1. Context
In patients with cirrhosis, spontaneous bacterial peritonitis (SBP) is a frequent and serious consequence that can be fatal (1). Bacterial translocation from the intestinal flora to the mesenteric lymph nodes is the first stage in the pathophysiology of SBP (2). In liver cirrhosis, there is a clear increase in gut permeability and small intestinal bacterial overgrowth (SIBO), both of which can promote bacterial translocation (2, 3). Through a variety of causes, including weakened immunity brought on by a reduction in the reticuloendothelial system’s phagocytic activity, a lack of complement, and neutrophil dysfunction, cirrhotic individuals are more vulnerable to infections (4). Reduced gastric acidity leads to an increase in bacterial growth in the stomach and small intestine. Gastric acidity is a defense mechanism against ingested germs. Enteric infections become more likely as a result (5).
Strong stomach acid inhibitors known as proton pump inhibitors (PPIs) have been linked to a higher risk of developing enteric infections brought on by different enteropathogens, such as Salmonella, Campylobacter, and Clostridium difficile (6, 7). Additionally, some results raise doubts about the relationship between PPI usage and the emergence of SBP in cirrhotic patients with ascites (8-10). The higher prevalence of enteric infections linked to PPI medication has been explained by a variety of reasons. In vitro, reduction of neutrophil function, altered microbial flora, increased small intestine overgrowth, and delayed stomach emptying are a few of the aforementioned results (6).
Proton pump inhibitor metabolism (except for rabeprazole) might be greatly hindered in advanced cirrhosis, which might have an impact on the infection risk associated with PPI use. As a result, higher exposure to PPIs might occur (11). Proton pump inhibitors work well and are well tolerated. In several acid-related illnesses, they are widely used and possibly misused (12-14). There is evidence of PPI usage in cirrhotic individuals in the literature (15, 16). The term “hepatic encephalopathy” (HE) refers to a variety of neuropsychiatric symptoms connected to both acute and long-term liver impairment (17, 18). Proton pump inhibitor abuse is widespread among cirrhotic patients. Proton pump inhibitor misuse, however, can potentially result in uncommon but severe negative effects.
Proton pump inhibitor use has been linked to an increased risk of HE in patients with liver impairment, according to previous studies (19). The most thoroughly researched adverse effect is the link between PPI use and the occurrence of SBP or HE in cirrhotic patients. Some studies found the link; however, there was no such link in other studies.
2. Objectives
The aim of the current systematic review and meta-analysis was to evaluate the association between PPI administration and the occurrence of SBP or HE in cirrhotic patients.
3. Data Sources
A systematic search of sources was conducted in order to evaluate any relationship between PPI and the risk of SBP or HE. Medline, ScienceDirect, Scopus, Ovid, ProQuest, Google Scholar, and Web of Science were searched to find any evidence in this regard from 1980 to November 2021. Iranian information databases included IranMedex, Barakat Knowledge Network System, Magiran, Scientific Information Database (SID), and Iranian Research Institute for Information Science (IranDoc). Grey literature and conference articles were also searched (Appendix I).
Search keywords were liver diseases, liver cirrhosis, hepatic cirrhosis, proton pump inhibitors (PPIs), spontaneous bacterial peritonitis (SBP), hepatic encephalopathy (HE).
PICO Definition of the Study:
P: Patients with liver diseases, including liver cirrhosis
I: Proton pump inhibitors
C: Placebo or no treatment
O: Outcome: Primary outcome was the occurrence of SBP and HE. The secondary outcomes were the occurrence of gastrointestinal bleeding, hepatorenal syndrome, mortality, and SIBO.
4. Study Selection
4.1. Inclusion Criteria
All studies that evaluated the association between PPIs and the occurrence of SBP or HE, or other complications in cirrhotic patients.
4.2. Exclusion Criteria
Animal studies, non-English language studies, and studies that did not have the required quality.
4.3. Screening Selection and Quality Assessment
The articles were evaluated by two individual researchers according to PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-analyses). After deleting the duplicates, first, the titles of the studies were evaluated, and then the whole content of the articles was studied. Any disagreement between the two researchers was solved by discussion or a third reviewer. This process was managed through EndNote 8 software.
The evaluation of the quality of the articles was carried out by one researcher, and the second researcher evaluated the articles accidentally. Appropriate Critical Appraisal Checklists of Joanna Briggs Institute (JBI) were used for the critical appraisal of the eligible studies.
