1. Context
Non-alcoholic fatty liver disease (NAFLD) is the hepatic manifestation of metabolic syndrome as the main complication of obesity. Parallel to the global epidemic of obesity, NAFLD is becoming the main liver disease and the main cause of liver transplantation worldwide (1). Universal NAFLD prevalence is 24% - 25% (2). NAFLD mirrors a spectrum extending from simple steatosis to non-alcoholic steatohepatitis (NASH) with the capability of progressing toward cirrhosis and even hepatocellular carcinoma (3). More than 80% of the patients with obesity submitted to bariatric surgery suffer from NAFLD, with 25% - 55% resulting in NASH and 2% - 12% resulting in fibrosis and cirrhosis (4).
Sustained weight loss through bariatric surgery is the most effective and durable management of this widespread and potentially dangerous disease (5).
Approximately during the past three decades, much more information has become available concerning the histologic effects of bariatric interventions on the liver of obese patients via sequential biopsies. In 2008, Mummadi et al. published a systematic review in which they included all 15 paired-biopsy studies available until that time, excluding cirrhotic patients. They claimed that the histopathologic features of NAFLD improved in more than 75% of the cases of less advanced liver disease (6). However, two years later in a Cochrane review, due to the lack of eligible studies, comments on the positive or negative outcomes of bariatric surgeries in NASH patients were avoided (7). Despite the limitations, the heterogeneity of the existing studies, and the lack of randomized controlled trials, the last attempt to illuminate the influence of bariatric surgeries on liver histology and biochemistry was performed in 2015 by Bower et al. They conducted a meta-analysis of 29 studies covering all the previous paired biopsy studies along with biochemistry-based interventions. They announced a significant improvement in steatosis, steatohepatitis, and fibrosis accompanied by an improvement in levels of aspartate aminotransferase, alanine aminotransferase, and gamma-glutamyl transferase (8).
Apart from multiple reports regarding jejunoileal and jejunocolic bypass bariatric procedures that were formerly abandoned due to several metabolic side effects such as hepatic failure, the clinical outcomes of current bariatric surgeries on the liver tends to improve. However, sometimes, we encounter reports in the literature regarding individuals experiencing bariatric surgeries presenting with hepatic impairment such as serious complications that can lead to liver transplantation or even death. Although hepatic impairment following biliopancreatic diversion with duodenal switch (BPD/DS), and Roux-en-Y gastric bypass (RYGB) was recently reviewed (9, 10), the evidence regarding this event is distracting, and our information about the underlying mechanisms, possible risk factors, and best managements is restricted to each author’s discussion.
This paper systematically reviews the data from two main sources, including paired-biopsy studies and post-bariatric-surgery hepatic failure case reports, with the aim of elucidating the hepatic effects of bariatric surgeries from another perspective.
2. Evidence Acquisition
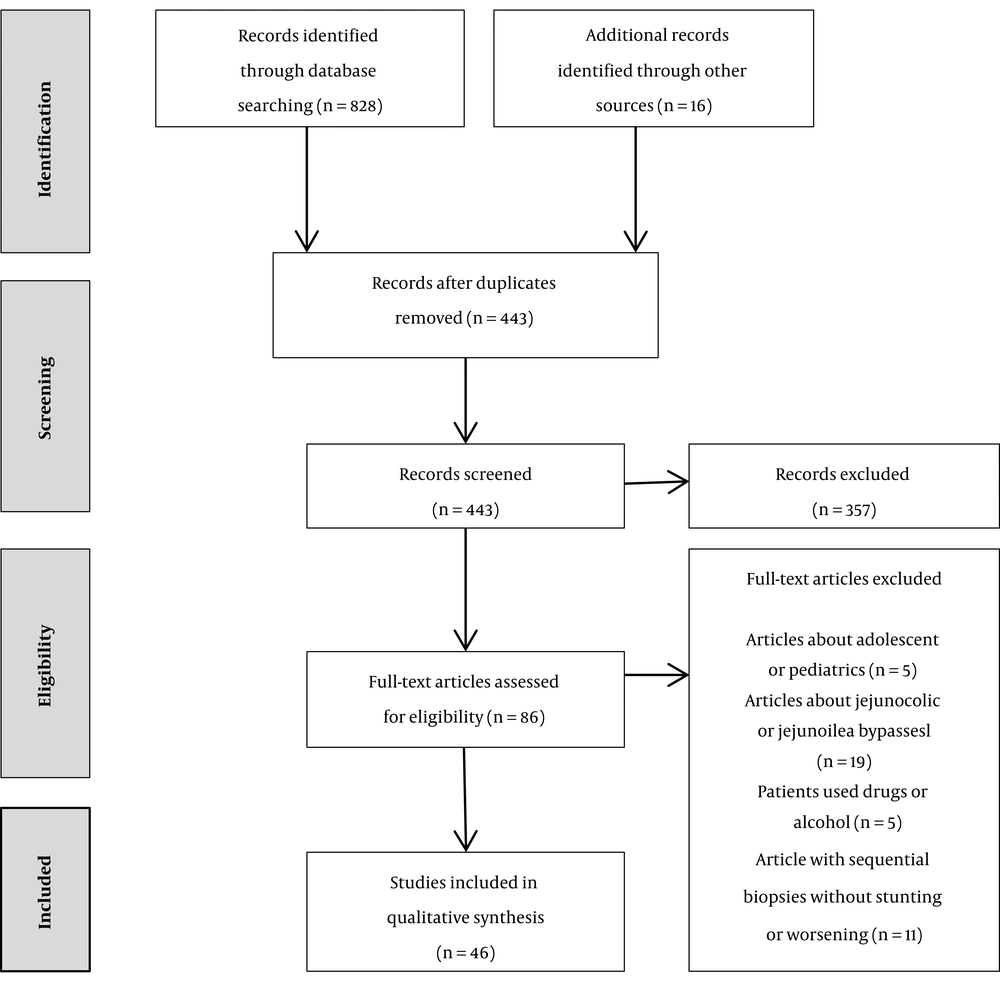
Online electronic searches were conducted in MEDLINE (via PubMed), Google Scholar, Scopus, and the Cochrane Controlled Trials Register to identify studies that provided information about histologic stunting or deterioration after any bariatric surgery through sequential liver biopsies and studies that reported hepatic dysfunction (clinical or pathological) following any of the bariatric surgical procedures up to March 2018. The literature search was concomitant using MeSH terms and was based on keywords as follows: “bariatric surgery”, “obesity surgery”, “Roux-en-Y gastric bypass”, “mini gastric bypass”, “biliopancreatic diversion”, “duodenal switch”, “sleeve gastrectomy”, “nonalcoholic fatty liver disease”, “steatohepatitis”, “liver failure”, and “cirrhosis”. Boolean logic was used to combine keywords. The limitations “humans” and “English language” were applied. The full versions of the review articles were checked to find other qualified studies. We excluded the studies if jejunoileal or jejunocolic bypasses included the bariatric surgical procedures or performed on pediatrics or adolescents. In addition, they were excluded if alcohol, drug abuse, pregnancy, hepatotropic viruses, and autoimmunity were the leading causes of hepatic failure in the case reports or the patients had a positive history of hepatic disease prior to bariatric surgery. The study attrition diagram is shown in Figure 1 according to the preferred reporting items for systematic reviews and meta-analyses (PRISMA).
