Nonalcoholic fatty liver disease (NAFLD) comprehends a spectrum of liver disorders ranging from simple fat accumulation in the hepatocytes (hepatic steatosis), to liver inflammation and hepatocyte injury (nonalcoholic steatohepatitis, NASH) with increasing levels of fibrosis to cirrhosis and hepatocellular carcinoma (HCC) in the worst-case scenario (
1-
3). Patients with NAFLD have a higher risk of morbidity and mortality (
3-
5). It is estimated to have a prevalence of 20%-30% in the western general population (
2,
6,
7). NAFLD has become the most common cause of chronic liver disease (
8). In the Asian-Pacific region, the prevalence of NAFLD has been reported to range from 12% to 24% (
3,
7,
8) and it is rapidly on the rise (
3,
6). In Iran, the prevalence is 2.04% in the adult population (
9) and 7.1% in the adolescent population (
10). In addition, NAFLD appears to have similar clinical and paraclinical features in Iran’s population compared to other areas of the world (
11).
For definite diagnosis and staging of NAFLD, liver biopsy remains the gold standard. Liver biopsy may, however, over- or underestimates the stage of liver disease, because it only samples a very small portion of the liver. The invasive nature of this procedure plus practical difficulties and high expenses further justify the need for a safe and reliable approach for follow-up of patients with NAFLD (
12-
16). In the recent years, emergence of noninvasive methods has provided clinicians with the possibility to identify severe liver fibrosis. However, these approaches do not fully correlate with histological stage of NAFLD in the absence of substantial liver fibrosis (
16,
17). In clinical practice, on the other hand, NAFLD is diagnosed based on ultrasonographic findings of fatty liver, elevated liver enzymes that cannot be otherwise explained and by excluding other causes of liver disease (
18,
19).
Thrombopoietin (TPO) is a hormone produced mainly by hepatocytes, which promotes platelet production by the bone marrow (
20). Studies indicated that the level of TPO is correlated to the degree of liver fibrosis and impairment of hepatocyte function (
20,
21), both of which contribute to NAFLD (
1-
3). Therefore, peripheral platelet count can possibly reflect the severity of liver injury (
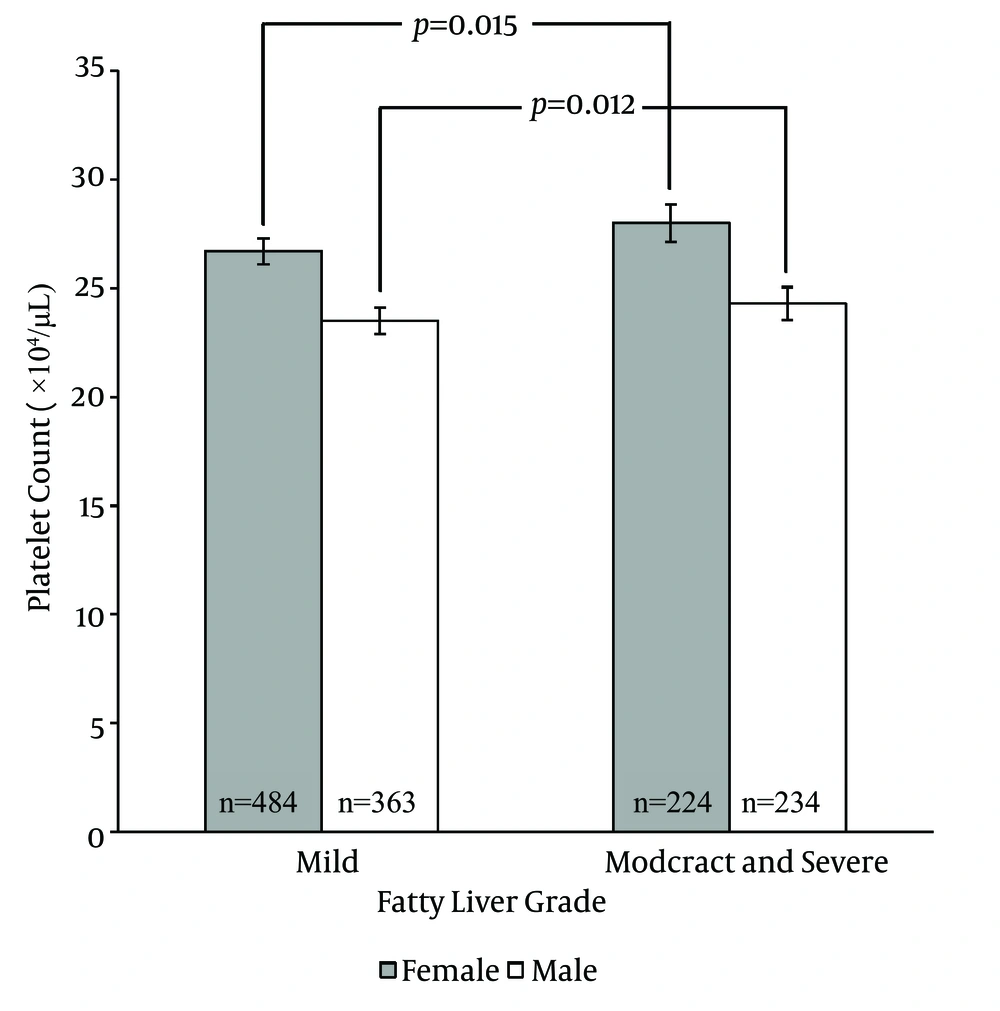
22). A number of studies demonstrated an association between platelet counts and the severity of liver injury (
23-
27), the results are controversial however. Several systems of scoring liver fibrosis such as the NAFLD fibrosis score (
28), aspartate aminotransferase (AST)-to-platelet ratio index (APRI) (
29) and the FIB-4 index (
30) incorporated the platelet count as one of their variables emphasizing the potential role of platelet count as an indicator of liver injury. There is a definite need for further research regarding the association between platelet counts and noninvasive measures of the severity of NAFLD.