1. Context
Hepatitis B virus (HBV) has infected more than one-third of the population around the world. It is estimated that more than 350 million people are chronic carriers of HBV, worldwide. HBV is one of the main causes of chronic hepatitis, with the disease progressing to hepatic cirrhosis, renal failure, or liver cancer in infected people (1, 2). Half of the infections with HBV, especially in countries with the moderate-to-high prevalence, occur in prenatal or early-childhood periods (3, 4) due to the high prevalence of hepatitis B surface antigen (HBsAg) among fertile women who transmit this infection to newborns (5). HBsAg is the main blood indicator of acute HBV infection, the existence of which is in the body for over 6 months indicates chronic infection (6).
Without any intervention or treatment for HBV infection in pregnant women, this infection will be transmitted to the fetus or newborns in 70% - 90% of cases, in which the mother’s hepatitis B e-antigen and HBsAg are both positive (7, 8), and in 10% of cases, in which only the HBsAg is positive (9, 10). Currently, the hepatitis B vaccine, that is highly reliable and effective in preventing the infection, is widely available to the public, especially pregnant women (11, 12). Nevertheless, HBV infection management during pregnancy is extremely difficult with respect to the risk of transmitting the infection to the fetus (11, 13). Screening programs for mothers aiming to identify those with positive HBsAg are implemented in many countries as a part of usual pregnancy examinations. If a mother is HBsAg positive, her neonate must receive hepatitis B immune globulin in order to decrease the chance of HBV vertical transmission (13). Although prophylaxis is effective in diminishing HBV infection transmission from the mother to the newborns, 3% - 13% of children born from HBsAg-positive mothers are still infected with HBsAg, a rate that is not negligi6ble (13, 14).
The World Health Organization (WHO) has estimated the prevalence of HBV in the Eastern Mediterranean Regional Office (EMRO) to be high, approximately 4.3 million (15). WHO has classified HBV prevalence as low (< 2%), moderate (2% - 8%), and high (> 8%) (16). Low HBV prevalence areas include the United States, Eastern Europe, Australia, and New Zealand, moderate HBV prevalence areas include the Mediterranean countries, Japan, Central Asia, the Middle East, and Southern USA, and high HBV prevalence areas are placed in Southeast Asia, China, and sub-Saharan Africa (17). Iran, as an important country in the Middle East, has a moderate level of HBV prevalence (18). A systematic review by Alavian et al. on Iranian people revealed that more than one million people have HBV infection (19). The prevalence of HBV varies among pregnant women residing in EMRO countries and the Middle East, reported being 1.2% in Iran (20), 2.2% in Egypt (21), and 4% in Turkey (22).
Identifying the prevalence of HBV among pregnant women can have an important role in determining control and prevention policies so that the vertical transmission rate of the disease can be decreased in the region by implementing more extensive control programs. Comprehensive review studies examining the prevalence of HBsAg in EMRO and Middle Eastern countries have not investigated the prevalence among pregnant women. Lack of reliable information on the prevalence of HBsAg among pregnant women residing in these countries is noticeable. Therefore, a meta-analytic and review study in these countries seems useful. Motivated by the abovementioned reasons, the present study was conducted on cross-sectional studies published from 2000 to 2016 in order to determine the prevalence of HBsAg among pregnant women residing in EMRO and Middle Eastern countries.
2. Method
2.1. Sources of Data
We searched Pubmed, ISI Web of Science, ScienceDirect, Ovid, Scopus, and Google Scholar electronic databases for paper titles and/or abstracts in order to access papers published from January 1, 2000 to December 31, 2016. At this step, the number of papers found in each database and the final number of searched papers was recorded. We conducted searches using different combinations of keywords and MeSH terms including:
“epidemiology,” “prevalence,” “HBV,” “hepatitis b virus,” “HBsAg,” and “Pregnant women,” along with the names of the EMRO and Middle East countries, including: Afghanistan, Bahrain, Cyprus, Djibouti, Egypt, Iran, Iraq, Jordan, Kuwait, Lebanon, Libya, Morocco, Oman, Pakistan, Palestine, Qatar, Saudi Arabia, Somalia, Sudan, Syria, Tunisia, Turkey, United Arab Emirates, and Yemen.
Additionally, using the mentioned keywords in English and Persian, Iranian databases including Scientific Information Database, Iran Medex, Magiran, and Medlib, as well as other related international sources such as PakMediNet (comprehensive Pakistani database) and WHO Eastern Mediterranean Health Journal were included in our searches.