3. Results
Among the paired-biopsy studies that explained the impact of bariatric surgeries on the NAFLD spectrum, 23 studies contained a stunting or deterioration report of second hepatic pathology in any pathological classification. They are summarized in Table 1.
| Author, Year | Study Type | Operation | Number; Female:Male | Age, y | Pre and Post-op. BMI, kg/m2 | Follow-up Duration | Pathologic Result | Explanation | ||
|---|---|---|---|---|---|---|---|---|---|---|
| Steatosis | Inflammation | Fibrosis | ||||||||
| Silverman et al. 1995 (11) | RC | RYGB | 91; 87:4 | 39 | 47 - 33.5 | 18.4 m | Improved | Worsened | Improved | Stunting: 5/73 (6.1%) of HS, 2/12 (16%) of PSF, and 2/9 (22%) of PF; worsening: 3/73 (4%) of HS and PI, and 1/13 (7.7%) of PSF |
| Luyckx et al. 1997 (12) | PC | VBG or AGB | 69; 59:10 | 36 ± 11 | 43.9 ± 8.3; 31.7 ± 4.1 | 27 ± 15 m | Improved | Worsened | NC | Stunting: F; worsening: LI (14% to 28%) |
| Dixon et al. 2004 (13) | PC | LAGB | 36; 25:11 | 43 ± 1.3 | 47 ± 10.6; 34 ± 5.5 | 25.6 ± 10 m | Improved | Improved | Improved | Stunting: PI and PF |
| Kral et al. 2004 (14) | PC | BPD; BPD/DS | 104; 84:20 | 40.3 ± 9 | 47 ± 8.4; 31 ± 8 (kg WL) | 41 ± 25 m | Improved | Improved | Worsened | Stunting: 34/104 (32.7%) of F; worsening: 10/104 (11.6%) of I and 42/104 (40.4%) of F and three new onset cirrhosis cases |
| Clark et al. 2005 (15) | PC | RYGB | 16; 8:8 | 43.9 ± 8.1 | 51.1 ± 6.1; 32.9 ± 5.1 | 305 ± 131 d | Improved | Improved | Improved | Stunting: 3/15 (20%) of I, 2/14 (14%) of BD, 8/14 (57%) of PSF and 7/13 (54%) of PF |
| Mottin et al. 2005 (16) | RC | RYGB | 90 | 35.6 | 46.7 - 81.4 (%EWL) | 12 m | Improved | NR | NR | Stunting: 16/90 (17.8%) of HS |
| Mattar et al. 2005 (17) | PC | RYGB (41) SG (23); AGB (6) | 70; 48:22 | 49 ± 9 | 56 ± 11 39 ± 10 | 15 ± 9 m | Improved | Improved | Improved | Stunting: 2/70 (2.8%) of F (in stage 4 fibrosis) |
| Stratopoulos et al. 2005 (18) | PC | VBG | 51; 33:18 | NR | 52.8 ± 1; 66% EWL | 18 ± 9.6 m | Improved | Improved | Improved | Stunting: 8/51 (15.6%) of HS, 7/51 (13.7%) of I and 21/51 (41.1%) of F; worsening: 6/51 (11.7%) of F |
| Barker et al. 2006 (19) | PC | RYGB | 19; 17:2 | 48.6 | 46.8 - 28.8 | 21.4 m | Improved | Improved | Improved | Stunting: 4/19 (21%) of PF; worsening: 1/11 (9%) of LF and 1/19 (5.2%) of PF |
| Klein et al. 2006 (20) | PC | RYGB | 7; 6:1 | 40 ± 5 | 58 ± 4 - 41 ± 5 | 12 m | Improved | NC | NC | Stunting: I and F |
| Cendes et al. 2006 (21) | PC | RYGB | 16; 15:1 | 46.2 | 44.3 - 28.6 | 17.5 m | Improved | Improved | NC | Stunting: F, 1/16 (6.7%) of HS and 1/16 (6.7%) F; worsening: 1/16 (6.7%) HS |
| de Almedia et al. 2006 (22) | PC | RYGB | 16; 14:2 | 40.2 ± 9.5 | 53.4 ± 8.8; 31.1 ± 4.7 | 23.5 ± 8.4 m | Improved | Improved | NC | Stunting: 2/4 (50%) of F; Worsening: 1/4 (25%) B |
| Liu et al. 2007 (23) | RC | RYGB | 39; 33:6 | 41.1 ± 9 | 47.7 ± 6.2; 29.5 ± 5.6 | 18 m | Improved | NC | Improved | Stunting: 10/16 (62.5%) of PI, 2/15 (13.3%) of PSF and 8/16 (50%) of PF; worsening: 6/39 (15.4%) of PI, 2/39 (5.1%) of LI and 1/16 (6.25%) of PF |
| Furuya et al. 2007 (24) | PC | RYGB | 18; 17:1 | 46.6 ± 7.3 | 51.7 ± 7.4; 31 ± 2 | 24 m | Improved | NC | Improved | Stunting: LI (81%) |
| Mathurin et al. 2009 (25) | PC | LAGB(214) BIB (87) RYGB (80) | 381 | 41.5 ± 9.6 | 50 ± 7.8; 39 ± 8.2 (1y); 37.7 ± 8.4(5y) | 1 , 5 y | Improved | NC | Worsened | Stunting: I; worsening: F |
| Moretto et al. 2012 (26) | RC | RYGB | 78; 59:19 | 39.5 ± 11.4 | 45.4 ± 8.1; 29.3 ± 5.8 | NR | NR | NR | Improved | Stunting: 10/31 (32.2%) of LF; worsening: (12.9%) of LF and 5/43 (11.6%) of F |
| Tai et al. 2012 (27) | PC | RYGB | 21; 13:8 | 29.9 ± 8.1 | 43.8 ± 7.5; 28.3 ± 4.6 | 12 m | Improved | Improved | Improved | Stunting: 1/19 (5.2%) of HS, 7/18 (38.9%) of BD and 1/15 (6.7%) of LI; worsening: 5/15 (33.3%) of LI |
| Vargas et al. 2012 (28) | PC | RYGB | 26; 19:7 | 45 ± 2 | 49.3 ± 4.8; 30.9 ± 4.3 | 16.3 ± 3 m | Improved | Improved | Improved | Stunting: F (in five patients) |
| Taitano et al. 2015 (29) | PC | Long limb and short limb; RYGB; LAGB | 160; 134:26 | 47 ± 12 | 52 ± 10; 33 ± 8 | 31 ± 26 m | Improved | Improved | Improved | Stunting: 46/160 (28.8%) of HS, 45/160 (28%) of I, and 68/158 (43%) of F; worsening: 8/160 (8%) of HS, 3/156 (1.9%) of I, and 25/158 (15.9%) of F |
| Praveenraj et al. 2015 (30) | PC | RYGB (10); SG (20) | 30; 15:15 | 43.3 | 45.9 ± 11.7; 35.3 ± 8.1 | 7.1 m | Improved | NC | Improved | Stunting: 19/24 (79%) of PI |
| Froylich et al. 2015 (31) | RC | RYGB (14); SG (11) | 25; 16:9 | 56.2 ± 8.6 | 51 ± 13.5; 34.5 ± 11.3 | 1.7 ± 0.7 y | Improved | Improved | NC | Stunting: F |
| Aldoheyan et al. 2016 (32) | PC | SG | 27; 18:9 | 35 ± 8 | 44.6 ± 7.8; 34.2 ± 6.3 | 3 m | Improved | NC | Improved | Stunting: I |
| Schneck et al. 2016 (33) | PC | RYGB | 9; 9:0 | 51 | 42 - 27.1 | 45 m | Improved | Improved | Improved | Stunting: 1/9 (11.1%) of BD; worsening: 1/9 (11.1%) of F |
Abbreviations: BD, ballooning degeneration; BIB, biliointestinal bypass; BMI, body mass index; BPD/DS, biliopancreatic diversion/duodenal switch; d, day(s); %EWL, percentage excess weight loss; F, fibrosis; GB, gastric bypass; HS, hepatic steatosis; I, inflammation; LAGB, laparoscopic adjustable gastric banding; LI, lobular inflammation; m, month(s); NC, not changed; NR, not reported; PC, prospective cohort; PF, portal fibrosis; PI, portal inflammation; PSF, perisinosoidal fibrosis; RC, retrospective cohort; RYGB, Roux-en-Y gastric bypass; SG, sleeve gastrectomy, VBG, vertical banded gastroplasty.
