1. Background
The world health organization (WHO) aims at a world, in which viral hepatitis is eliminated as a public health threat by 2030 (1). Approximately, 248 million people were hepatitis B surface antigen (HBsAg) positive with a seroprevalence of 3.61% in 2010 (2). Recently, a decreasing prevalence of HBsAg was reported in 50 countries worldwide, amongst which Turkey was classified as a country with a medium relative decrease in prevalence (3).
The estimated overall population prevalence of HBsAg in Turkey was reported as 4% to 4.57% (4, 5). However, the epidemiological data on hepatitis B in Turkey is usually obtained from individuals, who apply to health institutions as patients or blood donors (5).
In Turkey, routine hepatitis B vaccination was initiated in 1998, and a catch-up immunization program was implemented for all students attending the 3rd and 4th grade in 2007 (6). Overall, a vaccination rate as high as 97% was reached in 2015 (7).
The association between HBV infection and low socioeconomic status is strong. Low education, low social class, and crowded urban residence are among socioeconomic factors that have been found to be associated with high HBV carriage in both developed and developing countries (8).
2. Objectives
This study aimed at determining the seroprevalence of hepatitis B in the pre- and post-vaccination periods in the population above two years of age in Manisa province during year 2014 and to evaluate the relationship between being vaccinated and infected with some social determinants of health.
3. Methods
This cross-sectional study was carried out with the collaboration of Dokuz Eylul University and Manisa provincial public health directorate as part of the research project titled “Determination of the seroprevalance of some vaccine-preventable diseases in Manisa, 2014”. The study was approved by the Dokuz Eylul University clinical research ethics committee. The study was financially supported by Dokuz Eylul University Scientific research grants (2015.KB.SAG.51).
3.1. Study Population
The study population consisted of all individuals above two years of age (N = 1, 317, 917) registered at the family Medicine information system (FMIS) of Manisa. Assuming the expected HBsAg positivity as 2.0% and absolute precision as 0.75%, at 95% confidence level, the minimum sample size was estimated as 1337. The sample size was inflated by 30% to reach 1740 to overcome nonresponse bias. The sampling frame consisted of all individuals older than two years of age, who were registered under the Manisa province family Medicine information system during October 2013. The study sample was selected from older than two-year old population from the FMIS records using the simple random sampling method.
The participants were then invited to 158 family health centres in the Manisa province for face to face interviews and blood sampling. Blood samples were centrifuged on the same day and kept frozen at -80°C until tested for further evaluation. During the data collection phase, 168 individuals were not found at their addresses, 312 refused to participate in the study, and data from 30 individuals were excluded due to insufficient blood sample, hemolysis or lipolysis. In total, 1230 individuals were analyzed (response rate of 70.6%) in this study.
3.2. Serological Tests
The tests were performed by a Cobas e 411 analyzer operating with the electrochemiluminescence method and by HBsAg II, Anti-HBc, and Elecsys Anti-HBs II kits (Roche Diagnostics GmbH, Mannheim, Germany) of the same brand compatible with this device. The samples with an HBsAg threshold index value of ≥ 1.0 IU/L, HBs antibody (anti-HBs) value of ≥ 10 IU/L, and hepatitis B core antibody (anti-HBc) threshold index value of ≤ 1.0 IU/L were considered as positive, and samples outside these limits were considered as negative. The measurement range for the anti-HBs test was 2 to 1000 IU/L.
3.3. Evaluation of Social Determinants of Health
The dependent variables of the study were determined as being vaccinated and infected. Gender, age group, educational status, professional status, annual per capita equivalent income, perceived income status, household density (number of persons per room), area of residence, and area of residence during childhood, as social determinants of health, were the independent variables (9). The educational level was grouped by asking the last school graduated. Individuals, who did not receive formal education were included in groups as ‘illiterate’ or ‘literate’. Individuals at the primary school age were grouped as ‘currently in primary school’ or individuals below the primary school age as ‘not at the primary school age’. Open-ended questions were asked in the questionnaire for age, annual per capita equivalent income, occupational status, number of people in the household, and number of rooms in the household, and the responses were grouped for the analysis. The professional status was classified as unemployed (unemployed and looking for a job and housewife and looking for a job), individuals out of the workforce (unemployed and not looking for a job, housewife and not looking for a job), student, production worker (working in jobs in the industry or at a factory, etc.), skill-requiring/learned professions (doctor, engineer, etc.), employer (employing regular workers and landowners employing workers), individuals working in jobs without a regular income (salesman, waiter, etc.), agricultural workers, and self-employed craftsman. The unemployed group consisted of individuals, who were unemployed and out of the workforce; the employed group consisted of other professional groups (10). The annual per capita equivalent income was calculated by dividing the household total annual income by the equivalent household size (11). The median of the annual per capita equivalent income (3265 TL) was used as the cutoff point for grouping participants to two groups as above or below 3265 TL.