The sensitivity of the searches was examined by checking for duplicate papers. When the full-text of a paper was not available, we requested the paper from the author via email. In cases where the author did not answer the email, we used the abstract. Papers lacking the required information were excluded from the study.
2.2. Selecting the Studies
Full texts of English or Persian papers were included if they were cross-sectional and contained specific information on the prevalence of HBsAg among pregnant women residing in EMRO and Middle Eastern countries. At this step, studies conducted on the general population were also reviewed so that they would be included if any of their sub-groups consisted of pregnant women. The exclusion criteria were: (1) studies that were not cross-sectional and (2) studies that had not clearly reported the prevalence of HBsAg and only mentioned the general prevalence of hepatitis B (with respect to the aim of the study, i.e. investigating the prevalence of HBsAg among pregnant women). The names of authors or titles of journals had no effect on the selection of papers.
Journals had no effect on the selection of papers.
2.3. Data Extraction
Both authors evaluated the quality assessment of papers, independently. First, the first author (M.B.), who was an epidemiologist, extracted the data in order to identify eligible studies. The other author (S.M.A.), who was an expert in hepatology and had not participated in the process of searching, conducted a detailed quality evaluation on the papers. At the next step and before the critical evaluation, the authors held a session on the research questions and in this session, all articles were assessed one by one by both authors and inclusion or exclusion of articles was specified. After the final evaluation, the authors recorded the selected studies and required data, i.e. name of the first author, publication year, country, sample size, the prevalence of HBsAg, and standard error (SE), and entered them into the Excel software. SE was calculated using Cochran’s test.
2.4. Statistical Analysis
Heterogeneity was assessed based on Cochran’s Q-test results. However, since this test may fail to exactly discover true heterogeneity, it was complemented with Higgins and Thompson’s I2. The command “metaprop” was then used to apply a fixed or random effects model based on the results and significance of the Cochran’s test results or a large I2 value, respectively. By using metaprop, no studies with 0% or 100% proportions were excluded from the meta-analysis. Furthermore, study specific and pooled confidence intervals were always within admissible values and avoid confidence intervals exceeding the 0 to 1 range (23). Data aggregation and production of the pooled estimates were performed using the above-mentioned methods. Forest plots with descriptions of the findings were then developed to describe the results and calculate the point estimations and 95% confidence intervals (CIs).
3. Results
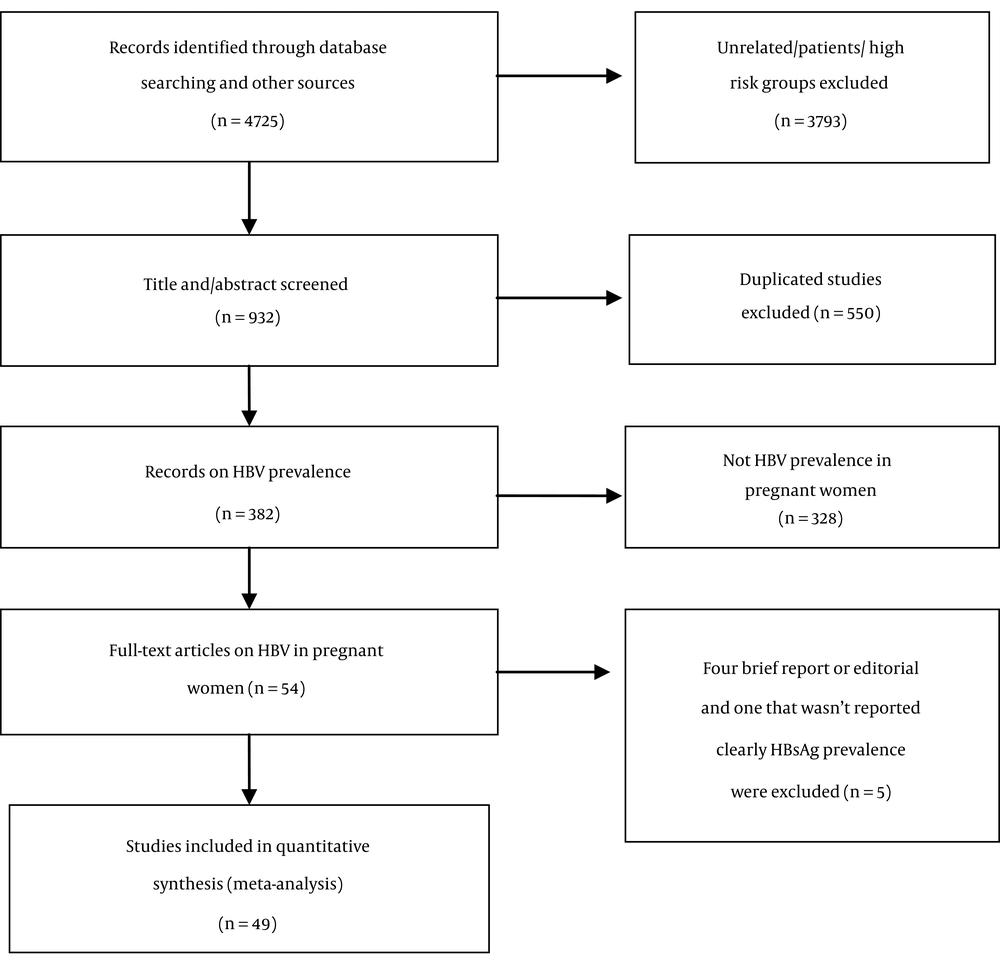
