1. Background
As of December 31, 2019, the novel coronavirus, also known as severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), was reported for the first time by the World Health Organization (WHO) as a cause of a multitude of pneumonia cases in the city of Wuhan, China. A few months later, the WHO officially declared coronavirus disease 2019 (COVID-19), caused by SARS-CoV-2, a pandemic. In contrast to other coronaviruses, which usually cause minor respiratory infections, this unique virus strain has high pathogenicity and capacity to cause life-threatening respiratory syndromes (1, 2).
Angiotensin-converting enzyme 2 (ACE2) receptors are key factors associated with the virus's transmissibility. These receptors are expressed in many extrapulmonary cells, including hepatocytes and cholangiocytes. As more cases were diagnosed, physicians became aware of the multisystem presentation of COVID-19. In some reports, the liver is not an exception to this rule, with more than 50% of infected patients having detectable abnormal liver biochemistries (2, 3). As known, COVID-19-associated liver injury includes direct damage to cholangiocytes, indirect damage caused by the drugs used for treating SARS-CoV-2 infection, systemic inflammatory response, and sepsis, in addition to multiorgan dysfunction. Old age, male sex, elevated body mass index, and pre-existing liver diseases (especially alcoholic liver disease, non-alcoholic liver disease, and chronic hepatitis) have also been associated with abnormal liver function tests (LFT) results (4, 5).
2. Objectives
This study aimed to examine the prevalence, behavior, risk factors, and mortality associated with hepatic involvement in admitted COVID-19 patients in a large academic medical center in Jeddah, Saudi Arabia.
3. Methods
3.1. Study Design and Setting
In this retrospective cohort study, we reviewed the data of 2,319 consecutive patients diagnosed with COVID-19, confirmed by polymerase chain reaction (PCR) testing, who were seen at King Abdulaziz University Hospital (KAUH) from April 2020 to November 2020. Informed consent was not obtained from the patients, as this was a retrospective cohort review study. This study was approved by the research ethics committee of the university hospital (Reference No. 491-20).
In this study, COVID-19 was defined by the presence of respiratory symptoms or fever, in addition to the confirmed PCR test results for SARS-CoV-2. We included all admitted and non-admitted COVID-19 patients aged > 18 years during the study period.
3.2. Data Collection
Data were collected from the electronic medical records of the patients. The primary outcome was the prevalence of hepatic injury among the study population. The secondary outcomes were morbidities and mortalities associated with hepatic injury. Demographic data of all patients, including age, sex, and nationality (Saudi or non-Saudi), were collected. Baseline laboratory investigations included complete blood count with white blood cell count (normal range, 3 - 11 KU/L), hemoglobin (Hb; normal range, 12 - 17 g/dL), platelet count (plat; normal range, 100 - 400 KU/L), and International Normalized Ratio (INR normal range, 0.85 - 1.2). The LFT results were collected at baseline, 24 - 48 h after hospital admission, and before mortality or discharge. The LFTs were assessed using the Dimension Clinical Chemistry System Flex reagent (cartridge serum ALT [normal range, 19 - 49 U/L], AST [normal range, 15 - 37 U/L], alkaline phosphatase [normal range, 50 - 136 U/L], gamma-glutamyl transferase [normal range, 5 - 85 U/L], albumin [normal range, 35 - 50 g/L], and total bilirubin [normal range, 0 - 17 µmol/L]). For all patients who underwent LFTs at baseline, Saudis and non-Saudis were compared in terms of hospital admission rate, elevated serum ALT, morbidity, and mortality. In addition, serum ALT levels at baseline were compared between male and female patients, and the ALT difference between admitted and non-admitted patients was recorded. Moreover, the difference in the serum ALT levels between survivors and those who died in the hospital was noted.
3.3. Statistical Analysis
Statistical analyses were performed using SPSS version 22.0 software (IBM Corp., Armonk, NY, USA). Means and standard deviations (SDs) were used to summarize continuous variables, while categorical variables were described using proportions. The chi-square test was employed to compare categorical data. The student's t-test was used to compare the difference in the mean serum ALT level at baseline between men and women, admitted and non-admitted patients, and the deceased and survivors. The paired sample t-test was also used to compare the difference in serum ALT levels at baseline and 24 - 48 h after admission, in addition to the difference in serum ALT levels at baseline and discharge or mortality (final outcomes). Significance was set at P < 0.05.
4. Results
4.1. Demographic Data and Patient Characteristics
During the study period, 2,319 patients were diagnosed with COVID-19, of whom 1,356 (58.5%) were men and 1,328 were non-Saudis (57.3%). The mean (± SD) age was 41.67 ± 18.3 years. A total of 1,022 (44.1%) patients had moderate-to-severe COVID-19 symptoms that required hospital admission, while the rest had home or quarantine center isolations. Besides, LFTs and basic blood work were performed in 1,226 patients. These included 913 patients who required admission and 213 who did not.
