3.1. Epidemiology
Chronic hepatitis B (CHB) infection, which embraces a large spectrum of the disease, remains a serious public health problem in the world with 257 million people affected and causes around 900000 deaths annually. The African and Western-Pacific World Health Organization’s regions harbor 68% of all infections globally (
8). Most of the countries of these regions are of higher-intermediate endemicity (prevalence 5% - 7.9%) or highly endemic (prevalence ≥ 8%) for HBV. The European region is of lower-intermediate endemicity (prevalence 2% - 4.9%) and most of the countries in the American region have low endemicity (prevalence < 2%) (
9). Globally, it is estimated that HEV affects a total of 20 million cases, 70000 deaths, and 3000 stillbirths (
10). The HEV is endemic in many countries of Asia, Central America, and Africa. South-East Asia harbors most of the infections (60%) and deaths (65%) (
11). Epidemiology of HEV infection varies greatly in each geographical location with a well-characterized viral genotype distribution.
While there is a decline in the prevalence of HBV in many regions, the prevalence of HEV is generally increasing in most countries. A recent systemic review of the studies published between 1965 and 2013 by Schweitzer et al. (
8), has shown a clear decline in HBV infection rates in many countries in several WHO regions, including Western-Pacific, South-East Asia, America, Europe and Eastern-Mediterranean during the periods 1965 - 1989 and 1990 - 2013. However, this trend was not seen in the African region throughout the periods considered. Another national-wide review from China, which is one of the most affected countries by HBV infection, also showed a significant decrease of HBV seroprevalence in the general population from 7.1% in 2006 to 6.1% in 2013 (
12). This pattern clearly deciphers an expanded adaptation of universal HBV immunization recommendations in newborns from the year 1992 in several countries (
13), with a poor response by many African countries due to economic constraints. These data are summarized in
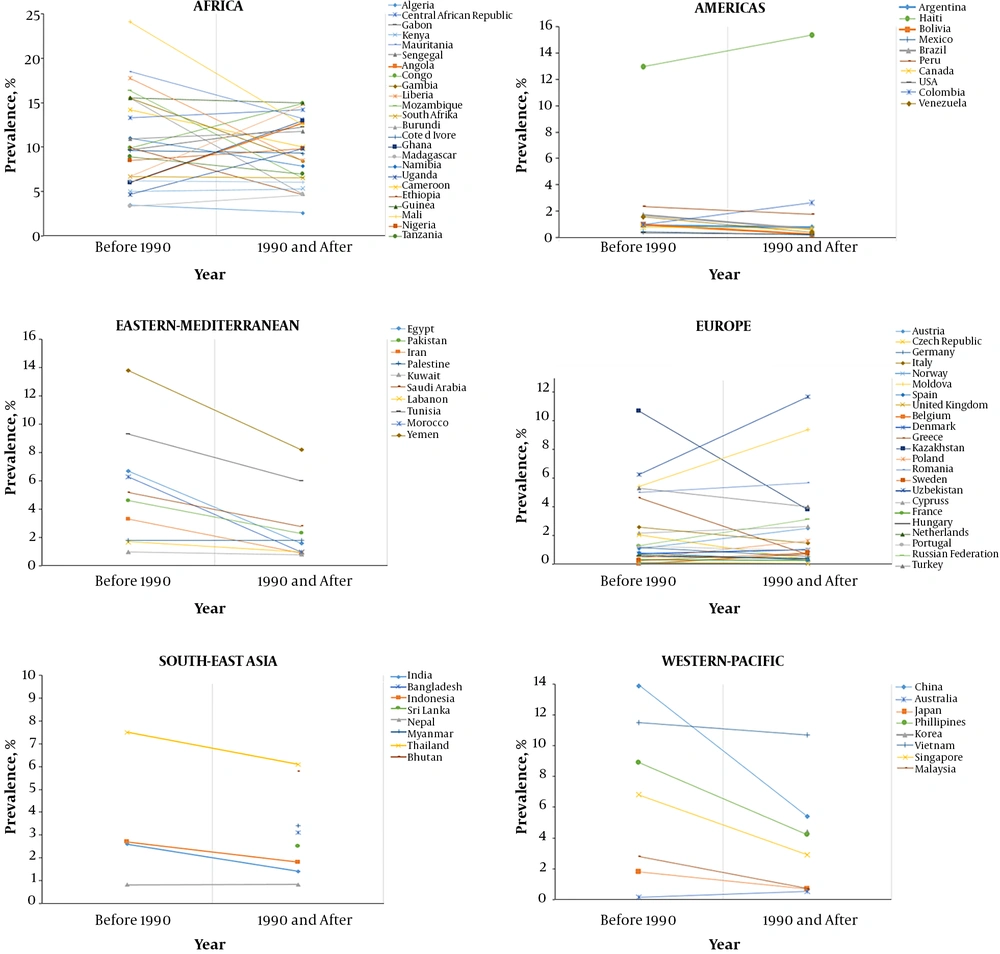
Figure 1.