In the first step, 4725 papers were found based on titles and/or abstracts, 932 papers of which were potentially relevant to the aims of the study and selected. In the next step, 550 duplicate papers were excluded, leaving 382 papers related to the prevalence of HBV in EMRO and Middle Eastern countries. Based on the available data, 55 papers reporting the prevalence of HBV among pregnant women were selected. Due to the importance of papers’ quality evaluation, 5 papers lacking the inclusion criteria were excluded from the study, 4 of which were in the form of a short report or letter to the editor (24-27), and 1 had not clearly reported the prevalence of HBsAg among pregnant women, only mentioning the prevalence of HBV (28). At the end, 49 papers, which had clearly reported the prevalence of HBsAg among pregnant women, remained for the analysis (Figure 1). Data on HBsAg prevalence in pregnant women wasn’t found for other EMRO and Middle Eastern countries.
3.1. Characteristics of Studies
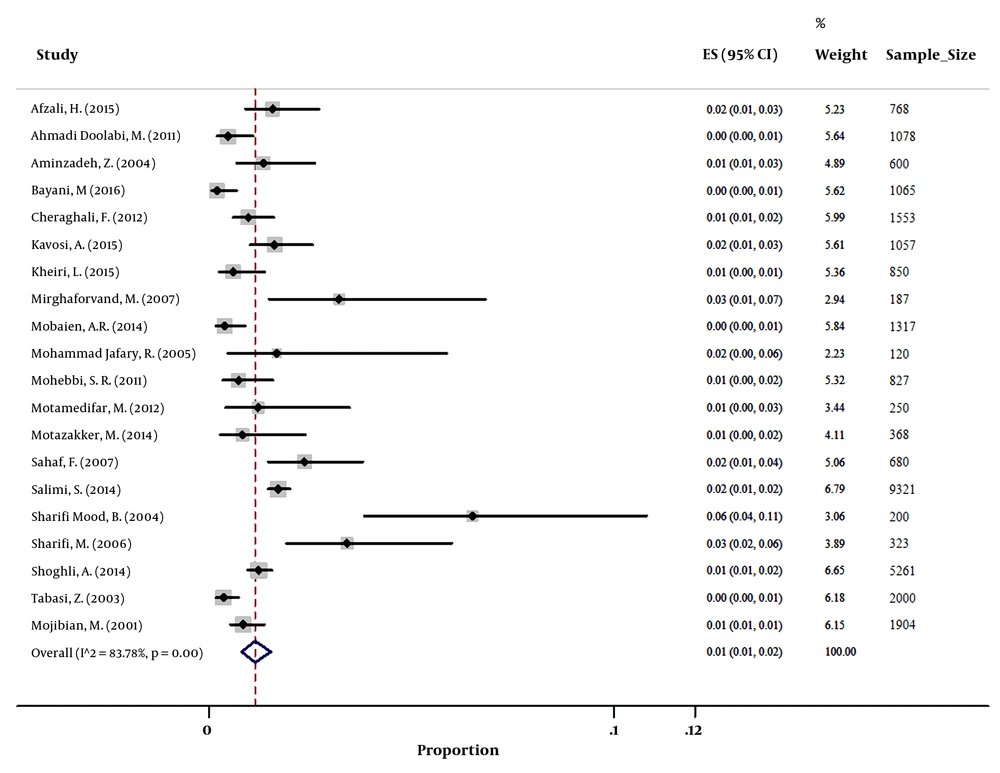
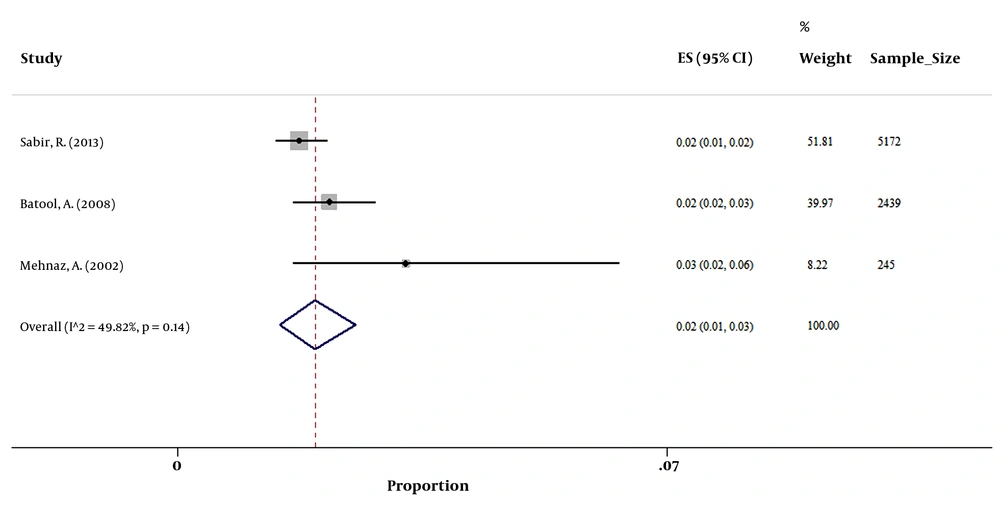
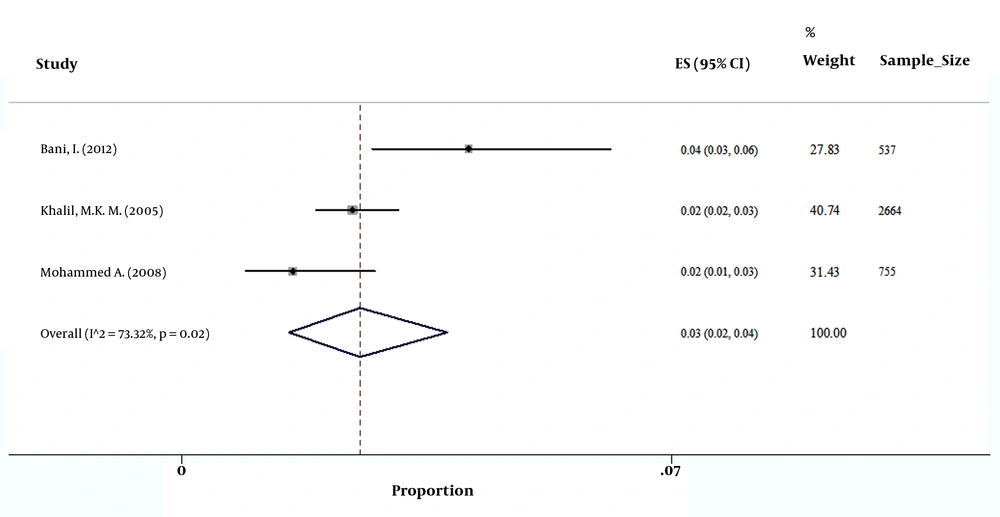
Generally, 49 papers were selected from 13 EMRO and Middle Eastern countries, reporting the prevalence of HBsAg among pregnant women, with the sample size of 89452. From among them, 2 studies had been conducted in Egypt (21, 29), 20 in Iran (30-49), 1 in Jordan (50), 1 in Libya (51), 1 in Oman (52), 3 in Pakistan (53-55), 1 in Qatar (52), 3 in Saudi Arabia (56-58), 3 in Sudan (59-61), 1 in Tunisia (62), 11 in Turkey (22, 63-72), 1 in the United Arab Emirates (52), and 1 in Yemen (73).