5. Data Extraction
Data extraction was performed through the modified JBI data extraction table. The extracted data included the year of issue of the article, authors, country, period of study, type of study, number of cases, mean age of patients, male-to-female ratio, outcome, type of drug, and side effects.
5.1. Data Synthesis and Analysis
Statistical analysis was performed by CMA software (version 2.0), and a P-value of less than 0.05 was considered a significant level. For this purpose, the heterogeneity of the studies was evaluated by Cochrane (Q) and I2, measuring the percentage of differences. If the amount of I2 was less than 50% constant, the Mantel-Haenszel method was used, and if it was more than 50% or P-value < 0.05, a random effect model was used.
6. Results
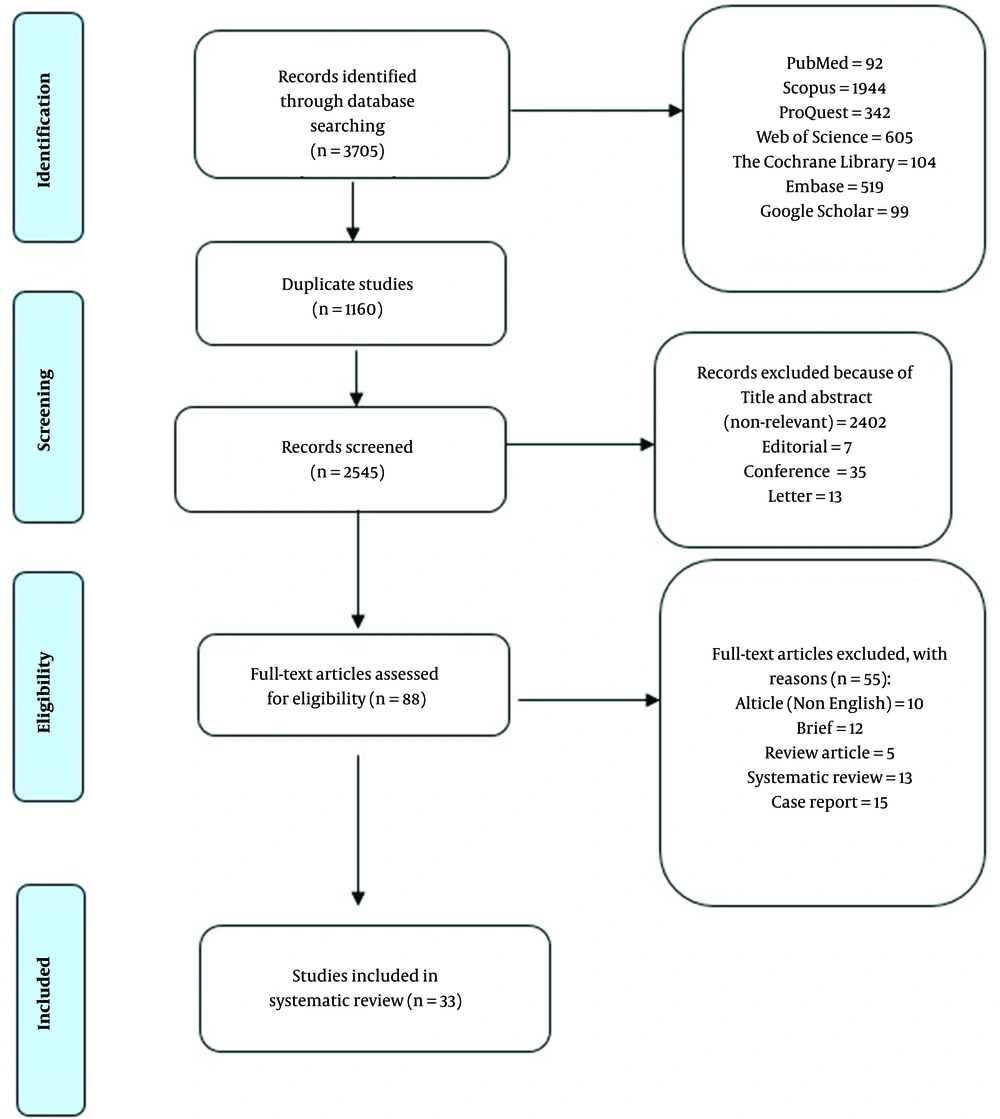
A total number of 3705 articles were obtained through a systematic electronic databases search. Of these papers, 1160 articles were omitted due to duplication, and 2457 articles were excluded after screening the titles and abstracts. Moreover, 55 articles were excluded after reading the full text. Finally, 33 studies entered the systematic review and meta-analysis (Figure 1). The characteristics of the included studies are summarized in Table 1. A total of 94.93% of the studies reported SBP as the outcome.