3.4. Statistical Analysis
The study findings were presented as frequency for categorized variables. The association between independent variables and Hepatitis B was assessed by multinomial logistic regression analysis by considering Hepatitis B as a categorical dependent variable consisting of two or more possible outcomes (12). In the multinomial modelling, hepatitis B seronegativity was considered as the reference group. The association between the social determinants of health and hepatitis B seronegativity, being vaccinated against hepatitis B and being infected with hepatitis B was examined with age-adjusted odds ratio (OR) and 95% Confidence Intervals (CI). The data analysis was performed using the SPSS 15.0 program.
4. Results
The results were presented for two separate groups to reflect pre and post-vaccination periods. A preliminary evaluation was performed on the test results to determine the cutoff age. Among the individuals aged 21, 22, and 23 years old, who were expected to be vaccinated in 2014, when blood samples were collected, the percentage of participants with vaccinated profile was 92%, 44%, and 33%, respectively. The low rate of vaccinated profile in the last two ages suggested a failure of catch-up immunization in the adolescents. Therefore, age of 21 years old with higher vaccination level was accepted as the cut-off. The first group consisted of participants between two and twenty-one years of age, born in 1993 and later, who were expected to be vaccinated by routine vaccination program, known as the “young” group. The second group was called the “old” group and consisted of participants above 22 years of age, who were susceptible to infection, although some of them may have been vaccinated by themselves.
The participants were divided to five different groups based on their serological profiles. Individuals with all three negative tests were defined as “seronegative”, individuals with only anti-HBs positivity as “vaccinated”, and individuals with HBsAg and/or anti-HBc positivity as “infected”. The infected group was further divided to three groups within itself; individuals with anti-HBs/anti-HBc positivity as a “past infection”, with HBsAg/anti-HBc positivity as “HBsAg carrier”, and with only anti-HBc positivity as “isolated anti-HBc” group.
Of the study group, 52.1% were female, 77.4% were living in the surrounding districts, 25.9% were out of the workforce, 18.1% were production workers, and 15.3% were agricultural workers (Table 1).
| Characteristic | No. (%) |
|---|---|
| Gender (n = 1230) | |
| Male | 589 (47.9) |
| Female | 641 (52.1) |
| Age group, y (n = 1230) | |
| 2 - 9 | 136 (11.0) |
| 10 - 19 | 189 (15.4) |
| 20 - 29 | 166 (13.5) |
| 30 - 39 | 185 (15.0) |
| 40 - 49 | 200 (16.3) |
| 50 - 59 | 152 (12.4) |
| 60 - 69 | 107 (8.7) |
| 70 - 79 | 72 (5.9) |
| ≥ 80 | 23 (1.9) |
| Area of living (n = 1230) | |
| Rural district | 952 (77.4) |
| City center | 278 (22.6) |
| Area of living during the childhood period (n = 1225) | |
| City center | 226 (18.6) |
| District | 421 (34.4) |
| Village | 567 (46.3) |
| Abroad | 11 (0.9) |
| Educational status (n = 1231) | |
| Illiterate | 99 (8.0) |
| Literate | 37 (3.1) |
| Primary School | 526 (42.7) |
| Secondary School | 176 (14.4) |
| High School | 146 (11.4) |
| University or above | 88 (7.1) |
| Non-school-age | 62 (5.0) |
| Currently primary school student | 95 (7.7) |
| Professional status (n = 1226) | |
| Unemployed (unemployed or housewives looking for a job | 68 (5.5) |
| Individuals out of the workforce (unemployed or housewives not looking for a job | 317 (25.9) |
| Students | 88 (7.2) |
| Production workers (working in jobs in the industry or at a factory, etc.) | 222 (18.1) |
| Skill-requiring/learned professions (doctor, engineer, etc.) | 113 (9.2) |
| Employer (employing less or more than 3 workers, landowners employing workers) | 47 (3.8) |
| Individuals working in jobs without a regular income (salesman, waiter, etc.) | 133 (10.8) |
| Agricultural workers | 188 (15.3) |
| Self-working craftsman (who does not employ any workers) | 50 (4.1) |
| Perceived income level (n = 1226) | |
| Income is more than the expense | 79 (6.4) |
| Income equals the expense | 632 (51.6) |
| Income is less than the expense | 514 (42.0) |
| Annual per capita equivalent income (TL) (n = 1188) | |
| ≤ 3265 | 603 (50.8) |
| > 3265 | 585 (49.2) |
| Household density (n = 1229) | |
| ≤ 1 person per room | 689 (56.1) |
| > 1 person per room | 540 (43.9) |
Hepatitis B seronegativity was 50.8% in the study group (22% in young and 62.3% in the older groups) with the highest in the age group of 30 to 39 years old (77.8%) (Table 2). Of the 77 seronegative participants in the young group, who were considered to be vaccinated, 19 displayed non-protective anti-HBs titers (2 to < 10 IU/L) while the remaining 58 had an undetectable level (< 2 IU/L) of anti-HBs. Of the 548 participants, whose vaccination history was unknown and who were determined to be seronegative in the old group, anti-HBs was found to be 2 to < 10 IU/L in 11 (2%) and < 2 IU/L in the others. Of the 55 participants, who were infected and had isolated anti-HBc profile, anti-HBs was found to be 2 to < 10 IU/L in 16 (29.1%) and < 2 IU/L in the others.