Other than hepatic decompensations after post-jejunoileal and post-jejunocolic bypasses that were excluded from the review, hepatic impairment following other bariatric surgeries was noted implicitly through some studies (34, 35). However, we reviewed the studies that reported the post-bariatric hepatic failure as an explicit target event. Table 2 lists 23 studies accounting for 59 cases who exhibited symptoms attributable to hepatic deterioration following one of the bariatric procedures.
| Author/Year/Location | Operation | Age, (y); Female/Male | ∆BMI, kg/m2 | Signs and Symptoms | Paraclinical Data | Index Biopsy | Post-Hepatic Failure Biopsy | Duration | Management and Outcome |
|---|---|---|---|---|---|---|---|---|---|
| Grimm et al. 1992 USA (36) | BPD | 43 F | 54.5 -27.6 | Weakness, jaundice, edema alopecia, temporal wasting, cheilosis, atrophic glossitis, ascites, RUQ tenderness, HE | T. Bili: 9.8 (D: 6.4); Alb: 2.1; ALP: 127 AST/ALT: 91/28 Plt: 212; HCT: 34.1 PT: 17 (n: 13) | Mild HS | Hepatocyte degeneration, Mallory bodies, Periportal and lobular PMN infiltrates, cholestasis | 10 m | TPN, vitamin supplementation, Metronidazole, Pancreatic enzyme-LT candidate-died |
| Langdon et al. 1993 USA (37) | BPD | 53a F | NR | Diarrhea, anasarca, cutaneous ulcer, HE | GGT and ALP: 1.5 - 2 of normal AST/ALT: normal or near normal; Alb: ↓; PT: mildly prolonged | HS | HS without cirrhosis | 3 y | TPN-reversion candidate-died |
| 44b F | NR | NR | HS | HS without cirrhosis | 6 y | EN-Gastrogastrostomy hole-reversion-died | |||
| Castillo et al. 2001 Spain (38) | BPD | 41 F | 46 - NR | Vomiting, weakness, abdominal pain, myalgia, jaundice, ascites | T. Bili: 13.4; ALP: 476 AST/ALT: 302/283; INR: 2 Alb: 2.; Plt: 120 | Mild HS | Submissive necrosis, periportal and lobular PMN, infiltration and cholestasis | 12 m | LT, shortening of bypassed bowel (40 cm) |
| Baltasar et al. 2003 Spain (39) | BPD/DS | 43 F | 61 - NR | jaundice | T. Bili: 6.7 (D: 4.7); ALP:240; AST/ALT: 146/61; INR: 1.2 | NR | HS3 L2 P2 F2 | 7 m | Improved with nutritional assistance |
| 45 F | 40 - NR | jaundice | T. Bili: 10.9 (D: 7.3); INR:1.3; ALP:230; AST/ALT: 803/800 | NR | NP | 1 m | Improved with nutritional assistance | ||
| 26 F | 44 - NR | jaundice | T. Bili: 23 (D:14); ALP: 667 AST/ALT: 2644/4240; INR:1.4 | NR | HS3 L2 P2 F2 | 14 w | Improved with nutritional assistance | ||
| 42 F | 49 - NR | jaundice | T. Bili: 22.6 (D: 20.3) ALP: 435; AST/ALT: 1188/1194 INR: 1.7 | NP | NP | NR | Improved with nutritional assistance | ||
| 41 F | 64 - NR | Persistent vomiting, jaundice, encephalopathy | T. Bili: 18.9 (D: 12.6); ALP: 644 AST/ALT: 236/123; INR: 1.64 | NR | HS3 L0 P0 F0 | 3 m | LT candidate-died | ||
| 31 F | 41 - NR | Generalized malaise, fever, weakness, anorexia | T. Bili: 12 (D: 8.6); ALP: 330 AST/ALT: 700/700; INR: 1.4 | NR | HS3 L1 P1 F2 | 2 w | Improved | ||
| 34 M | 45 - NR | Jaundice, prolonged fever for 30 days | T. Bili: 29.6 (D: 19.5); ALP: 353; AST/ALT: 716/1179; INR: 1.4 | NP | HS3 L2 P3 F3 | 10 d | Improved | ||
| 20 F | 60 - NR | Nausea, vomiting, wernicke-korsakoff neuropathy | T. Bili:1.2 (D: 0.66); ALP: 120 AST/ALT: 692/1406; INR: 1.31 | NP | NP | 1 m | Improved with adequate supplementation | ||
| 25 M | 47 - NR | Asymptomatic | T. Bili: 2.5 (D: 1.6); ALP: 126 AST/ALT: 189/766; INR: 2.3 | NP | NP | 1 m | Improved with supportive care | ||
| 50 F | 61 - NR | Asymptomatic | T. Bili: 1.6 (D: 0.9); ALP:383 AST/ALT: 1126/1658; INR:1.4 | NP | HS3 L2 P1 F3 | NR | Improved spontaneously | ||
| Cotler et al. 2004 USA (40) | RYGB | 34 F | 86 - NR | Muscle wasting, weakness, ascites, anasarca, tender hepatomegaly, ↓oral intake | T. Bili: 1.6 (D: 0.8); T.pro:7.5; AST/ALT: 74/21; Alb: 1.6; Plt:91; BUN/Cr: 11/0.9 | NR | Steatosis with steatohepatitis and cirrhosis | 17 m | LT candidate |
| 37 F | 61 - NR | Muscle wasting, jaundice, tender hepatomegaly, renal failure, ↓oral intake | T. Bili:13.3 (D: 8.1); T. pro: 5.8 AST/ALT:137/70; Alb: 1.5; Plt: 112; BUN/Cr: 4/3.1 | NR | Massive HS and mild F | 7 m | EN, volume repletion, lactulose, antibiotic-improved | ||
| 54 F | 49 - NR | Muscle wasting, jaundice, ascites, encephalopathy, renal failure, ↓oral intake | T. Bili: 11.7 (D: 6.9); T. pro: 5.9; AST/ALT: 50/26; Alb: 2.7 Plt: 136; INR: 2; BUN/Cr: 10/1.9 | NR | Severe steatosis with steatohepatitis and cirrhosis | 7 m | Medical therapy-diedc | ||
| D’Albuquerque et al. 2008 Brazil (41) | BPD | 29 F | 63 - 26 | Encephalopathy, anasarca, ascites | NR | NR | Active cirrhosis with inflammatory activity | 10 m | LT, reversion |
| 29 F | 60 - 35 | Anasarca, ascites, jaundice, shock, encephalopathy | NR | NR | Cholestatic acute hepatitis, intense macrovesicular HS | 6 m | LT candidate-died | ||
| 38 F | 56 -29.9 | Visceromegaly, encephalopathy | LFT: Impaired; MELD: 44.1 | NR | Massive necrosis, diffuse septa formation | 7 m | LT | ||
| Aasheim et al. 2010 Norway (42) | BPD/DS | 41d F | 57- NR | Ascites, Moderate encephalopathy | T. Bili: 19; Alb: 20; INR:1.7; child score: Class C | NR | NASH and bridging fibrosis, cirrhosis | 11 m | Improved to compensated cirrhosis |
| Geerts et al. 2010 Belgium (43) | BPD | 52 F | 65 - 41 | NR | NR | NP | NR | 13 m | LT, reversion |
| 38 M | 48 - 23 | NR | NR | NP | NR | 27 m | LT, reversion | ||
| 29 F | 40 - 20 | NR | NR | NP | NR | 84 m | LT candidate | ||
| 19 F | 41- 20 | NR | NR | NP | NR | 62 m | LT-retransplantation, reversion eight weeks later | ||