| Complications of Liver Cirrhosis | Number of Studies Reported, No. (%) |
|---|---|
| SBP | 31 (93.94) |
| HE | 14 (42.42) |
| Infection | 11 (33.33) |
| Variceal bleeding | 9 (27.27) |
| Ascites | 9 (27.27) |
| Mortality | 9 (27.27) |
| GIB | 3 (9.09) |
| Cirrhosis | 2 (6.06) |
| Hepatorenal syndrome | 1 (3.03) |
| SIBO | 1 (3.03) |
Prevalence of Complications in Studied Articles
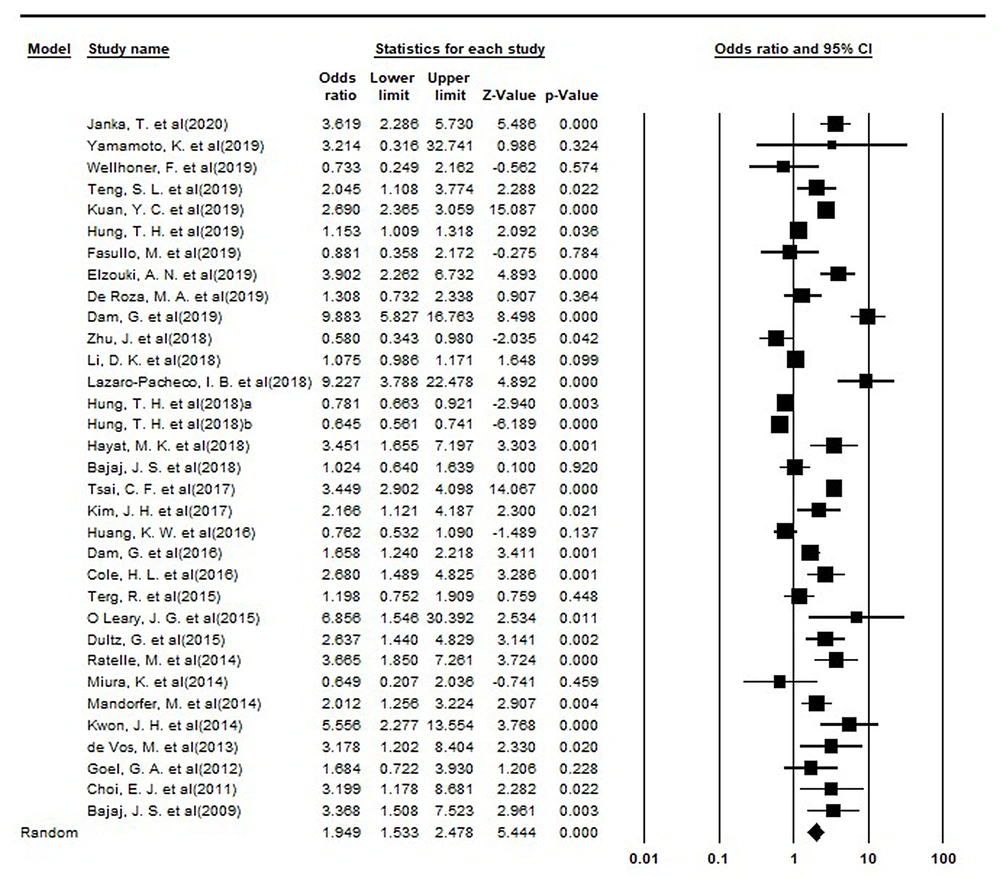
In the eligible articles for this systematic review and meta-analysis, 28778 patients used PPI, and 34644 cases were non-users. Of these cases, 6370 and 8037 patients in the intervention and control groups experienced at least one of the complications of liver cirrhosis, respectively. According to the meta-analysis, the risk of complications in the intervention group was 1.95 times higher than in the control group (RR = 1.95; 95% confidence interval [CI]: 1.53 - 2.48, P < 0.001). The forest plot of the meta-analysis is illustrated in Figure 2.