| Hepatitis B Serological Status | Young (Age 2 - 21) | Old (Age 22 - 89) | Total (Age 2 - 89) |
|---|---|---|---|
| Seronegative | 77 (22.0) | 548 (62.3) | 625 (50.8) |
| Vaccinated | 263 (75.1) | 81 (9.2) | 344 (28.0) |
| Infected | 10 (2.9) | 251 (28.5) | 261 (21.2) |
| Past infection | 5 (1.4) | 179 (20.4) | 184 (15.0) |
| HBsAg carrier | 2 (0.6) | 17 (1.9) | 19 (1.5) |
| Isolated anti-HBc | 3 (0.9) | 55 (6.2) | 58 (4.7) |
| Total | 350 (28.5) | 880 (71.5) | 1230 (100) |
aValues are expressed as No. (%).
bHepatitis B infected group consists of HBsAg carriers, immune individuals due to hepatitis B infection, and individuals with isolated anti-HBc positivity.
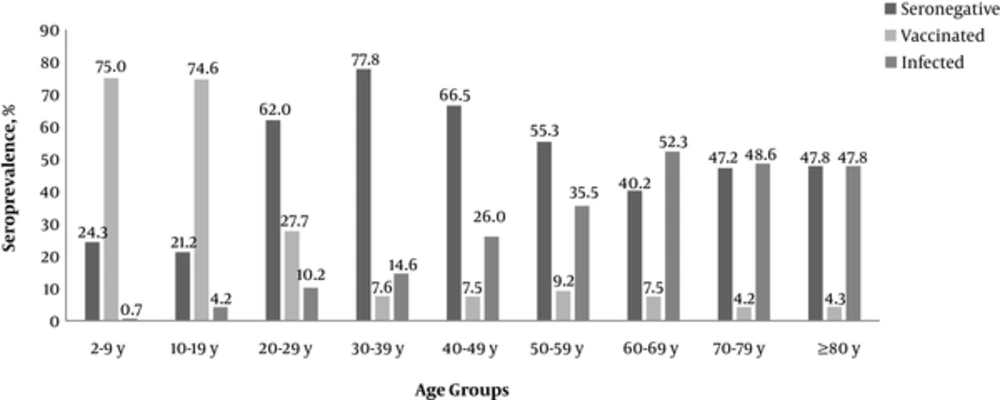
The prevalence of being vaccinated against hepatitis B was 28.0%. The age groups with the highest vaccination rate were 2 to 9 and 10 to 19 year-olds, with a rate of 75.0% and 74.6%, respectively. The prevalence of hepatitis B infection was 21.2%. Age groups of 2 to 9 and 10 to 19 year-olds had the lowest infection rate of 0.7% and 4.2%, compared to age groups of 60 to 69 and 70 to 79, with higher rates of 52.3% and 48.6% (Figure 1).