Chronic liver disease (CLD) was reported in 23/1022 (2.25%) patients admitted to the hospital. Nonalcoholic fatty liver disease (NAFLD) was the most common etiology of CLD in 12/23 (52%) patients. Viral hepatitis was the second most common infection in 8/23 (34.7%) patients. In addition, two patients had secondary thalassemia and sickle cell anemia with hemosiderosis, and one had progressive familial intrahepatic cholestasis. All patients with CLD had a stable compensated disease, except for one with NAFLD, who had decompensated cirrhosis.
In 185/1022 (18%) admitted cases, the ICU transfer was required. Male patients were more likely to be transferred to the ICU than female patients [136 (73.5%) vs. 49 (26.5%); P < 0.001]. Non-Saudi patients were more likely to develop the severe disease as reflected by hospital and ICU admissions, in addition to higher morbidity and mortality (P < 0.001) (Table 1). Non hepatic comorbidities at presentation were reported in more than one-third of the patients (n = 774). The presence of comorbidities was associated with hospital admission in 602/1022 (61.5%) admitted patients (P < 0.001). The most common comorbidity was diabetes with or without hypertension, encountered in 367 (47.5%) admitted patients. The second common comorbidity was isolated hypertension, reported in 107 (13.8%) patients.
| Saudi, 991 Patients | Non-Saudi, 1328 Patients | Total | P-Value | |
|---|---|---|---|---|
| Sex | < 0.001 | |||
| Male | 513 | 823 | 1336 | |
| Female | 478 | 505 | 983 | |
| Hospital admission | 316 | 706 | 1022 | < 0.001 |
| Morbidity on admission | 263 | 511 | < 0.001 | |
| ALT above normal at baseline | 288 | 509 | 797 | < 0.001 |
| ICU admission | 27 | 158 | 185 | < 0.001 |
| Morbidity | 263 | 511 | 774 | < 0.001 |
| Mortality | 26 | 156 | 182 | < 0.001 |
4.2. Laboratory Results
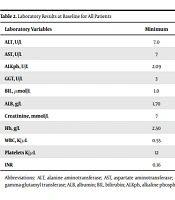
The results of baseline laboratory tests varied, ranging from normal liver enzymes and bilirubin in non-admitted patients to severe, mainly hepatocellular, injury in patients with severe COVID-19 symptoms, particularly in those who underwent ICU transfer. Moreover, deceased patients experienced severe elevation of all liver enzymes (Table 2). Apart from eight patients with chronic viral hepatitis, all patients with liver injury had negative viral hepatitis serologies. Liver injury mainly showed a hepatocellular pattern. Elevated serum ALT levels at baseline were reported in 797 (34.4%) patients. The baseline serum ALT level was higher in men than in women (67.6 ± 202 vs. 39.6 ± 46.5; P = 0.001). Besides, ALT was also significantly higher in admitted patients than in non-admitted patients (60 ± 174 vs. 39.4 ± 36.2; P = 0.001).
| Laboratory Variables | Minimum | Maximum | Mean ± Standard Deviation |
|---|---|---|---|
| ALT, U/L | 7.0 | 3300.0 | 55.985 ± 158.0546 |
| AST, U/L | 7 | 4891 | 60.40 ± 229.279 |
| ALKph, U/L | 2.09 | 997.00 | 97.6718 ± 85.72170 |
| GGT, U/L | 3 | 1107 | 74.06 ± 106.246 |
| BIL, µmol/L | 1.0 | 353.0 | 11.067 ± 17.2237 |
| ALB, g/L | 1.70 | 615.00 | 35.8930 ± 33.00484 |
| Creatinine, mmol/L | 7 | 2100 | 123.57 ± 171.761 |
| Hb, g/L | 2.50 | 690.00 | 13.5025 ± 21.64563 |
| WBC, K/µL | 0.55 | 383.00 | 8.5251 ± 17.27660 |
| Platelets K/µL | 12 | 946 | 248.67 ± 113.854 |
| INR | 0.16 | 5.68 | 1.1959 ± 0.43390 |
Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase; INR, international normalized ratio; WBC, white blood cell; Hb, hemoglobin; GGT, gamma-glutamyl transferase; ALB, albumin; BIL, bilirubin; ALKph, alkaline phosphatase.
Of 797 patients with elevated ALT at baseline, 633 (79.4%) had hospital admissions versus 164 (20.6%) who did not have hospital admissions (P = 0.023). Additionally, patients who had baseline elevated serum ALT had a higher chance of ICU transfer [103/185 (55.7%) patients who had ICU transfer compared to 82/185 (44.3%) patients who had normal ALT at baseline; P < 0.001]. Like ALT, elevated AST and low albumin levels significantly correlated with older age (P = 0.04, 0.004, and 0.011, respectively).
A progressive elevation in serum ALT was observed among patients who had follow-up LFTs during admission [mean ALT at 24 - 48 hours was 105 U/L and mean ALT at the time of discharge or death was 138.12 U/L, with P < 0.001 compared to baseline for both]. Most of these patients had moderate to severe disease and required a prolonged hospital stay.