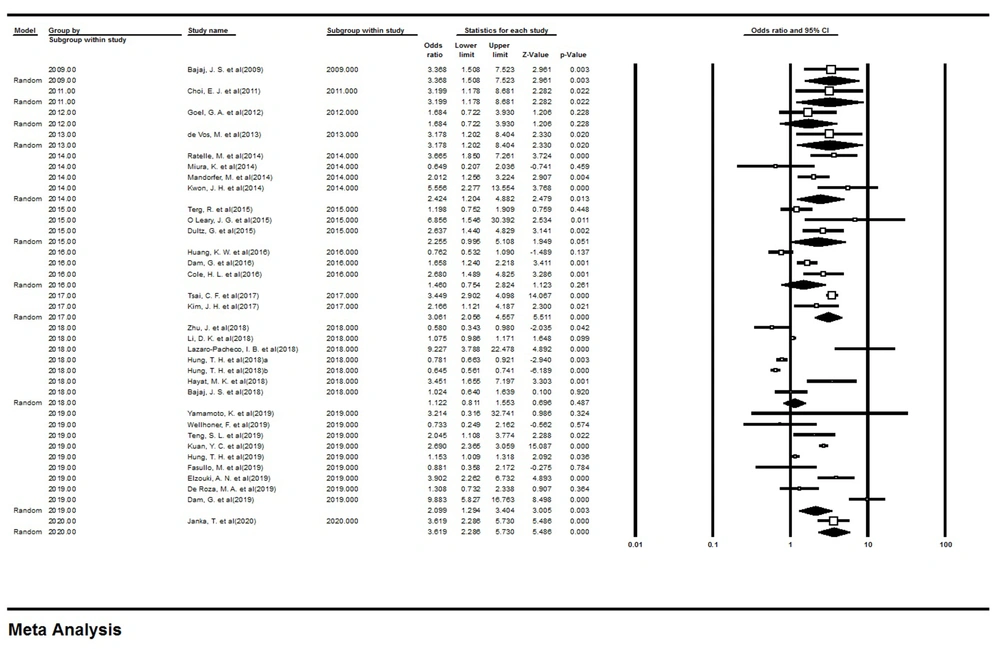
Since there was heterogeneity among studies, a subgroup analysis was performed based on the date of publishing the papers, and the results showed that there was no significant difference between the studies based on the years of publication (Figure 3). The odds ratio (OR) value for studies by the years of publication of the articles is shown in Table 2.
| Group | Number of Studies | Point Estimate | Lower Limit | Upper Limit | Z-Value | P-Value | Q-Value | df (Q) | P-Value | I-Squared |
|---|---|---|---|---|---|---|---|---|---|---|
| 2009 | 1.00 | 3.37 | 1.51 | 7.52 | 2.96 | 0.00 | 0.00 | 0.00 | 1.00 | 0.00 |
| 2011 | 1.00 | 3.20 | 1.18 | 8.68 | 2.28 | 0.02 | 0.00 | 0.00 | 1.00 | 0.00 |
| 2012 | 1.00 | 1.68 | 0.72 | 3.93 | 1.21 | 0.23 | 0.00 | 0.00 | 1.00 | 0.00 |
| 2013 | 1.00 | 3.18 | 1.20 | 8.40 | 2.33 | 0.02 | 0.00 | 0.00 | 1.00 | 0.00 |
| 2014 | 4.00 | 2.42 | 1.20 | 4.88 | 2.48 | 0.01 | 10.43 | 3.00 | 0.02 | 71.24 |
| 2015 | 3.00 | 2.26 | 1.00 | 5.11 | 1.95 | 0.05 | 7.54 | 2.00 | 0.02 | 73.46 |
| 2016 | 3.00 | 1.46 | 0.75 | 2.82 | 1.12 | 0.26 | 16.94 | 2.00 | 0.00 | 88.19 |
| 2017 | 2.00 | 3.06 | 2.06 | 4.56 | 5.51 | 0.00 | 1.79 | 1.00 | 0.18 | 44.13 |
| 2018 | 7.00 | 1.12 | 0.81 | 1.55 | 0.70 | 0.49 | 82.85 | 6.00 | 0.00 | 92.76 |
| 2019 | 9.00 | 2.10 | 1.29 | 3.40 | 3.01 | 0.00 | 133.51 | 8.00 | 0.00 | 94.01 |
| 2020 | 1.00 | 3.62 | 2.29 | 5.73 | 5.49 | 0.00 | 0.00 | 0.00 | 1.00 | 0.00 |
Results of Subgroup Analysis Based on the Year of Publication
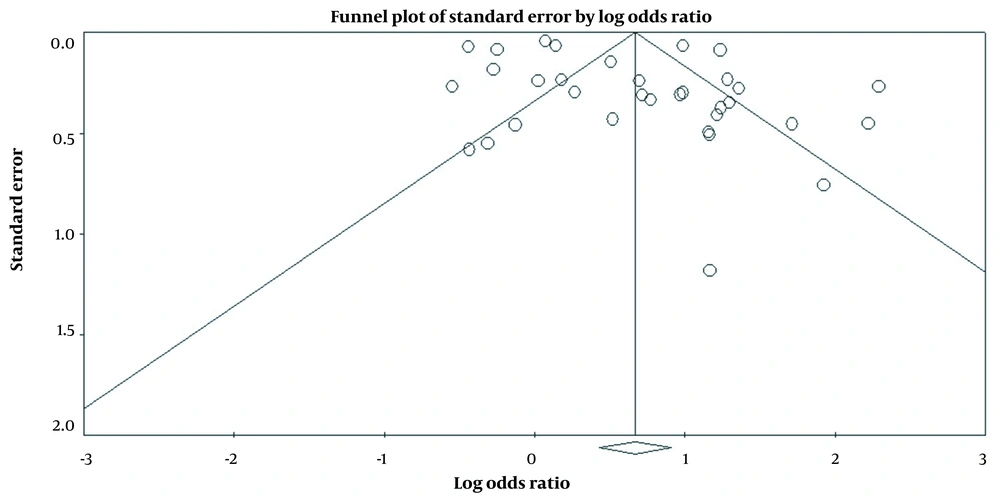
6.1. Publication Bias
According to the results of the funnel plot, there was no publication bias in the included studies (t-value = 1.94, df = 31, P = 0.06) (Figure 4).
The Critical Appraisal Checklists of the JBI (Available at https://jbi.global/critical-appraisal-tools) were used to evaluate the methodological quality, and according to the results, the studies had moderate quality (Table 3).
| Author | Ref | Q1 a | Q2 b | Q3 c | Q4 d | Q5 e | Q6 f | Q7 g | Q8 h | Q9 i | Q10 j | Q11 k |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Janka et al., 2019 | (20) | Yes | Yes | Yes | Unclear | Unclear | Yes | Yes | Yes | Yes | Yes | Yes |
| Yamamoto et al., 2019 | (21) | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| Wellhöner, 2019 | (22) | Yes | Yes | Yes | Unclear | Unclear | Yes | Yes | Yes | Yes | Yes | Yes |
| Teng et al., 2019 | (23) | Yes | Yes | Yes | Unclear | Unclear | Yes | Yes | Yes | Yes | Yes | Yes |
| Kuan et al., 2019 | (24) | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| Huang et al., 2019 | (25) | Yes | Yes | Yes | Unclear | Unclear | Yes | Yes | Yes | Yes | Yes | Yes |