The relationship between social determinants of health and being vaccinated and infected in the young and old groups is presented in Table 3. In the young group, the vaccination rate was higher in individuals, who lived in a village or rural district during their childhood compared with individuals, who lived in the city center or abroad (OR: 3.13, 95% CI 1.49 to 6.58, OR: 4.46, 95% CI 1.97 to 10.08, respectively). Participants with low annual per capita equivalent income were more likely to be vaccinated (OR: 1.78, 95% CI 1.05 to 3.01).
| Characteristics | Young (2 - 21 Years Old) N = 350 | Old (22 Years and Above) N = 880 | ||
|---|---|---|---|---|
| Being Vaccinated Against Hepatitis B, OR (%95 CI) | Being Infected with Hepatitis B, OR (%95 CI) | Being Vaccinated Against Hepatitis B, OR (%95 CI) | Being Infected with Hepatitis B, OR (%95 CI) | |
| Gender | ||||
| Male | 1.00 (0.59 - 1.68) | 0.78 (0.20 - 3.08) | 1.08 (0.68 - 1.74) | 1.40 (1.03 - 1.92) |
| Area of living | ||||
| Central districts | 1.12 (0.59 - 2.13) | 1.03 (0.19 - 5.44) | 1.02 (0.59 - 1.74) | 0.89 (0.60 - 1.31) |
| Area of living during childhood | ||||
| District | 4.46 (1.97 - 10.08) | 0.41 (0.06 - 2.58) | 2.13 (1.01 - 4.51) | 0.64 (0.42 - 0.97) |
| Village | 3.13 (1.49 - 6.58) | 0.43 (0.09 - 1.99) | 1.79 (0.86 - 3.72) | 0.50 (0.33 - 0.75) |
| Professional status** | ||||
| Employed | 2.17 (0.65 - 7.25) | 4.67 (0.46 - 6.81) | 1.15 (0.71 - 1.87) | 1.27 (0.92 - 1.76) |
| Perceived income level | ||||
| Income ≥ the expense | 0.99 (0.58 - 1.70) | 0.29 (0.07 - 1.20) | 1.49 (0.91 - 2.45) | 0.91 (0.66 - 1.24) |
| Annual per capita equivalent income (TL) ≤ 3265 | 1.78 (1.05 - 3.01) | 2.14 (0.48 - 9.43) | 0.56 (0.34 - 0.91) | 1.12 (0.81 - 1.54) |
| Household density | ||||
| More than 1 person | 1.36 (0.81 - 2.28) | 2.10 (0.49 - 8.91) | 0.81 (0.49 - 1.34) | 1.21 (0.87 - 1.69) |
Abbreviations: CI, Confidence Intervals; OR, Odds Ratio.
aIn the Multinomial Logistic Regression Hepatitis B seronegativity was the reference.
bStudents were excluded from the professional status during the analysis.
cReference categories: Gender- female, Area of living - Rural districts, Area of living during the childhood period- city/abroad, professional status- unemployed/out of the workforce, perceived income- income less than expense, annual per capita income- > 3265 TL, household density- 1or less person.
In the old group, individuals, who lived in the rural district during their childhood were 2.13 times (95% CI 1.01 to 4.51) more likely to be vaccinated for hepatitis B than individuals, who lived in a city or abroad. Vaccination rate was statistically lower in individuals with annual per capita equivalent income of ≤ 3265 TL compared with individuals with higher incomes (OR: 0.56, 95% CI 0.34 to 0.91).
In the older group, males were 1.40 times (95% CI 1.03 to 1.92) more infected with hepatitis B than females. The rate of being infected with hepatitis B was statistically lower in individuals, who lived in a village or rural district during their childhood compared to those who lived in a city or abroad (OR: 0.50, 95% CI 0.33 to 0.75, OR: 0.64, 95% CI 0.42 to 0.97, respectively).
5. Discussion
This study highlighted important findings on hepatitis B seroepidemiology in the population of older than two-year-olds in Manisa. It was shown that vaccine-induced immune response was higher in the age groups that received the routine vaccination program compared with the other age groups.
In the Manisa study, 75% of the population between two and twenty-one years of age were in the vaccinated group. It is possible to conclude that these participants are individually protected from the infection (13). On the other hand, 22% of the young group were seronegative. It is an important issue to lay emphasis on whether these participants are protected from the infection and whether an additional booster dose is needed. To test the immunity of these participants, it is necessary to investigate the presence of an anamnestic response by administering a booster dose (14). Studies investigating the status of the insufficient response of anti-HBs values prior to the booster dose may be indicative (15, 16). A 100% response was obtained with the booster dose in individuals with 1 to < 10 IU/L in one study and 2 to < 10 IU/L in another study. In the study of Manisa, 19 (24.7%) from a total of 77 seronegative participants had an anti-HBs value of 2 to < 10 IU/L. Following the above information, it could be assumed that these participants are protected from infection. Whether the remaining 58 (75.3%) participants need the booster dose has not been clarified yet (17).