3.2. The Total Prevalence of HBsAg Among Pregnant Women Residing in EMRO and Middle Eastern Countries
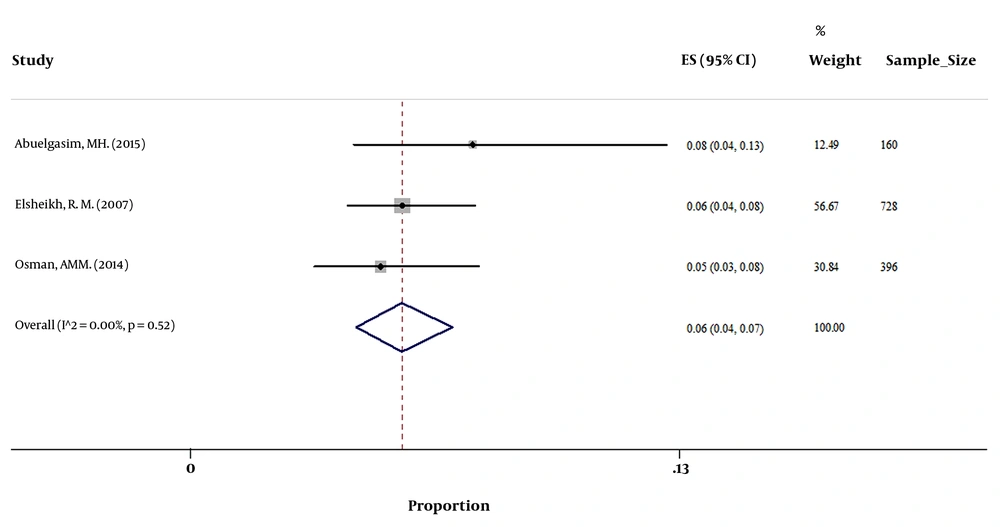
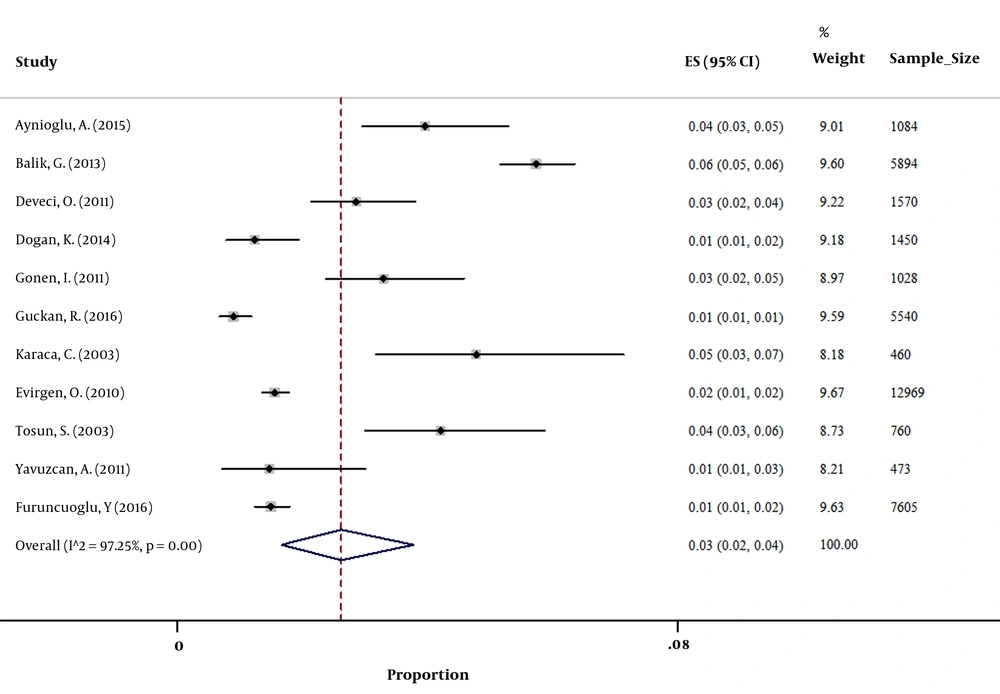
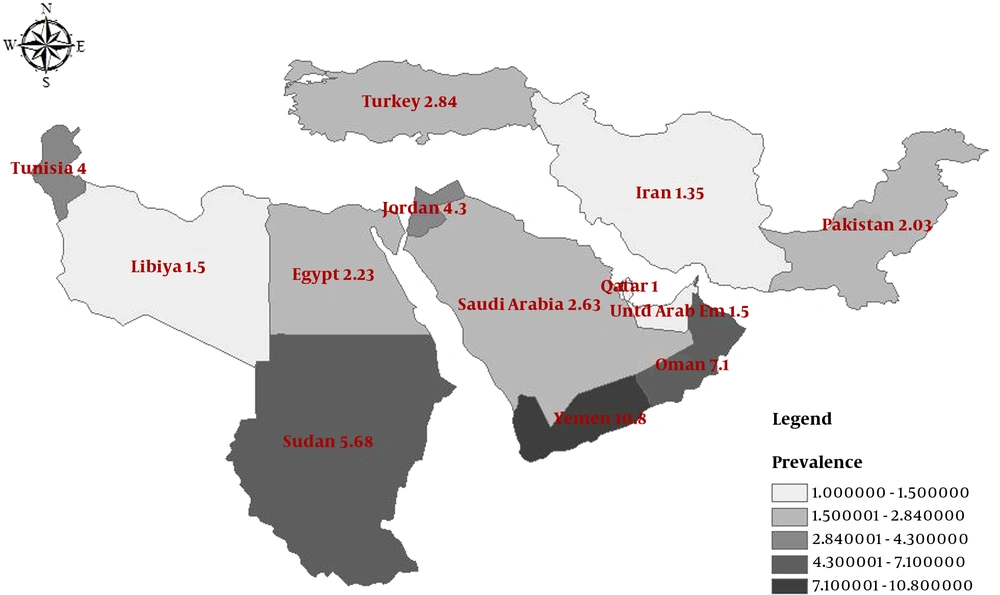
Based on the available data from 13 countries with at least one included study, the highest and lowest prevalence of HBsAg among pregnant women was 10.8% for Yemen and 1% for Qatar (Table 1). In addition, the pooled prevalence of HBsAg in countries with at least 2 papers revealed that the prevalence of HBsAg among pregnant women was 1.35% (95% CI: 1.22 - 1.48) in Iran and 2.03% (95% CI: 1.74 - 2.37) in Pakistan, which were lowest rates after the mentioned prevalence for Qatar. The prevalence in countries with at least 2 papers is available in Table 2 and Figures 2 - 6. Furthermore, the geographic distribution of HBsAg prevalence in pregnant women of EMRO and Middle East countries was shown in Figure 7.
| Countries | Authors’ Names | Pub. Year | Samples Size | HBsAg Positive, No | HBsAg Prevalence, % |
|---|---|---|---|---|---|
| Egypt (E and M) | |||||
| Abo-Salem MES (21) | 2014 | 397 | 9 | 2.3 | |
| Zahran KM (29) | 2010 | 500 | 20 | 4 | |
| Iran (E and M) | |||||
| Afzali H (30) | 2015 | 768 | 12 | 1.56 | |
| Ahmadi M (31) | 2011 | 1078 | 5 | 0.5 | |
| Aminzadeh Z (32) | 2004 | 600 | 8 | 1.3 | |
| Bayani M (33) | 2016 | 1065 | 2 | 0.18 | |
| Cheraghali F (34) | 2012 | 1553 | 15 | 1 | |
| Kavosi A (35) | 2015 | 1057 | 17 | 1.6 | |
| Kheiri L (36) | 2015 | 850 | 5 | 0.59 | |
| Mirghaforvand M (37) | 2007 | 187 | 6 | 3.2 | |
| Mobaien AR (38) | 2014 | 1317 | 5 | 0.4 | |
| Mohammad Jafari R (39) | 2005 | 120 | 2 | 1.7 | |
| Mohebbi SR (40) | 2011 | 827 | 6 | 0.7 | |
| Motamedifar M (41) | 2012 | 250 | 3 | 1.2 | |
| Motazakker M (42) | 2014 | 368 | 3 | 0.8 | |
| Sahaf F (43) | 2007 | 680 | 16 | 2.5 | |
| Salimi S (44) | 2014 | 9321 | 159 | 1.7 | |
| Sharifi Mood B (45) | 2004 | 200 | 13 | 6.5 | |
| Sharifi M (46) | 2006 | 323 | 11 | 3.4 | |
| Shoghli A (47) | 2014 | 5261 | 64 | 1.21 | |
| Tabasi Z (48) | 2003 | 2000 | 7 | 0.35 | |
| Mojibian M (49) | 2001 | 1904 | 16 | 0.84 | |
| Jordan (E and M) | Batayneh N (50) | 2002 | 1000 | 43 | 4.3 |