| Fasullo et al., 2019 | (26) | Yes | Yes | Yes | Unclear | Unclear | Yes | Yes | Yes | Yes | Yes | Yes |
| Elzouki et al., 2019 | (27) | Yes | Yes | Yes | Unclear | Unclear | Yes | Yes | Yes | Yes | Yes | Yes |
| De Roza et al., 2019 | (28) | Yes | Yes | Yes | Unclear | Unclear | Yes | Yes | Yes | Yes | Yes | Yes |
| Dam et al., 2019 | (29) | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| Zhu et al., 2018 | (30) | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | |||
| Li et al., 2017 | (31) | Yes | Yes | Yes | Unclear | Unclear | Yes | Yes | Yes | Yes | Yes | Yes |
| Lazaro-Pacheco et al., 2017 | (32) | Yes | Yes | Yes | Unclear | Unclear | Yes | Yes | Yes | Yes | Yes | Yes |
| Hung et al., 2018 | (33) | Yes | Yes | Yes | Unclear | Unclear | Yes | Yes | Yes | Yes | Yes | Yes |
| Hung et al., 2018 | (34) | Yes | Yes | Yes | Unclear | Unclear | Yes | Yes | Yes | Yes | Yes | Yes |
| Hayat et al., 2018 | (35) | Yes | Yes | Yes | Unclear | Unclear | Yes | Yes | Yes | Yes | Yes | Yes |
| Bajaj et al., 2009 | (10) | Yes | Yes | Yes | Unclear | Unclear | Yes | Yes | Yes | Yes | Yes | Yes |
| Tsai et al., 2017 | (36) | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| Kim et al., 2017 | (37) | Yes | Yes | Yes | Unclear | Unclear | Yes | Yes | Yes | Yes | Yes | Yes |
| Huang et al., 2016 | (38) | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| Dam et al., 2016 | (39) | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| Cole et al., 2016 | (40) | N/A | N/A | Yes | Unclear | Unclear | Yes | Yes | Yes | Yes | Yes | Yes |
| Terg et al., 2015 | (41) | Yes | Yes | Yes | Unclear | Unclear | Yes | Yes | Yes | Yes | Yes | Yes |
| O’Leary et al., 2015 | (42) | Yes | Yes | Yes | Unclear | Unclear | Yes | Yes | Yes | Yes | Yes | Yes |
| Dultz et al., 2015 | (43) | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| Ratelle et al., 2014 | (44) | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| Miura et al., 2013 | (45) | Yes | Yes | Yes | Unclear | Unclear | Yes | Yes | Yes | Yes | Yes | Yes |
| Mandorfer et al., 2014 | (46) | Yes | Yes | Yes | Unclear | Unclear | Yes | Yes | Yes | Yes | Yes | Yes |
| Kwon et al., 2014 | (47) | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| De Vos et al., 2013 | (48) | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| Goel et al., 2012 | (8) | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| Choi et al., 2011 | (9) | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| Bajaj et al., 2008 | (49) | Yes | Yes | Yes | Unclear | Unclear | Yes | Yes | Yes | Yes | Yes | Yes |
Methodology Quality According to the Joanna Briggs Institute (JBI) Critical Appraisal Checklist