| 46 F | 55 - 29 | NR | NR | NP | NR | 11 m | LT, reversion | ||
| 53 F | 40 - 24 | NR | NR | NP | NR | 18 m | LT, reversion | ||
| 35 F | 45 - 25 | NR | NR | NP | NR | 20 m | LT, reversion | ||
| 38 F | 40 - 22 | NR | NR | NP | NR | 21 m | Died | ||
| 40 F | 47 - 25 | NR | NR | NP | NR | 50 m | Died | ||
| Sagredo et al.e 2012 Chile (44) | GBS | 28 F | NA | NR | NR | NR | Severe steatohepatitis and fibrosis | 11 m | Improved with supportive care, EN, N acetylcysteine, zink, Vit. E |
| Sgambato et al. 2013 Italy (38) | BIB | 42 F | 54 - NR | Diarrhea, jaundice, paleness, splenomegaly, asthenia, hepatomegaly | T. Bili: 0.14 (n < 0.01) INR:1.9; AST/ALT: 135/150; Plt: 54; Alb: 31(35 -55); Hb:72(126 - 160) | NP | NP | 8 m | LT, reversion |
| Auclair et al. 2013 Canada (45) | BPD/DS | 37 F | 54 - 30 | Nausea, vomiting, ascites, abdominal pain, jaundice, dark urine, pale stool, pruritus | T. Bili: 136 (7 - 23); T.pro: 5.9 AST/ALT: 1115/1169; INR:1.9; Alb: 27 (36 - 45) Cr: 52 (53 - 112) | NP | Acute hepatitis, confluent bridging necrosis, mixed inflammatory infiltrate | 8 m | Medical therapy-LT |
| Baltasar 2014 Spain (46) | BPD/DS | 33 F | 49 - NR | Progressive jaundice, encephalopathy | T. Bili: 508 (0 - 18); D.Bili: 217 (0 - 8); INR: 6.5 | NR | Hepatocyte disappearance, cholangiolar metaplasia, severe bile stasis | 20 m | LT, CC lengthening (> 200 cm) |
| 41 F | 58 - 39 | Persistent vomiting, jaundice, encephalopathy | T. Bili: 18.9 (D: 12.6); ALP: 383 AST/ALT: 236/12; INR: 1.64 | NR | NASH-No postmortem pathology | 6 m | TPN-LT candidate -died | ||
| Marszalek et al. 2015 Poland (47) | RYGB | 56f F | 50.9 - 24.5 | NR | NR | NASH HS2 F3 | NR | 2 y | LT |
| Ossorio et al. 2015 Spain (48) | Distal GB | 42 F | 51 - NR | Wall abccess, entrocutnous and AL to CC fistula | AST/ALT: ↑; coagulopathy | NR | NR | 3 y | Abdominal wall mesh extraction, fistula closure-sepsis-died |
| 36g F | 52.2 - NR | Vomiting, jaundice, ascites, encephalopathy | NR | NR | NR | 2 m | Died | ||
| Silva et al. 2016 Spain (49) | BPD | 35 M | NR - 18 | NR | MELD: 40 | NR | Submassive necrosis and severe macro-microvesicular HS | 12 m | LT, reversion-died in refractory septic shock |
| Ralki et al. 2016 Belgium (50) | Long limb GB | 38 F | 39.5-22.9 | Diarrhea | T. Bili: 7.8; ALP: 118 AST/ALT:767/1438; GGT: 162; INR:1.5 | HS | NR | 7 m | Medical therapy-LT |
| Lefere et al. 2017 Belgium (51) | BPD | 24 F | 40 - 20.2 | Jaundice, encephalopathy | T. Bili: 11; Alb: 39 INR: 2.09; Cr: 0.62 Plt: 109; MELD: 24 | NR | At 1st OLT: pericellular and ceptal F, macrovesicular HS At Reversion: severe NASH | 5 y | LT-Reversion eight weeks later-SIBO treatment-Retransplantation 10 m afterward-Died |
| Tsai et al. 2017 USA (52) | Long limb RYGB | 33 F | 45.3 - 38.4 | Jaundice, nausea, vomiting, encephalopathy | T. Bili: 11.7; ALP: 189 AST/ALT: 105/39; Alb: 1.8 | NR | Stage 3 F, panacinar HS, ballooning, ductular reaction, Mallory bodies | 5 m | Died |
| RYGB | 37 F | 59.4 - 26.5 | Jaundice, asterixis, ascites, hepatomegaly | T. Bili: 27.6; ALP: 124 AST/ALT: 80/143; Alb: 2.4 | NR | Stage 4 F, panacinar HS, ballooning, ductular reaction, Mallory bodies | 3 y | LT | |
| RYGB | 37 F | 46 - 32 | Wernicke-Korsakoff neuropathy, Portal HTN, hepatomegaly | T. Bili: Nl; AST/ALT: ↑; coagulopathy | NR | Stage 3 - 4 F, ballooning, Mallory bodies | 7 y | Medical therapy-improved | |
| Kermansaravi et al. 2017 Iran (53) | MGB | 29 F | 55.7 - NR | Dyspnea, jaundice, nausea, vomiting, diarrhea, edema, mild abdominal tenderness | T. Bili: 7.7 (D: 4.8); ALP: 342; AST/ALT: 28/22; Alb: 2.3; Hb: 7; Plt: 195 | NR | PF (grade 3/3 and stage 2/4) | 8 m | Medical therapy-LT candidate-died |
| Kalantar et al. 2017 Iran (54) | MGB | 37 F | 44 - 27 | Generalized fatigue, weakness, jaundice, lower extremity edema | T. Bili: 1.1 - 7.5; ALP: 395 AST/ALT: 43/60; Plt: 55; Hb: 8; PT: 10 Alb: 2.4 - 1.8 - 1.7 | NR | Nonspecific inflammation in portal spaces and fatty change | 12 m | High protein regimen-gastrogastrostomy-died |
| Kalantar et al. 2017 Iran (55) | MGB | 57 F | 42.8 - 25.7 | Nausea, vomiting, edema, vague abdominal pain | T. Bili: 1.5; Hb: 11.2; T. pro: 5.2; Alb: 2.5 AST/ALT: 33/38; ALP: 82; MCV: 86.9; Plt: 314 | NAS: 2/8 | NAS: 7/8 | 8 m | TPN-gastrogastrostomy-improved |
| Eilenberg et al. 2017 Austria (56) | RYGB | 22 - 66; 8 F and 2 M | 48.2 - 21.9 | Dysphagia, epigastric pain, diarrhea, sarcopenia, leg edema, hepatomegaly | Alb: ↓; Plt: ↓; coagulopathy | NR | 90 to 100% micro and macrovesicular HS | 88 | |
| EN - Reversion | |||||||||
| MGBh | 33.3 - 22 | Sarcopenia, leg edema, splenomegaly, variceal bleeding, ascites, HE | Alb: ↓; Plt: ↓; coagulopathy | NR | Cirrhosis | 5 m | LT | ||
| MGB | 42.9 - 20.8 | Ascites, hepatosplenomegaly | AST/ALT: ↑; pancytopenia | NR | Cirrhosis | 12 m | Conversion to RYGB + CC lengthening | ||
| Distal GBi | 44.5 - 28.7 | General fatigue, leg edema | AST/ALT: ↑ Alb: ↓ coagulopathy | NR | NR | 12 m | EN-CC lengthening | ||
| Distal GBj | 28.1 - 20.3 | Diarrhea, dumping syndrome, leg edema, hepatomegaly | Alb: ↓; coagulopathy | NR | NR | 6 m | CC lengthening | ||
| RYGB | 58 - 28.7 | Epigastric pain, leg edema, HE, ascites, hepatosplenomegaly | AST/ALT: ↑; Alb: ↓; Plt: ↓ coagulopathy | NR | NR | 2 m | Improved | ||
| RYGB | 53.4 - 26.6 | Fatigue, ascites, Pleural effusion, sarcopenia, leg edema, hepatosplenomegaly | AST/ALT: ↑; Alb: ↓; Plt: ↓ | NR | 20% macrovesicular HS, NAS: 3/8 | 24 m | EN-CC lengthening | ||
