1. Introduction
2. Case Presentation
2.1. Clinical Summary
2.1.1. Case 1
The patient was a 43-year-old male who used to be physically healthy. On August 12, 2015, he underwent mediastinal tumor resection in our hospital. The postoperative pathological diagnosis was “thymoma, type B2”, and the patient received chemotherapy with cyclophosphamide plus cisplatin. On September 18, 2016, the contrast-enhanced chest computed tomography (CT) scan revealed a recurrence of thymoma with left pleural metastasis. On September 23, 2016, a mediastinal mass resection was performed on him, after which he received chemotherapy with docetaxel plus lobaplatin. In July 2018, the patient was admitted due to limb weakness. On admission, serum levels of creatine kinase (CK) (6370 U/L), Creatine Kinase-MB Isoenzyme (CK-MB) (97.7 U/L), and troponin T (571.8 pg/mL) increased significantly. The left ventricular ejection fraction (LVEF) was 73%, estimated by echocardiography on July 13, 2018. Moreover, biomarkers of vasculitis such as ANCA-nuclear, and antinuclear antibody (ANA) were positive. Electromyography (EMG) suggested partial myogenic damage. The patient was diagnosed with polymyositis (PM), and the symptom was alleviated after treatment with glucocorticoid. Meanwhile, the chest CT indicated the thymoma metastasis to the left thoracic cavity and, therefore, the surgery was performed. Later, multiple CT scans performed in 2019 showed that the thymoma was in progress. After the routine examination such as the electrocardiogram (ECG) showing substantially normal, therefore, the patients received chemotherapy with pembrolizumab (200 mg) plus paclitaxel (300 mg) on June 24, 2019.
On July 2, 2019, symptoms of chest tightness, shortness of breath, and limb weakness showed up. The myocardial enzyme was elevated, and chest CT scan showed reduced pleural metastasis lesion. The symptoms did not alleviate after the patient was given glucocorticoid (methylprednisolone) and diuretics. On July 6, 2019, moreover, tracheal intubation and ventilator-assisted ventilation were performed due to dyspnea and respiratory failure. The patient was transferred to our hospital for intensive care on July 7, 2019.
Physical examination (PE) on admission: Temperature (T): 36.5°C, pulse (P): 84 beats per minute (bpm), respiratory rate (R): 20 times per min, blood pressure (BP): 122/80 mm Hg, and weight: 70 kg. The patient was conscious, and there was no rash on the skin or mucosa. The lungs were clear with no pleural rub, and his heart rate (HR) was 80 bpm with sinus rhythm. No pathological murmur or pericardial friction rubs were detected through auscultation, and his limbs were not swollen.
The clinical characteristics are summarized in Table 1.
| No. | Case 1 | Case 2 | Case 3 |
|---|---|---|---|
| Age (y) | 43 | 27 | 52 |
| Gender | Male | Female | Male |
| Previous history | None | None | Kidney stone |
| Histological type | Type B2 | Type B2 | Type B1 or type AB |
| Therapeutic effect | Pleural metastases are slightly smaller | Not reviewed | Not reviewed |
| Duration between hospital admission and onset (days) | 8 | 12 | 15 |
| Symptoms | |||
| Limb weakness | + | + | + |
| Dyspnea | + | + | + |
| Ptosis | + | + | + |
| Laboratory tests | |||
| Index CK (U/L) | 5806 | 10988 | 7633 |
| Index CK-MB (U/L) | 193.5 | 293.7 | 159.5 |
| Index CK-MB/CK | 3.33% | 1.76% | 2.09% |
| Peak CK (U/L) | 7273 | 25862 | 7633 |
| Peak CK-MB (U/L) | 192.5 | 359.6 | 433.4 |
| Peak hsTNT (pg/ML) | > 10000 | 6558.0 | > 10000 |
| Peak NT-proBNP (pg/mL) | 7140 | 4030.0 | 5696 |
| MuSK.Ab | - | Not reviewed | - |
| RyR.Ab | + | Not reviewed | + |
| AchR.Ab | + | Not reviewed | + |
| Titin-Ab | - | Not reviewed | - |
| Auxiliary examination | |||
| Electrocardiogram | Atrioventricular dissociation, multi-source ventricular escape rhythm | Second-degree atrioventricular blocks: Mobitz type I, intraventricular aberrant conduction; refractory ventricular tachycardia after disease progression | Complete atrioventricular block |
| LVEF detected by echocardiogram | 50% | 41% | 21% |
| Biopsy | |||
| Myocardium | a | Not reviewed | Not reviewed |
| Skeleton muscle | Poor specimen sampling | Not reviewed | Not reviewed |
| irAEs | |||
| Myocarditis | + | + | + |
| Polymyostitis | Suspected | Suspected | Suspected |
| Myasthenia gravis | Suspected | Suspected | Suspected |
| Intervention | |||
| Anti-inflammatory agents | Methylprednisolone and intravenous immunoglobulin | Methylprednisolone | Methylprednisolone and intravenous immunoglobulin |
| Advanced life support | Mechanical ventilation; pacemaker | Mechanical ventilation; CRRT | Mechanical ventilation; pacemaker; CRRT; ECMO |
| Outcome | Disease progression and withdrawal of therapy | Death | Multiple organ dysfunction syndrome and withdrawal of therapy |
Abbreviations: CK, creatine kinase; CKMB, creatine kinase-MB; hsTNT, high-sensitive cardiac troponin T; NT-proBNP, N terminal pro B type natriuretic peptide; MuSK.Ab, anti-skeletal muscle receptor tyrosine kinase antibody; RyR.Ab, ryanodine receptor calcium release channel antibody; AchR.Ab, acetylcholine receptor antibody; Titin-Ab, connexin antibody/human actin antibody; LVEF, left ventricular ejection fraction; irAEs, immune-related adverse events; CRRT, continuous renal replacement therapy; ECMO, extra-corporeal membrane oxygenation.
