptoms had better quality of life scores (23). Contrary to other findings, McRae did not observe a significant effect of a 12-week daily walking regimen on the quality of life in his study subjects (11). However, the comparison conditions between our study and McRae's are not identical. In our study, the physical exercise intervention took place in green spaces and outdoors, whereas McRae's study involved older individuals with chronic illnesses who exercised indoors. Pederson's systematic review on the effects of physical activity across 26 groups of medical and psychiatric conditions, including depression, anxiety, and chronic medical illnesses, suggests that physical activity could serve as an alternative to mild medication (6, 22).
The period of social restrictions due to COVID-19 has prohibited group physical activities, which were limitations in our research. It is suggested that more comprehensive research be conducted after the coronavirus epidemic, with a larger sample size. Additionally, it is recommended to apply the management and planning model of leisure sports through the joint cooperation of various scientific groups across different fields.
5.1. Conclusions
Engaging in eight weeks of regular physical activity, such as solitary walks in the park by patients with Pemphigus Vulgaris, in comparison to a control group that only received medication and engaged in normal life activities, resulted in a significant improvement in quality of life (QoL) and a reduction in anxiety symptoms. However, no significant difference was observed in the reduction of depressive symptoms between the 2 groups. This discrepancy may be attributed to the duration of the study, suggesting that more time is needed to witness a significant change in depressive symptoms; 2 months of physical activity may not have been sufficient. Despite this, the experimental group did show a noticeable reduction in depressive symptoms over time. Additionally, our findings indicate no significant difference in the severity of symptoms post-intervention, possibly due to the need for a longer period to observe clinical symptom improvement; again, 2 months of physical activity might have been inadequate. Nonetheless, the critical takeaway is the significant positive impact of this intervention on the quality of life of these patients, who often experience concerns about their appearance and may have lower energy levels than the general population. Therefore, we recommend that patients with Pemphigus Vulgaris, particularly those with mild to moderate symptoms, consider engaging in moderate-intensity, enjoyable leisure exercises, with physician approval, in addition to medication, rather than as a substitute for pharmacological treatment.