| MGB | 40.8 - 21.7 | Fatigue, ascites, leg edema, sarcopenia, steatorrhea, diarrhea, splenomegaly | Alb: ↓; Plt: ↓; coagulopathy | NR | Cirrhosis | 35 m | Conversion to RYGB, CC lengthening | ||
| MGBk | 57.6 - 36.8 | Diarrhea, dysphagia, epigastric pain, sarcopenia, pleural effusions, ascites, steatorrhea, diarrhea, HE | AST/ALT: ↑; Alb: ↓ | NR | 90 to 100% micro and macrovesicular HS | 41 m | Conversion to RYGB, CC lengthening | ||
| MGB | 64 - 30.5 | Fatigue, dysphagia, leg edema, jaundice, pulmonary effusion, sarcopenia, ascites hepatosplenomegaly | AST/ALT: ↑; Alb: ↓; Plt: ↓; coagulopathy | NR | 85% macrovesicular HS, NAS: 7/8 | 20 m | Conversion to RYGB, CC lengthening | ||
| Lammers et al. 2018 Netherlands (57) | Distal GBl | 43 F | 32 - ? | Generalized edema, depression, anorexia, encephalopathy | T. Bili:53 (n < 17); ALP: 103 AST/ALT: 25/21; INR > 7 Alb: 12(n > 35); GGT: 76(n < 35) | NR | NP | 2 y | EN, lctulose, rifaximin-LT candidate-died |
| GBm | 34 F | 31 - 16 | Vomiting, depression, anorexia, anasarca, encephalopathy | T. Bili:9 (n < 17); ALP: 149 AST/ALT: 43/54; INR > 7 Alb: 10 (n > 35); GGT:55 (n < 35) | NR | NR | 28 m | EN, lctulose, rifaximin-died |
Abbreviations: AL, alimentary limb; BIB, biliointestinal bypass; BMI, body mass index; BPD/DS, biliopancreatic diversion/duodenal switch; CC, common channel; d, days; EN, enteral nutrition; F, fibrosis; GB, gastric bypass; HS, Hepatic steatosis; HE, hepatic encephalopathy; L, lobular inflammation; LT, liver transplantation; m, month(s); MGB, mini gastric bypass; NA, not available; NAS, NAFLD activity score; NASH, nonalcoholic steatohepatitis; NP, not performed; NR, not reported; P, portal inflammation; RYGB, Roux-en-Y gastric bypass; SIBO, small intestinal bacterial overgrowth; TPN, total parenteral nutrition; y, year(s); w, weeks.
aShe had a history of GB seven years before BPD.
b She had a history of vertical banded gastropexy before BPD.
cGastric remnant carcinoma with peritoneal metastases found in the autopsy.
dShe had a history of hyperinsulinemic hypoglycemia.
eThe main article is not written in English, so data were driven from the English abstract.
fShe had a questionable history of alcohol abuse.
gShe had a history of vertical banded gastroplasty before distal GB.
hThe patient had a history of gastric banding for 144 months that was removed and underwent MGB due to weight regain, dysphagia, band infection, pancytopenia, and elevated liver enzymes.
iThe patient had a history of RYGB for 85 months that was converted to distal GB due to weight regain.
jThe patient had a history of RYGB for 96 months that was converted to distal GB due to weight regain.
kThe patient had a history of gastric banding for 108 months that was removed and underwent MGB due to no weight loss.
lThe patient had a history of endoscopic gastric bypass for one year that underwent banded gastric bypass due to insufficient weight loss (∆BMI: 59 - 47) and revised to distal GB because of abdominal pain and dysphagia after other one year.
mThe patient had a history of SG that was revised to GB after five months due to anastomotic leakage, abdominal sepsis, and recurrent esophageal stenosis with stenting. An esophageal-jejunostomy was added three months later because of recurrent nausea and vomiting resulting from gastrojejunal ulceration distal to the esophageal stent.
4. Discussion
4.1. Possible Underlying Mechanisms
Due to the paucity of strong evidence, the influence of bariatric surgeries on the hepatic pathology remains controversial, as most of the time, it leads to improvement and sometimes can result in impairment (Table 1). The possible underlying mechanisms of bariatric surgery-induced hepatic deterioration can be explained under the following three headings: (1) rapid and drastic weight loss, (2) protein-calorie malnutrition, and (3) gut microbiota alteration and bacterial overgrowth.
From the era of the “two-hit hypothesis” until now with the “multi-hit hypothesis”, both have attempted to explain the agents accused of creating and progressing NAFLD, and hepatic fat accumulation has been a mainstay as the “first hit.” Indeed, together, dietary free fatty acids, de novo liver lipogenesis, and non-esterified free fatty acids from adipose tissue which is mediated by the effect of insulin resistance are related to fatty liver development. Triglyceride accumulation in the liver by itself is a defensive mechanism against free fatty-induced lipotoxicity. Nevertheless, excess free fatty acid accumulation increases liver vulnerability to oxidative stress, which is a “second hit” that leads to lipid peroxidation, hepatocyte degeneration, apoptosis, necrosis, release of pro-inflammatory cytokines, activation of liver satellite cells and finally, fibrogenesis (11, 12). Considering the mentioned aspects, it is not unreasonable to assume that a large free fatty acid influx to the liver from rapid and massive weight loss induced by bariatric surgeries might harm the preexisting unhealthy liver and cause hepatic dysfunction alone or along with other impacts.
Severe protein deficiency secondary to malabsorptive bariatric procedures can lead to decreased apolipoprotein synthesis, which alleviates the hepatic antioxidative capacity and worsens steatosis (13). Although hepatic impairment has occurred after all types of restrictive, malabsorptive, and hybrid bariatric procedures (Table 1), malabsorptive bariatric procedures are the most abundant past surgical history in case-reports (Table 2).