a See Figure 1.
Auxiliary examinations:
(A) Positive acetylcholine receptor antibody (> 20 nmol/L, normal range < 0.45 nmol/L); positive ryanodine receptor calcium release channel receptor antibody (normal range: Negative); negative idiopathic inflammatory myopathy panel; negative paraneoplastic syndrome panel.
(B) Bedside Chest X-ray: Postoperative mediastinal tumor; proposed left lung and pleural metastasis; unexclusive left 6th rib metastasis; left pneumonia with mild pleural effusion on the left.
(C) Muscle biopsy of the right quadriceps: Fragmentary specimens, fibrous tissue, fat, and muscle tissue.
(D) Bedside doppler ultrasound showed diastolic dysfunction, mild mitral regurgitation, and mild tricuspid regurgitation. There was no obvious enlargement of the atria and ventricles or pericardial effusion. The segmental motion was weakened (poor anteroseptal and anterior wall contraction). Left ventricular ejection fraction was estimated to be about 50%, and the mitral inflow E/A ratio was < 1.
Diagnosis: 1, acute severe myocarditis, arrhythmia, heart failure, New York Heart Association (NYHA) cardiac function grade IV; 2, suspected acute coronary syndrome; 3, PM; 4, suspected myasthenia gravis (MG); 5, thymic malignant tumor (recurrence).
Intervention: Methylprednisolone 80 mg ivgtt qd; intravenous immune globulin (IVIG) 25 g/day; pyridostigmine bromide 60 mg ig q6h; piperacillin sodium and tazobactam sodium 4.5 g ivgtt q6h. Shortness of breath was slightly alleviated after treatment. On July 9, 2019, however, the patient suffered atrioventricular dissociation and ventricular escape beat with an HR of 35 - 50 bpm. Temporary pacemaker implantation was performed and coronary angiography showed no occlusion or slow flow (TIMI level 2) of the vessels. Within 24 hours, cardiac arrest, ventricular tachycardia (VT), and ventricular fibrillation showed up repeatedly, and all rescue measures proved ineffectual.
2.1.2. Case 2
The patient was a 27-year-old female, who used to be physically healthy and, apparently, had no history of smoking. On May 12, 2019, a contrast-enhanced chest CT scan revealed a thymic tumor in the anterior superior mediastinum with pleural metastasis. The pleural biopsy indicated thymoma (type B2, stage IV with pleural metastasis). On June 5, 2019, the patient received chemotherapy with albumin-bound paclitaxel (200 mg), carboplatin (740 mg), and pembrolizumab (200 mg). On June 17, 2019, the patient started to suffer from limb weakness, shortness of breath, right eyelid ptosis, and stress urinary incontinence. The symptoms gradually worsened, and the patient was transferred to the intensive care unit on June 21, 2019.
PE: T: 36.2°C, P: 150 bpm, R: 20 times per min, BP: 124/89 mm Hg, weight: 56 kg. The patient was conscious and there was no rash on the skin or mucosa. The lungs were clear with no rales or pleural rub, and the HR was 134 bpm with sinus rhythm. No pathological murmur or pericardial friction rubs were detected through auscultation, and her limbs were not swollen.
Bedside echocardiogram: The heart rhythm was irregular, with mild to moderate pericardial effusion (left inferior ventricular wall 0.6 mm, left atrial free wall 0.4 mm, right inferior atrial free wall 12 mm, right ventricular free wall 7 mm). The estimated LVEF was 45%, inferior vena cava was 1.45 cm, and respiratory variation was < 50%.
Diagnosis: 1, acute severe myocarditis, arrhythmia, heart failure, NYHA cardiac function grade IV; 2, suspected MG; 3, suspected myositis; 4, thymoma (type B2, stage IV with pleural metastasis).