HBV seroprevalence in different countries in two-time periods (before and after the year 1990) (9). Graphs show the studies describing HBV seroprevalence (HBsAg) in countries from six WHO regions. The correlation was shown between studies that were published before the year 1990 and those published in 1990 and afterward. Apart from Africa and few countries in Europe and Americas, general declination of HBV seroprevalence is evident between two time periods.
On the other hand, gathered evidence suggests that the rate of HEV infection is clearly escalating in many countries (
14-
17). In Europe for instance, the number of autochronous symptomatic cases has steadily increased from 124 cases in 2003 to 1243 cases in 2016 in England (
18) and 31 cases in 2001 to 1991 cases in 2016 in Germany (
19). A large multicenter study in Scotland has recently reported an increase in HEV detection among blood donors from 4.5% in 2004 - 2005 to 9.3% in 2014 - 2015 (
20). Furthermore, in China, it was reported in the Third National Viral Hepatitis Prevalence Survey of 2005 - 2006 (
21) that HEV seroprevalence rate was 23.5% in the country, which was higher than the previously reported seroprevalence of 17.2% in 1997 (
22). These data are illustrated in
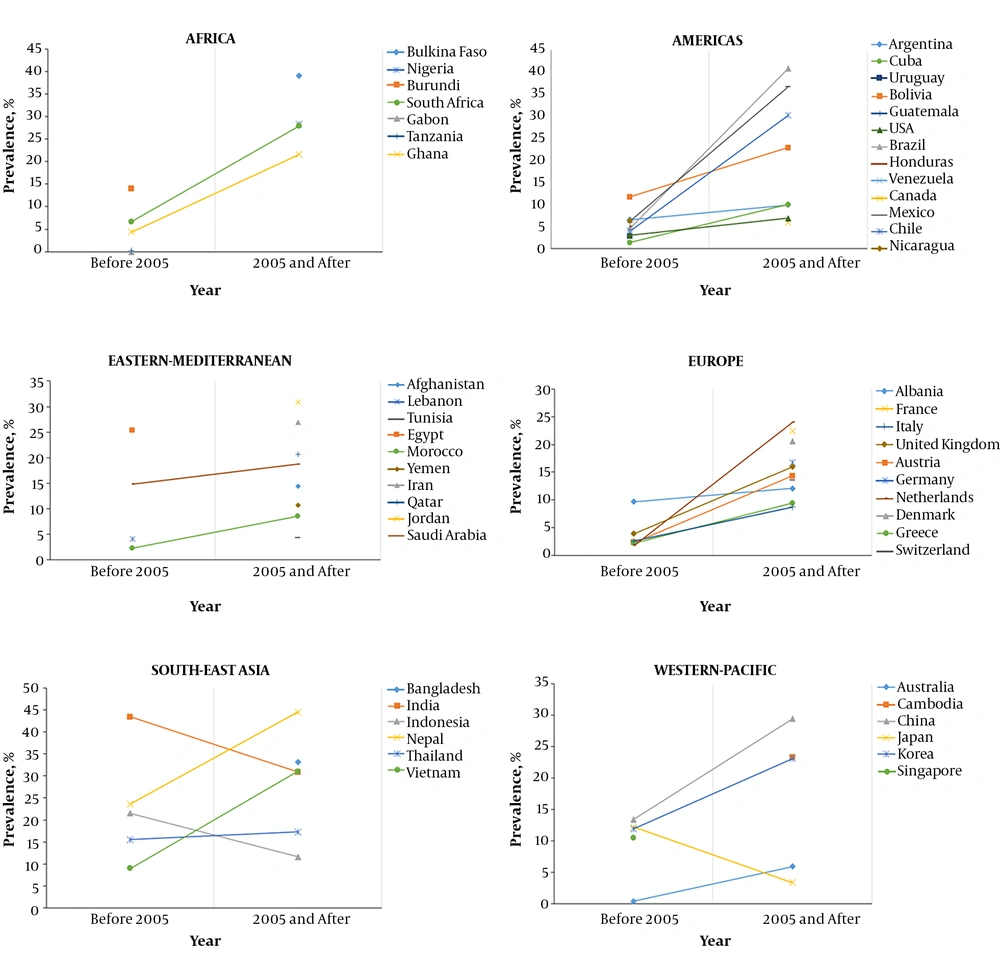
Figure 2 and Appendix 1 in Supplementary File. The increasing detection rate of HEV might be a result of increased awareness and hence increased testing for the virus and/or a true increase in numbers of new infections. Moreover, the new serological tests, particularly rapid Wantai, have shown to have a significantly higher sensitivity than most of the older tests (
23) and hence, it might possibly contribute to the variability.