| Libya (E and M) | El-Magrahe H (51) | 2010 | 1500 | 23 | 1.5 |
| Oman (E and M) | Al Awaidy S (52) | 2006 | 604 | 43 | 7.1 |
| Pakistan (E) | |||||
| Sabir R (53) | 2013 | 5172 | 90 | 1.74 | |
| Asia B (54) | 2008 | 2439 | 53 | 2.2 | |
| Mehnaz A (55) | 2002 | 245 | 8 | 3.26 | |
| Qatar (E and M) | Al Awaidy S (52) | 2006 | 495 | 5 | 1 |
| Saudi Arabia (E and M) | |||||
| Bani I (56) | 2012 | 537 | 22 | 4.1 | |
| Khalil MKM (57) | 2005 | 2664 | 65 | 2.44 | |
| Alrowaily MA (58) | 2008 | 755 | 12 | 1.6 | |
| Abuelgasim MH (59) | 2015 | 160 | 12 | 7.5 | |
| Sudan (E) | |||||
| Elsheikh RM (60) | 2007 | 728 | 41 | 5.6 | |
| Osman AMM (61) | 2014 | 396 | 20 | 5.1 | |
| Tunisia (E) | Hannachi N (62) | 2009 | 2303 | 92 | 4 |
| Turkey (M) | |||||
| Aynioglu A (22) | 2015 | 1084 | 43 | 4 | |
| Balik G (63) | 2013 | 5894 | 338 | 5.7 | |
| Deveci O (64) | 2011 | 1570 | 45 | 2.9 | |
| Dogan K (65) | 2014 | 1450 | 18 | 1.2 | |
| Gonen I (66) | 2011 | 1028 | 34 | 3.3 | |
| Guckan R (67) | 2016 | 5540 | 50 | 0.9 | |
| Karaca C (68) | 2003 | 460 | 22 | 4.7 | |
| Evirgen O (69) | 2010 | 12969 | 203 | 1.6 | |
| Tosun S (70) | 2003 | 760 | 32 | 4.2 | |
| Yavuzcan A (71) | 2011 | 473 | 7 | 1.47 | |
| Furuncuoglu Y (72) | 2016 | 7605 | 114 | 1.5 | |
| United Arab Emirates (E and M) | Al Awaidy S (52) | 2006 | 595 | 9 | 1.5 |
| Yemen (E and M) | Murad EA (73) | 2013 | 400 | 43 | 10.8 |
aE: EMRO countries; M: Middle East countries; E & M: All countries from two regions.
| Countries | Prevalence | Confidence Intervals |
|---|---|---|
| Egypt | 3.23 | 2.26 - 4.6 |
| Iran | 1.35 | 1.22 - 1.48 |
| Pakistan | 2.03 | 1.74 - 2.37 |
| Saudi Arabia | 2.63 | 2.17 - 3.18 |
| Sudan | 5.68 | 4.54 - 7.08 |
| Turkey | 2.84 | 2.68 - 3.01 |
4. Discussion
The present study was conducted to determine the prevalence of HBsAg among pregnant women from EMRO and Middle East countries during 2000 - 2016. In this study, the minimum and maximum prevalence of HBsAg among pregnant women in the countries of the EMRO and the Middle East was 1% and 10.8% for Qatar and Yemen, respectively. In the literature review, the prevalence of HBsAg among pregnant