Some authors revealed the role of the gut-liver axis in the pathogenesis and progression of NAFLD. Gut flora, named “gut microbiota,” plays a pivotal role in modulating the metabolic and immune functions of the digestive system. In the symbiosis relationship between gut microbiota and the host, it contributes to the energy homeostasis and guts barrier function. When the microbiota turns to dysbiosis, this can cause liver disease with respect to the unique condition of the liver as the first and major site for filtration of intestinal blood drainage. The systemic inflammation flare-up, special fibrogenic cytokine release, dietary choline metabolism change, endogenous ethanol production, impaired bile acid metabolism, and increased gut permeability are the intermediates in liver damage resulting from small intestine bacterial overgrowth (SIBO) (14-17).
Compared to healthy subjects, the prevalence of SIBO is higher in patients with morbid obesity and is associated with severe hepatic steatosis (18). In the context of post-bariatric interventions, the long length of the bypassed intestine in malabsorptive dominant procedures, dysmotility of the Roux limb, the impaired enterohepatic circuit of bile acids, the extended length of the bowel deprived from bile, gastric acid reduction in restrictive operations, and anastomosis stricture and subsequent partial bowel obstruction can potentially lead to SIBO and consequently liver damage.
4.2. Risk Factors
To prevent unwanted post-bariatric surgery events, it is crucial to individualize patient selection and management. We should be aware of high-risk patients. Obesity, type 2 diabetes, hyperlipidemia, hypertension, and metabolic syndrome are the well-known metabolic comorbidities associated with NAFLD (2) that should be considered in patients with underlying symptoms of high-risk patients.
4.2.1. The Severity of NAFLD
The severity of liver disease may affect bariatric surgery outcomes (19). As shown in Table 1, stunting and worsening reports of a second hepatic pathology are related to more advanced NAFLD rather than simple steatosis. Although some studies considered no role for NASH in increasing the risk of post-bariatric complication (20, 21), new data based on more participants show something to be different. According to the results of the post-RYGB outcomes, including 157,559 patients in the bariatric outcome longitudinal database (BOLD), liver disease is significantly associated with 30-day postoperative mortality (22). Additionally, it has been shown that NASH-diagnosed patients have a higher risk of liver-related mortality in the long-term follow-up (23). In the context of bariatric surgery, it is noted that histologically proven NASH is correlated with an increased risk of death within a median follow-up of 10.2 years (24). In a longitudinal study of patients with NAFLD, the fibrosis stage, but no other NASH features, was considered to associate independently with long-term overall mortality, liver transplantation, and liver-related events (25). Another study introduced steatosis accompanied by fibrosis, or so-called steatofibrosis, as the strongest predictor of mortality in NAFLD patients (26). Therefore, knowing the hepatic pathology may help in the decision-making process of high-risk patients.
Currently, some noninvasive modalities and models are available to assess liver fibrosis through serologic, radiologic, or genetic methods (27). Although they have progressed, they are expensive and their provided information is limited and inconclusive. Therefore, liver biopsy remains the gold standard to identify patients with most likely diagnosis of advanced disease (28). Brunt is the most widely used steatohepatitis classification system after staining liver biopsies, specimens with hematoxylin-eosin and Masson’s trichrome (29). Angulo et al. developed a NAFLD fibrosis score using age, hyperglycemia/diabetes, body mass index (BMI), platelet count, albumin, and AST/ALT ratio with two different cutoff values for predicting fibrosis to decrease the number of unnecessary biopsies (30). The NAFLD fibrosis score was evaluated in a population-based study among Chinese patients who were less susceptible to develop fibrosis. They suggested this score has a high negative predictive value; therefore, it can exclude patients who do not have advanced fibrosis and do not need a biopsy (31). The mean number of the NAFLD fibrosis scores was compared in morbidly obese patients who were operated with RYGB. As expected, RYGB relieved liver fibrosis and decreased the mean score after 12 months (32). Another study assessed the validity of this score in obese patients. A biopsy is taken routinely during the bariatric surgery but there is little chance to take a second biopsy. Therefore, they confirmed fibrosis by the biopsy and compared its prevalence using the NAFLD fibrosis scoring system cutoff. They suggested that the score has high negative predictive value (33); therefore, it can be used for evaluating patients after bariatric surgery or patients who develop liver failure without taking a biopsy (32). We recommend bariatric surgeons to use this score for their patients to evaluate the risk of fibrosis and liver function and estimate the outcomes of surgery or liver transplantation option.
Regarding available guidelines for perioperative nutritional, metabolic, and nonsurgical support of bariatric surgery patients, “consideration can be made for liver biopsy at the time of surgery to document steatohepatitis and/or cirrhosis that may otherwise be unknown due to normal appearance and/or liver function tests” (36). Thus, the preoperative selective liver biopsy of high-risk patients may be rational.
4.2.2. Revisional Bariatric Surgery
Revisional surgery is performed for two kinds of patients. The first is patients who fail to lose weight, which is prevalent in vertical banded gastroplasty (VBG), gastric banding (GB), and sleeve gastrectomy (SD); they should be converted to gastric bypass procedures for achieving sufficient weight loss (37). The risk of liver failure in these patients is not higher than what has been before (38). The second is patients who have malnutrition or severe persistent postoperative complications. In some cases, reversal surgery is inevitable (39). Surgeons who advocate revision rather than reverse are concerned with weight regain after reversing to the normal anatomy (40). Although approximately all of the obese patients have NAFLD and bariatric procedures help them to lose weight and improve their livers, weight regain after reversal surgery is not very much and the secondary weight will not reach the primary weight before the surgery; therefore, the risk of liver failure and NAFLD should be assessed per-case and interpreted according to their factors. Not performing a timely reversal surgery for those whose liver is likely fail, is very risky and puts the patients in high mortality (37-42).
In total, nine out of 59 post-bariatric surgery hepatic failure cases had a history of revisional bariatric surgery (five cases of VBG, three of GB, and one of SG). Two of them underwent BPD (43) and others were converted to (distal or mini) gastric bypasses (38, 44, 45). Considering approximately 10% - 25% as the rate of revisional bariatric surgeries (46), this point may be categorized as insignificant. Notwithstanding, it is rational to pay attention to the hepatic condition in revisional surgeries, especially based on the conclusion that this small population is not definitive. Although not specified on the hepatic consequences, a systematic review of 175 articles concluded a higher complication rate after reoperative bariatric surgery compared to initial surgery (47).
4.2.3. Gender
About 90% of the hepatic failure cases are females. According to the World Health Organization, in 2016, 15% of women and almost 11% of men over 18 years old were considered obese (48). Therefore, it is conceivable that the number of women undergoing bariatric surgery is also higher. The female-to-male distribution of 80% to 20%, respectively, for every year between 1998 until 2010 was reported in patients undergoing bariatric surgery (49). Thus, given the larger population, a higher number of post-bariatric complications such as hepatic failure seem to be expected. Moreover, NAFLD tended to be more frequent in men (50). This can be attributed to the defensive role of female sex hormones against hepatic fibrogenesis, which are capable of coordinating fatty acids into ketone body production instead of low-density lipoproteins or triglycerides. This impact was confirmed with more serious steatosis and fibrosing NASH in postmenopausal women (50, 51). In premenopausal women, the NAFLD prevalence and severity is higher than in those with polycystic ovary syndrome (PCOS), independent of obesity. Some researchers proposed the role of the hepato-ovarian axis to explain this non-incidental accompaniment. They suggested that advanced forms of NAFLD might augment hepatic and systemic insulin resistance and release mediators that can lead to PCOS (52). Without counting the role of bariatric surgeries and based on this hypothesis, we can suppose more advanced forms of NAFLD in some of our female cases. However, PCOS is not mentioned in any of our cases, and we do not know the perioperative NAFLD grading of most of the patients.