HEV seroprevalence in different countries in two-time periods (before and after year 2005). Graphs showing the studies describing HEV seroprevalence (anti-HEV IgG) in countries from six WHO regions. The correlation was shown between studies that were published before the year 2005 and those published in 2005 and afterward. A clear inclination of HEV seroprevalence was observed between two time periods in most countries.
Several meta-analysis studies have revealed that older subjects and males are more susceptible to HEV infection than younger people and females, respectively (
14,
24-
26). In most countries where HEV is prevalent, infection in children is relatively rare. However, in Egypt where the prevalence of HEV in the general population is among the highest in the world, a very high rate (56.7%) of HEV infection was seen in children among patients with acute viral hepatitis (
27). These observations were at least partly attributed to possible parenteral transmission as up to 20% of these children pre-received blood transfusion (BT) that was not screened for HEV. Indeed, BT has previously correlated with the possibility of HEV transmission (
28). Late presentation of HEV infection in life might be due to cumulative viral exposure in a lifetime and probably aging of the immune system. However, the reasons as to why males are more susceptible than females are yet to be clarified.
HBV affects different age groups depending on the time of infection. In countries with a high prevalence of HBV (prevalence > 2%), infection is usually acquired perinatally or during early childhood and the majority of the patients live with the infection to its chronic phase, while in countries with low prevalence (prevalence < 2%), HBV infection is usually acquired during adulthood, mostly through intravenous drug uses (IDU) or sexual intercourse (
29). To date, there is no evidence of any sex predilection of HBV infection, but male sex is known to be an independent risk factor for increased progression to cirrhosis and HCC (
30).
In endemic areas, HEV typically superimposes CHB in most cases since the former occurs during adulthood, while the latter occurs in early life in these areas. Thus older and male subjects comprise a higher risk group for HBV-HEV superinfection (
31-
33). The rates of HBV-HEV superinfection vary considerably. The prevalence of 9% - 38% has been reported in previous studies in China (
34-
38). In Southeast Asia, the HBV-HEV superinfection rate was reported to be 45%, 14% and 9.5% in Vietnam, India and Bangladesh, respectively (
39-
41). A clear territory-wise distribution of HBV-HEV superinfection has been observed in Africa where a higher prevalence of 56.7% was reported in Egypt in the northern part of the continent, while insignificant rates of 1% and 0% were reported in Kenya, the Eastern part of Africa (
27,
42,
43) (
Table 1).
| Country | Reference | Cohort | Prevalence (%) | Sample Size | Year of Sampling |
|---|
| HEV Diagnostic Method | Agea |
|---|
| IgG | IgM | RNA | |
|---|
| China | (44) | Pregnant women | 10.7 | 0 | | 26.4 ± 4.1 | 391 | 2016 |
| Turkey | (45) | Positive HBV DNA | 13.7 | 8.4 | 14.7 | 42.2 ± 9.1 | 190 | 2004 |
| China | (37) | General | 38.1 | 1.57 | | 52.1 ± 13.1 | 1022 | 2014 |
| India | (46) | Cirrhotics | | | 6.3 | 46 ± 14.6 | 192 | 2004 |
| Vietnam | (39) | CHB | 41 | 9 | | 41 (9 - 84) | 744 | 2013 |
| Hongkong | (32) | Acute HEV | 19 | | | 51 (22 - 85) | 161 | 2000 - 2012 |
| Spain | (47) | HIV-infected | 13.3 | 0 | 0 | 46.3 (21 - 80) | 448 | 2013 |
| Nepal | (48) | HIV-infected | | 14.3 | | 36.2 ± 10.3 | 459 | 2015 |
| Italy | (49) | HIV-infected | 5.9 | | | 41.7 ± 6.7 | 34 | 2013 |
| Kenya | (42) | Acute hepatitis | 1 | | | 38.9 (19.83) | 100 | 2012 |
| Bangladesh | (50) | Pregnant with acute viral hepatitis | | 9.4 | | 32 (8 - 82) | 31 | 2004 - 2006 |
| Ghana | (51) | Jaundiced | 18.7 | | | NS | 155 | 2016 |
| Central African Republic | (52) | Fever and jaundice | | | 4.9 | NS | 198 | 2008 - 2010 |
| India | (40) | Children with acute viral hepatitis | | 0.7 | | < 16 | 149 | 1998 - 2002 |
| Egypt | (27) | Children with sporadic acute viral hepatitis | 56.7 | | | NS | 162 | 2008 |
Abbreviations: IQR, interquartile range; NS, not specified; SD, standard deviation.
aValues are expressed as mean ± SD or mean (IQR).