women is different in various parts of the world. The prevalence among the African countries such as Ethiopia (3.8%) (74) as well as Nigeria (11.6% and 12.5%) (75, 76) and among the Western Pacific countries such as Taiwan (15.5%) (77), and China (5.49%) (78) is medium and high, while the European countries have a acceptable situation, in which the prevalence of HBsAg is much lower. The prevalence is low in European countries such as Germany (0.48%) (79), France (0.65%) (80), and England (0.9%) (81). The prevalence of HBsAg among pregnant women in the United States is different for different race: 6% among Asian women, 1% among African American women, 0.6% among non-Hispanic white women, and 0.1% among Hispanic women (3).
It is no doubt that hepatitis is still a global public health problem. For the first time in 2016, the World Health Assembly approved "the global health sector strategy on viral hepatitis in 2016 - 2020”. The prospect of this strategy is the elimination of viral hepatitis as a public health concern. The global objectives of this strategy are to reduce viral infections by 90% and decrease mortality caused by viral hepatitis by 65% by the year 2030 (82). However, almost 600000 people die annually due to complications such as acute liver failure, cirrhosis, and liver cancer (12). According to the Center for Disease Control, lack of the post exposure immune prophylaxis, nearly 40% of the infants in the United States with mothers infected with HBV have chronic HBV infection and, finally, one out of every four infants die of the chronic liver diseases (83). In addition to the problems associated with HBV infection in infants, other small-scale complications can occur in infants and women. Greater incidence of low birth weight and prematurity has been reported among pregnant women with acute hepatitis B infections. Gestational diabetes, antepartum hemorrhage, and preterm delivery are higher among mothers with chronic infection than the general population (3), while this problem can be more acute and widespread in less developed and non-developed countries with higher prevalence of infection among the pregnant women.