7. Discussion
This systematic review and meta-analysis showed that there was an association between PPI use and the occurrence of SBP or HE, and cirrhotic PPI users are at a two-fold risk of SBP or HE than non-users. Gastrointestinal bleeding, such as esophageal variceal bleeding, peptic ulcer bleeding, portal hypertensive gastropathy, and stomach vascular ectasia syndrome, are frequent in cirrhotic patients. Proton pump inhibitors are applied to these patients to treat or stop bleeding following endoscopic hemostasis (50, 51). Recent research, however, suggested that PPIs might make SBP more common in cirrhotic patients (39).
Proton pump inhibitors are frequently utilized in therapeutic settings for a variety of patient indications. The treatment of peptic ulcer disease, gastroesophageal reflux disease, Zollinger-Ellison syndrome, non-steroidal anti-inflammatory drug (NSAID)-associated ulcers, and elimination of Helicobacter pylori are among the indications for PPIs (52, 53). In order to avoid peptic problems in patients receiving multidrug therapy for variceal or hypertensive gastropathy hemorrhage, PPIs are frequently used in patients with cirrhosis, sometimes even in the absence of a specific acid-related condition. The use of this class of medications appears to be more habitual than evidence-based ones, ultimately jeopardizing patient safety and raising healthcare expenditures (51).
Healthcare professionals managing patients with cirrhosis should be aware that the majority of these individuals do not require the usage of PPIs and should take all reasonable steps to actively review and reassess the current PPI therapy. Prescribers should use PPIs sparingly and only when there is clear evidence of their benefits. Although a small number of the studies included in the present systematic review contradicted this relationship, most of the included studies demonstrated a risk between the use of PPIs and the emergence of SBP. The patients with considerable liver damage in the included studies might be responsible for the disparity. Additionally, drug dosage or types might have an impact on the outcome of treatment. The use of PPIs and its relationship with the prevalence of SBP in cirrhotic patients is debatable, which is likely due to the heterogeneity of the individuals included between studies and other methodological problems, such as retrospective design and inadequate follow-up. Additionally, the negative effects of PPIs might only be experienced by certain subgroups, such as patients with decompensated cirrhosis, particularly in the case of ascites presence.