The seroprevalence of HBV infected participants among the young group was 2.9% in this study. Although a population-based study representing the Manisa province has not been encountered, the study of Buran et al. performed retrospectively on children, who were admitted to the hospital was partly informative about the Manisa province (18). The authors evaluated hepatitis B serological test results of patients, who were admitted to three state hospitals in Manisa between 2006 and 2012 for various reasons. They grouped the patient results by birth dates and published HBsAg positivity rates by age groups. They reported HBsAg positivity as 1.6%, 0.9%, 0.3%, and 0.6% in cases born between 1993 and 1997, 1998 and 2002, 2003 and 2007, and 2008 and 2012, respectively (18). The results indicated a statistically significant decrease in HBsAg positivity after the routine vaccination program. If similar community-based studies are conducted in Manisa in the coming years, and the results are compared with this study, it will be possible to make a more accurate interpretation of changing trends in hepatitis B seroprevalence for Manisa.
Hepatitis B virus infection is strongly associated with low socioeconomic status. The effect of poverty on HBV endemic is observed mainly in the young age group (8). In the Manisa study, however, there was no relationship between the social determinants of health and being infected in the young group, in which the vaccination rate was relatively high. Similarly, in a study conducted on children in Erzurum province, no significant relationship was found between HBV carriage and socioeconomic status, suggesting that the development of sanitation conditions in the last decade may have reduced the seroprevalence of HBV infection (19). In the present study, living in a village or rural district during childhood and having a low income were significant determinants of being vaccinated. This result can be an indirect evidence of the efficiency of primary health care services provided for disadvantaged groups of population in Turkey.
In the Manisa study, of the participants between 22 and 89 years of age, named as the old group, 62.3% were seronegative, 28.5% were infected, and 9.2% were vaccinated. These results were similar to the results of the TURHEP study carried out nationwide on individuals aged over 18 years old (4). The rates of seronegativity, being infected, and vaccinated in the TURHEP study were reported to be 59.5%, 30.6%, and 8.4%, respectively. In TURHEP, HBsAg positivity in Turkey was 4.0% and 2.3% in the Aegean region. In the Manisa study, HBsAg positivity was 1.9%. The difference may be a result of Manisa’s hygiene conditions being better when compared to the other Aegean provinces.
In Manisa and TURHEP studies, the relationship of some common social determinants that may be associated with HBsAg positivity was examined. In the TURHEP study, being a male, married, and having high school and below education were associated with HBsAg positivity (4). However, in the Manisa study, while a relationship was found between being infected with hepatitis B and being a male and living in a city/abroad during childhood in the old group, no relationship was found with other social determinants of health, including household density and professional status. This finding may indicate that males exhibit more repetitive risky behaviours than females, and that crowded urban life increases HBV carriage (8). In the Manisa study, there was a relationship between high income and being vaccinated against hepatitis B in the old group. Since there was no vaccination program in the older group, it was thought that they could have access to the vaccine by paying for it.
Although there are studies on hepatitis seroepidemiology in Turkey, the generalizability of their results is limited, since the majority of studies are based on patients admitted to the hospital, they have a small sample size, sample selection is not performed, or the selected sample group does not represent the population (5). The relatively large study population that was randomly selected from the population was the strength of the Manisa hepatitis B study. Another power of the study was examining the association between social determinants of health and being hepatitis B seronegative, vaccinated or infected.
The main limitation of the study was that no medical history was obtained from individuals on the possible mode of transmission, vaccination, and disease symptoms. The researchers assumed that this information could not be remembered. Another limitation of the study was that approximately 30% of the sample population could not be reached or included in the study. The relatively low response rate was mostly due to refusal to participate in the study. In population-based studies like the Manisa study, response rates are getting lower in many countries. The current study compared individuals, who participated and did not participate in the study regarding age and gender. There was no difference in mean age between the individuals, who participated and did not participate in the study (P: 0.69), yet the participants of the study were mainly females (P < 0.001). However, this may not have changed the overall results of this study. Another limitation of the study was that no intensified data from people, who were engaged in high-risk behaviors, such as injection drug users, persons with multiple sexual partners, males, who had sex with males or who were high-risk individuals for hepatitis B, like health-care workers, patients with hemodialysis, were collected (20).
5.1. Conclusions
The Manisa Study provided up-to-date data on hepatitis B seroepidemiology in a sample representing the general population. It could be concluded that routine hepatitis B vaccination initiated 16 years ago induced high levels of immunity in the great majority of the population aged < 22 years living in Manisa. Repeated population-based seroprevalence studies are useful in monitoring the impact of the vaccine programs, determining the modifications needed in the vaccination schedule for the control of hepatitis B infections in the society.