The safe and effective hepatitis B vaccine became commercially available in 1982 (84). In December 2006, 164 of the 193 countries in the World Health Organization included hepatitis B vaccination in their child-care program (82). According to the World Health Organization, the global vaccination coverage in the third injection was 84% in 2015 (82). The vaccination and increase in the number of people resistant to hepatitis B can reduce the burden of hepatitis in the society and, as a result, decrease its transmission to women of reproductive ages and, thus, to pregnant women. Studies in Asia have shown the effect of vaccination on reducing the prevalence of chronic infection and hepatocellular carcinoma among children (85-87). Another factor affecting the reduction of prevalence can be prevention strategies such as screening blood donations among the volunteers and quality control. In 2013, 97% of blood donors underwent screening and quality control. This screening had a positive effect on reducing transmission to the society. The decrease in the unsafe injection from 39% to 5% from 2000 to 2010 in the world can also be an effective factor for the prevalence decline (82). However, hepatitis B is still a public health problem with high and medium prevalence in Asian countries. On the other hand, many people with chronic infection are asymptomatic until the cirrhosis or final stages of the liver disease and studies show that many people with chronic infection are unaware of the disease (88-90).
In view of the fact that nearly 40% - 50% of carriers of hepatitis B are infected through the mother-to-child way, (91) one of the mother-to-child transmission prevention strategies can be screening pregnant women to identify those with hepatitis B and, as a result, strategies such as hepatitis B and immunoglobulin vaccination at birth. In 2004, the US Preventive Services suggested the screening of pregnant women in the first prenatal visit (92). In American and European countries, infection in the society is low (3, 79-81); however, by screening, the pregnant women had more opportunities for preventing this infection among their children. It seems that in less- and non-developed countries with high and medium prevalence, screening has an important role in reducing infection transmitted from mother to newborn. Studies have demonstrated that the infection is higher among immigrants, particularly those from endemic areas, than the native population (3, 79). It seems that this group of people should be considered as high-risk groups in the studies on pregnant women. Since vaccination during pregnancy has no risk to the mother and fetus, it can be one of the strategies to deal with high-risk pregnant women (93).
However, in addition to the injection of vaccine and immunoglobulin into infants at birth, one of the ways to control the disease transmission is to use drugs such as telbivudine, lamivudine, and tenofovir, as a drug prevention strategy, among pregnant women who are diagnosed with the disease in screening. This strategy can prevent the viral transmission to children without adverse effects on children and mothers (94). Since 3% - 13% of the children with infected mothers fail in active and passive preventions (13, 14), measures such as maternal screening and drug therapy for reducing the risk of transmission and incidence of chronic disease in children can be the possible ways to achieve the goals of the World Health Organization in terms of reducing the incidence and mortality rate of this disease (95).
4.1. Limitations and Strengths
The first limitation of this study was the limited number of studies compared to the number of countries in the region. In order to solve this problem, we also used several studies performed on the prevalence of HBsAg in the general population in which pregnant women were studied as a subgroup. The second limitation was that we attempted to assess the prevalence of HBsAg among pregnant women in published papers and conference abstracts and grey literature were excluded, which should be considered in future studies. The third limitation was a lack of reporting the separated prevalence of HBsAg among pregnant women in terms of age, making it impossible to report the prevalence in different age groups. The fourth limitation was that we searched the English and Persian literatures, however, not the Arabic literature, which should be considered in future studies. As the last limitation, data on HBsAg prevalence in pregnant women wasn’t found for some EMRO and Middle Eastern countries that should be considered in future studies. The most important strength of the present study was the high sensitivity of searching to find the relevant studies. We tried to use all the relevant keywords for searching.
4.2. Conclusion
Based on the WHO classification of HBV prevalence, the available data on the prevalence of HBsAg in pregnant women of EMRO and Middle East countries showed that there was a different pattern of HBsAg prevalence in studied countries. Low, moderate, and high prevalence of HBsAg was observed for EMRO and Middle East countries during 2000 - 2016. Although there were countries with low prevalence of HBsAg, the lowest frequency in our study was higher than the reported prevalence in developed countries. Increasing the rate of Hepatitis b vaccination is suggested to reduce the prevalence of HBsAg in EMRO and Middle East countries, especially in countries with high prevalence of HBsAg. Additionally, screening of women during the pregnancy in all studied countries is suggested.