Using PPI can interfere with the immune response to pathogens that enter through the mouth, decrease the motility of the digestion system, delay the evacuation of gastric, and decrease gastric mucous viscosity. All of the aforementioned issues can affect intestine flora which might cause SIBO. In addition to acid suppression, PPIs interfere with neutrophil action and innate immunity. The appropriate action of neutrophils is required for the release of active oxygen and the production of interleukin 8 (IL-8). These mechanisms shed light on the role of PPIs in the spread of pneumonia and other serious healthcare-associated and hospital-acquired infections (15, 20). With a 4-fold increase, infection is the primary factor in cirrhotic patients’ mortality (16, 54). The presence of infection, even after it has resolved, is suggestive of the next prognostic stage; therefore, it must be taken into account when grading cirrhosis, according to a recent study by Arvaniti et al. (16, 55).
Spontaneous bacterial peritonitis is the illness that cirrhotic patients encounter the most commonly and is always linked to a poor prognosis (56). The prevalence changes throughout time and depending on the population. However, over the past two decades, the prevalence has ranged from 10% to 30% (55, 57, 58), with the most recent systematic study indicating a pooled frequency of 17.12% (59). Spontaneous bacterial peritonitis has a 40% (60) survival rate after the initial episode and a 70% (61) recurrence rate, accounting for 4% (59) of emergency department visits in the cirrhotic population. About 33 - 50% of patients with SBP have been observed to have acute renal damage (62).
Prophylactic and therapeutic antibiotic use, intravenous albumin infusion, and risk factor modification are all examples of management options for SBP. Old age, female gender, HE, coagulopathy, and variceal hemorrhage are previously recognized risk factors for SBP based on research (63). In addition, the use of PPI but not H2-blocker is an independent risk factor for the development of SBP in cirrhotic patients with ascites. The use of β-blockers is protective against the development of SBP, as has already been highlighted in the literature (64, 65). The infection with Clostridium difficile and nosocomial and community-acquired pneumonia have all been linked to long-term PPI use (66, 67).
A substantial body of evidence has emerged recently linking the usage of PPIs with the emergence of SBP in cirrhotic individuals. Regarding immunological dysfunction, PPIs and liver dysfunction share a similar mechanism, going back to the physiopathology of cirrhosis and SBP. Proton pump inhibitor use has been linked to increased risks of bacterial infections, such as SBP, by inhibiting phagocytosis, reducing oxidative burst, and promoting bacterial translocation (68-71). Clinical results on this subject, however, are still debatable. This issue makes it difficult to provide specific recommendations and emphasizes the need for larger, population-based research to support this association.
Some earlier studies identified no link between PPI use and the onset of SBP (37, 41, 46, 72, 73). The aforementioned studies, however, used smaller samples, had inconsistent definitions of exposures and outcomes, or failed to take suitable confounders into account. However, a growing number of studies have found a positive correlation (41, 42, 68, 74), and the majority of these studies have stronger levels of evidence (67, 75, 76). Variceal bleeding is much more common among PPI users, as demonstrated by Mandorfer et al. (46). A previous history of SBP episodes is a substantial risk factor for the development of a second episode; nevertheless, the database used does not provide any information regarding those episodes.
In terms of the association of PPIs and HE, short-term PPI consumption is associated with a significant risk of HE in patients with decompensated cirrhosis at varied periods of time, regardless of age, gender, and recent comorbidities. The highest risk is for the 28-day window, and the lowest risk is for the 7-day window. Esomeprazole, rabeprazole, and lansoprazole (more potent to suppress the effect of gastric acid) were associated with the risk of HE, but not omeprazole and pantoprazole (24). The use of PPIs might affect cirrhotic patients by changing the pH of the stomach and leading to the proliferation of the intestinal microbiome; therefore, it increases ammonia production and bacterial transport. Proton pump inhibitor use is associated with worse degrees of HE (26). The use of PPIs in patients who suffer from decompensated liver cirrhosis is associated with a higher mortality rate and major liver impairment complications that require hospitalization (28). The role of PPIs in cirrhotic patients with HE should focus on Helicobacter pylori eradication, not gastric acid suppression (33).
The current review is more recent and thorough, although recent systematic reviews have sought to investigate this association (77-80). With a larger patient group, this review included 33 published papers in this analysis, which is more than performed in earlier assessments. Although liver failure can occur due to various reasons, further prospective studies that stratify the population at risk of developing SBP according to the severity of their liver illness would be interesting, given that the risk of SBP is higher in individuals with severe liver disease (81-84).
7.1. Conclusions
Proton pump inhibitors increase the risk of SBP and HE and are associated with an increased risk of mortality in hospitalized or outpatient cirrhotic patients. In addition, subgroup analysis is suggested to evaluate which type and dosage of PPIs have a higher association with SBP or HE.