4.2.4. BMI Reduction Rate
The rate of BMI reduction in some cases and reports of hepatic failure is calculable; it spans from 0.23 to 14.65 kg/m2/month. The ideal weight loss rate after bariatric surgeries remains undefined. Andersen et al. observed that rapid weight loss of more than 1.6 kg/week could lead to portal inflammation and a change in fibrosis. This effect is more obvious in patients with morbid obesity at higher degrees (53).
4.2.5. Obesity, Gut Hormones, Cytokines, and Inflammation
It has been revealed that obesity has an inflammatory component in our body due to increasing atherosclerosis progression. Bariatric surgery can decrease C-reactive protein as an acute phase reactant marker and interleukin-6 (IL-6) as an inflammatory cytokine. Both of them indicate obesity can develop a gradual chronic inflammation in our body. IL-6 can increase insulin resistance and increase the chance of type 2 diabetes mellitus. Visceral fat tissues can secret IL-6 into the portal circulation, which is one of the involving markers in making metabolic symptoms of obesity. Moreover, inflammatory processes have been involved in NAFLD and NASH pathogenesis. All experiments have shown that NASH is related to obesity and body inflammation due to increased pro-inflammatory cytokines, tumor necrosis factor-alpha (TNF-α). TNF increases insulin resistance and its expression increases in liver and adipose tissue of obese patients. As a result, the bariatric procedure has a great role in decreasing the inflammatory status of obese patients and relieving NAFLD-NASH in their livers. In contrast to a previous hypothesis about the functions of adipose tissue, recent research suggests that this tissue can be a part of endocrine system due to producing adipokines and a part of the innate immune system due to producing adipocytokines. Leptin, which is expressed in adipose tissue, has a positive correlation with BMI. Leptin has a major role in controlling eating behavior and balancing energy. A negative correlation with BMI can be seen in the adiponectin level, which is one of the adipose-tissue specific proteins. Low-level of adiponectin has been related to decreased insulin sensitivity, increased inflammatory processes, and atherogenic effects. Inflammatory cytokines like TNF-α and IL-6 can suppress its production. Weight loss and fat removal processes can increase the adiponectin level in serum (54-56).
Our body has a neuroendocrine system for energy homeostasis and eating behavior control, which is working abnormally in obese patients. Various markers and body organs run this system. Ghrelin regulates appetite and energy expenditure. Its producing cells are in gastric fundus; therefore, different bariatric procedures alter its level variously. An interesting correlation has been found between ghrelin and its co-products, and NAFLD. They were higher in patients with NASH and positively correlated with liver fibrosis. It has been shown that the ghrelin level has a relationship with liver enzyme tests. Ghrelin agonists, which can be used in obese patients or any patient who has NAFLD, are a potential site for liver drug category (57). Glucagon-like-peptide-1 and 2 are secreted from intestinal L-cells. GLP-1 decreases blood glucose and increases the insulin level (58, 59). GLP-2, which increases after bariatric procedures like RYGB and SG, can decrease intestinal apoptosis and therefore, cause Ileal cell hyperplasia. Its functions were evaluated through experiments showing that it can stabilize weight loss, decrease malabsorption, reduce diarrhea, and regulate some aspects of the satiety-appetite circle (59, 60). An experiment in rat showed that GLP-2 is a signal that depends on nutrients and involves in absorbing lipids through the intestine (61). Bariatric procedures increase the level of GLP-2 in serum but the available length of intestine for lipid absorption is less than normal. Other effects of GLP-2 are exerted more on weight loss than on making obesity like suppressing appetite. In an experimental model of BPD, serum concentrations of GLP-1, 2, and pancreatic peptide YY (PYY) were evaluated before and after the surgery between BPD and sham-operated groups. They revealed that all of the markers increased significantly postoperatively and BPD rats had a significantly lower appetite (59). PYY has multiple roles in regulating appetite, decreasing emptying stomach, modifying colonic motility, and decreasing insulin production postprandially. Obese patients have lower fasting and postprandial PYY concentration (56, 57, 59, 60, 62-65).
4.3. The Most Appropriate Operation for High-Risk Patients
Besides the recognition of high-risk patients, it is important to choose the most appropriate strategy to manage them. A recent review concluded that the ideal outcome of RYGB is achieved when the combined length of the biliopancreatic or alimentary limb is 100 - 200 cm (66). Compared to RYGB, mini-gastric bypass (MGB) shows predominance in terms of weight loss. The difference might be explained by a longer bypassed small bowel in MGB and therefore a more malabsorptive effect (67). Greater weight loss and transient deterioration in several liver parameters were observed in the MGB group compared to RYGB or SG groups (68, 69).
Lee et al. tailored the bypassed limb according to the body weight in MGB candidates. In three groups of patients with lower (< 40 kg/m2), moderate (40 - 50 kg/m2), and higher (> 50 kg/m2) BMI, they intraoperatively considered a 150-cm, 250-cm, and 350-cm bypassed limb, respectively. After two years, the weight loss trend and resolving of comorbidity were compatible with the length of the bypassed limb (70). A total number of 118 surgeons from 30 countries with the experience of 47,364 MGB procedures reported the highest rate of revision due to protein-calorie malnutrition (0.6%) with a > 250 cm biliopancreatic limb length and the lowest rate (0%) with a biliopancreatic limb length of 150 cm (71).
Hess et al. proposed a guideline to avoid hypoproteinemia or excess weight loss in BPD/DS submitted patients. According to this guideline, < 3% of revisions of distal RYGB were reported in 120 patients after 10 years. Based on the different lengths of the small bowel in patients, the surgeon selected 10%, 40%, and 50% of the total small bowel length for the common channel, alimentary limb, and biliopancreatic limb, respectively. Some variations existed according to the patient’s gender and height, and attention was paid to the size of the stomach and type of anastomosis considered in the suggested guideline (72).
Jan et al. in a systematic review of 11 studies on bariatric surgeries in patients with cirrhosis, confirmed that the rate of hepatic decompensation was higher after SG than after RYGB and GB, but it was more likely to be self-limiting after SG than after BPD or RYGB. They recommended SG and GB followed by RYGB as the procedure of choice for higher-risk patients with more advanced liver disease (73).
4.4. Recognition and Management
4.4.1. Liver Transplantation and Bariatric Surgery
According to the high prevalence of obesity worldwide and its increasing rate, NAFLD is the most common liver injury and it is becoming the leading indication for transplantation. Transplantation is hard in obese patients; however, in late-stage or prolonged NAFLD, fibrosis progresses in the liver and obese patients face liver failure due to prolonged obesity and cirrhosis. Transplantation can be risky for obese patients without decreasing any fat mass and liver function preoperatively. Therefore, it is better to perform bariatric surgeries for patients who have its indications in order to subside the NAFLD-NASH, prevent further progression to cirrhosis, and decrease the chances of needing liver transplant. In some situations, transplantation and bariatric surgery can be performed together (74, 75).