4.3. Mortality
The mortality rate among admitted patients was 17.8% (182/1022) versus 7.8% (182/2319) among all patients. Mortality was higher among male patients than among female patients [111 (61%) vs. 71 (39%); P = 0.23], and older age was associated with higher mortality (59.6 ± 18 vs. 47.1 ± 18.4; P < 0.001). Interestingly, deceased patients had higher baseline serum ALT levels on admission than survivors (108 ± 389.4 vs. 47 ± 47; P = 0.044).
Among mortality cases, comorbidities on admission were reported in 143/182 (78.6%) patients (P < 0.001). On multiple regression analysis, background pattern, sex (P = 0.014), nationality (P = 0.046), age (P < 0.001), comorbidity (P = 0.013), ICU transfer (P < 0.001), AST (P = 0.008), Hb (P = 0.008), and INR (P < 0.001) were significantly associated with mortality. Among patients with CLD, four died from COVID-19 complications. No significant difference was found in the mortality and morbidity rate among patients with CLD compared with patients without liver disease.
5. Discussion
We found that more than one-third of patients with COVID-19 had associated liver injury. Similarly, previous studies have shown that patients with COVID-19 may experience the involvement of other systems, including liver damage, regardless of the respiratory symptoms (2, 3). Among recent publications about COVID-19, many studies have investigated hepatic involvement, usually represented by abnormal LFTs, including ALT and AST, and this is similar to our findings of hepatocellular injury (3, 6, 7). Analysis of patients’ data showed that abnormal hepatic enzyme levels were associated with disease severity (4, 5, 8, 9). Saudi Arabia and Middle-East countries are among countries with the highest obesity and NAFLD prevalence (10). In addition, viral hepatitis has an intermediate prevalence in Saudi Arabia. These factors may suggest a higher chance of COVID-19 liver injury, and they raise the relevance of liver enzyme interpretation for further therapeutic actions in patients with COVID-19 (11-13). Irrespective of liver function on admission, Fan et al. found that liver enzymes were high 7 - 11 days after admission of patients with COVID-19 (14). Zhang et al. also found that the enzymes in patients with COVID-19 were significantly higher than those in patients with community-acquired pneumonia (15).
In this study, the prevalence of severe COVID-19 was significantly higher among patients with hepatocellular injury than in those with normal hepatic tests on admission. Elevated ALT levels were also significantly associated with a higher likelihood of hospital admission. Moreover, the baseline elevation of serum ALT levels was associated with an increased rate of ICU transfer compared with normal ALT levels at baseline. This is similar to the findings from previous studies (4, 6, 8, 9).
This cohort study observed a relatively small number of CLD cases, predominantly among NAFLD patients. Besides, patients with CLD were comparable with those without CLD in terms of disease severity and mortality. However, pre-existing CLDs on admission and abnormal liver chemistries were reported to be associated with severe COVID-19 (4, 9). In early studies, COVID-19 patients with NAFLD showed rapid disease progression and extended periods of viral shedding compared with those without NAFLD. Moreover, NAFLD also increases the hepatotoxicity of certain medications, such as acetaminophen, which may exacerbate COVID-19 liver chemistries (16). Previous reports showed that the prevalence of the pre-existing liver disease is variable in patients with COVID-19 (3, 6, 9).
This study found an association between male sex and COVID-19. Besides, 71% of COVID-19 cases were recorded in male patients, compared with only 39% in female patients. These findings are supported by various population-based and epidemiological studies from other countries.9,18 Our findings showed that the ALT and AST levels were higher in male patients than in female patients (5, 9). Furthermore, higher ALT levels correlated with older age. Feng et al. assumed that older age implied an increased risk of liver impairment, given that children with COVID-19 had no liver damage (12). By contrast, Xie et al. did not find any age differences between people with and without hepatic injury (17). However, Nikpouraghdam et al. showed that COVID-19 is more common among adult male individuals (median age 34 - 59 years) (18).
Compared with previously published reports, the mortality rate was 17.8% among hospitalized patients. Previous similar reports showed higher mortality rates among patients with COVID-19 who had hospital admissions from China.8 Other reports from the United States showed low mortality among on-ICU patients (19). In addition to that and similar to our findings, comorbidities, and ICU admission were associated with a higher mortality rate (8). Moreover, in our cohort, deceased patients had higher baseline serum ALT levels on admission than survivors, which is similar to the finding from previous studies on liver injury in COVID-19-infected patients (1, 6, 9).
5.1. Study Limitations
The study's retrospective nature is a limitation because the data collection and the time of performing LFT were not preplanned and standardized for all patients. Moreover, the number of patients with CLD was small; thus, definite conclusions can be drawn from the results obtained about COVID-19 in patients with pre-existing liver disease.
5.2. Conclusion and Perspectives
This study indicated that men and adults accounted for the majority of COVID-19 cases in the study hospital. Abnormal liver chemistries in patients with COVID-19 were common and related to adverse clinical outcomes. Therefore, clinicians need to monitor liver biochemistries closely and use medications with the lowest hepatotoxicity in these patients sufficiently and cautiously. Future multicenter studies on patients from the Middle East with pre-existing CLD who had COVID-19 infection will guide the care of that category of patients.