Besides what indicated before, sometimes after bariatric procedures, malnutrition may occur in methods with malabsorptive components or methods with highly restrictive parts. Malabsorption can cause hypoalbuminemia, which can lead to NASH by itself. NASH can make portal hypertension, thus decreasing intestinal absorption and making a vicious cycle (41, 42, 76). Revisional or reversal surgery can correct it and brake the circle. Two women expired due to not responding to protein supplement and intravenous albumin after MGB surgery. Their livers failed and needed transplant; however, according to bad timing decision, death was their fate (41, 42). There is another report of three patients who received Scopinaro's biliopancreatic diversion and developed hepatocellular failure after the surgery. One of them expired while waiting for transplantation (76). Malnutrition is rare but can be fatal after malabsorptive bariatric procedures; therefore, patients should be followed frequently to monitor their liver enzymes and use high-protein supplements. Liver transplantation for patients with acute liver failure and liver dysfunction can be very risky due to impaired coagulation profile and postoperative complications like extended intensive care unit, increased infection rate, and increased need for blood products transfusion (74-78).
The literature is sparse concerning sufficient data for evidence-based management of post-bariatric surgery of hepatic failure. In addition to careful history taking and physical examination, routine serologic laboratory data should be checked. Viral, autoimmune, and metabolic liver disease should be ruled out using related exams even through liver biopsy. A hydrogen breath test or other relevant tests should be requested when SIBO is suspected. Radiologic or endoscopic evaluation should be performed in cases suspicious of gastrointestinal obstruction. The choline level should be checked to distinguish total parenteral nutrition (TPN)-induced hepatopathy in cases that receive long-term intravenous protein for possible intestinal failure following bariatric surgery from other causes (79).
SIBO presents with symptoms such as bloating, flatulence, abdominal discomfort, diarrhea, abdominal pain, weight loss, and steatorrhea. It can be recognized using the quantitative culture of the small bowel aspirate, 14C-D-Xylose, hydrogen breath tests, or urinary or serum tests. SIBO is mirrored in lab tests with mostly macrocytic or microcytic anemia (due to vitamin B12 deficiency, occult bleeding from ulcers of inactive excluded intestinal loops and trace element deficiencies), lymphopenia, low serum pre-albumin and transferrin, fat-soluble vitamin deficiencies, and elevated levels of serum folate and vitamin K. The strategy of SIBO treatment is based on the elimination of the underlying cause, eradication of overgrowth, and correction of nutritional deficiencies (80, 81).
Overall, 7 to 10-day intraluminal metronidazole is the first-line antibiotic therapy for SIBO that may need to be repeated. Other antibiotic treatment options include amoxicillin with clavulanic acid, trimethoprim-sulfamethoxazole (cotrimoxazole) with metronidazole, metronidazole with cephalexin, ciprofloxacin, and norfloxacin (80). Rifaximin is another antibiotic introduced for SIBO eradication (82). Probiotics are also highly effective (83).
Accepting the causative role of gastric achlorhydria in SIBO appearance (84-86) and considering the result of research that propounds microbiota alteration even with the normal breath test in gastric acid-suppressed patients (85), proton pump inhibitor (PPI) cessation or scale down should be postulated as a SIBO medical therapy. However, in another study on a group of 1190 patients using the breath test, the SIBO prevalence was not statistically different between PPI receivers and non-receivers (87).
Protein-calorie malnutrition could occur after malabsorptive dominant bariatric procedures characterized by hypoalbuminemia, edema, anemia, and asthenia. Nutritional assistance should begin to manage the nutritional deficiency induced by bariatric surgery by itself or through SIBO. Considering the privilege of enteral feedings, such as a physiologic and immune enhancing route, and on the other hand, the potency of total parenteral nutrition to induce or aggravate steatosis and catheter-related complications, enteral feeding is preferred to correct a nutritional deficit. Pancreatic enzymes should be added to increase protein absorption. The recommendation and restriction of feeding in liver failure should be considered. We may encounter thiamine deficiency as a potential outcome of SIBO, protein-calorie malnutrition, or during prolonged TPN in the course of malnutrition treatment, especially after intractable vomiting. Thus, it seems reasonable to consider intravenous thiamine before glucose-containing solutions (88, 89). Other vitamins and micronutrients should also be given according to each situation. A nasogastric tube should be inserted to avoid aspiration in the decreased consciousness condition. Nevertheless, the advantages of this policy do not cover the bypassed stomach and biliopancreatic limb; so, some authors advocate for gastrostomy tube insertion in the bypassed partition of the stomach to benefit from early digestion in the upper bowel, SIBO reversal in the excluded digestive path, and immune-enhancing capacity (90).
However, in patients who cannot benefit from enteral feeding due to low compliance or technical difficulties, TPN with vitamins and trace elements should be initiated.
Some patients recover with this nutritional assistance; but for others for which the medical management of protein-calorie malnutrition fails, surgery should be undertaken to shorten or eliminate the bypassed bowel. The optimal time for this intervention is unclear and therefore, quick action will end weight regain and its complications and late intervention exposes the patient to advanced liver failure that may lead to death. The evaluation of patients for a possible need for liver transplantation is rational before surgical intervention, especially in patients with encephalopathy attack.
Sometimes, performing a corrective surgery in the presence of coagulopathy and imposing anesthesia risk on the diseased liver in malnourished patients may be harmful. Therefore, less invasive procedures are preferred in these situations. Langdon et al. created a hole between two partitions of the stomach using the polypectomy snare tip of the endoscope and dilated it to 15 mm with a balloon to alleviate the malabsorptive effect of BPD (43). Motamedi et al. performed gastrogastrostomy surgically in two cases of MGB (42, 91). Baltasar suggested 60 cm (in length) side-to-side anastomosis from the so-called “kissing–X” of the alimentary limb to the biliopancreatic loop and 100 cm proximal to the Roux-en-Y anastomosis for lengthening of the common channel that can be done laparoscopically in the case of BPD/DS (92, 93).
If the patient becomes eligible for liver transplantation, most surgeons prefer to reverse bariatric surgery or lengthen the common channel simultaneously or as soon as the general condition allows. This decision is case-dependent; two cases that bypassed the bowel were not restored at the time of liver transplantation, and one needed retransplantation and reversion two months later (94), whereas reversion was not needed for another case (76). In addition, the gastrostomy tube that was inserted in the bypassed stomach prudently at the time of liver transplantation never needed to be used in another case (90).
Bariatric surgeons should be cautious that hepatic transplantation and bariatric surgery reversal might not be the end of hepatic surveillance in such patients because hepatic pathology may return more intensely. However, two intended cases were reversed two months after liver transplantation (77, 94, 95).
Collectively, this paper enhances our knowledge regarding the possibility of hepatic failure occurrence following bariatric surgeries through sequential liver biopsies and reviews the case reports and series about this event. The current study proposes to identify susceptible patients by considering risk factors for severe NAFLD, conduct more intensive preoperative evaluation of the liver condition (especially through liver biopsy as the gold standard of NAFLD recognition), select the appropriate operation type, conduct careful follow-ups, and pay attention to suspected signs and symptoms and available managements for this condition. However, this study is limited because the suggestions are based on just a few case reports. Inaccessibility to some reports and non-English literature is another limitation. It seems that the first step in expanding our knowledge regarding this entity is the special attention of bariatric surgeons and a report of the events. Therefore, further research can be undertaken to solve this enigma.
5. Conclusions
Hepatic failure can occur after bariatric surgeries although rarely. Given the paucity of data available and a lack of strong evidence, determination of risk factors, causes, and proper managements are needed. None of the bariatric surgeries is free of risks; however, the risk seems to occur more often after malabsorptive dominant procedures; thus, it is reasonable to avoid these surgeries in high-risk patients. Moreover, microbiota alteration alongside rapid and dramatic weight loss is suspected as another possible cause. Bariatric surgeons should be aware of this catastrophic event, recognize high-risk patients, use the best technique, and follow the patients carefully including a prompt diagnosis and performance of the appropriate action.