Intervention: The patient was treated with methylprednisolone 80 mg ivgtt qd. The onset of VT (electrocardiogram: VT, HR: 163 bpm, QRS: 86 ms, QT/QTc: 292/493 ms, QRS axis: +97°, RV5/SV1: 0.38/0.14 mV) started on the day of admission, accompanied by the decreasing BP. Cardioversion helped to convert sinus rhythm (electrocardiogram: Sinus tachycardia, burst polymorphic VT; suspected second-degree atrioventricular block type 1; intraventricular block; abnormal Q wave in anterior and high lateral walls; QRS complex low voltage; ST-T changes; HR: 110 bpm, QRS: 106 ms, QT/QTc: 324/438 ms, QRS axis: +70°, RV5/SV1: 0.49/0.00 mV, P: 5000 ms). Continuous renal replacement therapy (CRRT) was initiated the same day due to anuria. However, the patient suffered from frequent premature ventricular contraction and recurrent VT (electrocardiogram: VT, HR: 117 bpm, QRS 176 ms, QT/QTc: 432/605 ms, QRS axis: +39°, RV5/SV1: 1.04/0.00 mV, P: 5000 ms). Lidocaine was used as antiarrhythmics and electrolyte disturbance was remedied. The dose of methylprednisolone was increased to 120 mg ivgtt qd. Unfortunately, the patient developed a pulseless VT (electrocardiogram: VT, HR: 111 bpm, QRS: 130 ms, QT/QTc: 436/603 ms, QRS axis: -45°, RV5/SV1: 0.82/0.00 mV, P: 5000 ms) on June 23, 2019, and cardiac arrest occurred after cardioversion. All rescue measures proved ineffectual and no return of spontaneous cardiac rhythm occurred.
2.1.3. Case 3
The patient was a 52-year-old male, with a medical history of kidney stones and smoking for more than 30 years. In December 2017, a mass in the lung was found through an annual health check. On March 27, 2018, the CT scan found a right lung neoplasm with pleural metastasis. The lung biopsy showed a thymoma (suspected type B1 or AB). On April 19, 2018, the therapy of albumin-bound Paclitaxel (200 mg), carboplatin (740 mg), and pembrolizumab (200 mg) were administered. On May 3, 2018, the patient developed fever, soreness, limb weakness, shortness of breath, and left eyelid ptosis. The patient was admitted to the community hospital on May 8, 2018. Tests on myocardial enzymes showed elevated CK (25612 U/L) and CK-MB (969 U/L), positive acetylcholine receptor antibody (5.50 nmol/L, normal range < 0.45 nmol/L), positive ryanodine receptor calcium release channel receptor antibody (normal range: Negative), and positive connexin antibody (normal range: Negative). Chest CT scan showed a mass in the right lung, and pneumonia on both sides. Between May 8 and May 10, 2018, the patient was treated with methylprednisolone 40 mg/day intravenously, IVIG 35 g/day, and piperacillin-tazobactam. On May 10, 2018, the patient developed a complete atrioventricular block with the HR decreasing to 24 - 28 bpm. Emergent implantation of the temporary pacemaker was performed. Between May 11 and May 13, 2018, the dose of methylprednisolone was increased to 500 mg/day, combined with IVIG 35 g/day. The disease showed no improvement and on May 14, 2018, the patient was transferred to the intensive care unit in our hospital.
PE: T: 36.0°C, P: 130 bpm, R: 25 times per min, BP: 78/58 mm Hg. The patient was in a coma. There was no rash on the skin or mucosa. The pupils were equal and round with a diameter of 3 mm, but no light reflex was observed on either side. The lungs were rough with moist rales of both lower lungs, and no pleural rub was heard. The HR was 130 bpm with sinus rhythm. No pathological murmur or pericardial friction rubs were detected through auscultation, and his limbs were not swollen.
Auxiliary examinations:
(A) Electrocardiogram: Wide QRS complex tachycardia with fusion wave, left axis deviation, HR 152 bpm, QRS: 122 ms; QT/QTc: 332/527 ms, RR/PP: 394/377 ms.
(B) Bedside echocardiogram: The left ventricle was enlarged, the end-diastolic diameter was 52 mm, and the upper and lower diameter of the section of the apex was about 90 mm. The left ventricle wall movement was not coordinated, and the degree of contraction diffusively decreased obviously, especially in the middle and lower part of the ventricular wall. The estimated LVEF was 21%. The aortic valve was slightly enhanced with mild regurgitation; mild regurgitation of the mitral and tricuspid valve; there was no obvious fluid sonolucent area in the pericardial cavity. Conclusion: Left ventricular enlargement; left ventricular systolic function was significantly reduced; Mild mitral regurgitation; Mild tricuspid regurgitation.
Diagnosis: (1) acute severe myocarditis, arrhythmia, heart failure, NYHA cardiac function grade IV; (2) MG; (3) suspected myositis; (4) thymoma (type B1 or AB).
Intervention: The patient was treated with imipenem and cilastatin sodium 1 g ivgtt q8h, terlipressin, noradrenaline, dopamine, and dobutamine. Within 24 hours after admission, the ventricular escape rhythm emerged with an HR of approximately 30 bpm (electrocardiogram: Ventricular rhythm, non-specific intraventricular block; HR 40 bpm, QRS 144 ms, QT/QTc: 408/332 ms, RR/PP:1492/0 ms), accompanied by the loss of BP and oxygen saturation. After cardiopulmonary resuscitation, the patients returned to sinus rhythm but no spontaneous circulation occurred. Extra-corporeal membrane oxygenation was implemented. However, the heart function did not improve and was succeeded by multiple organ failure. The family withdrew treatment on May 15, 2018.
2.2. Pathological Findings
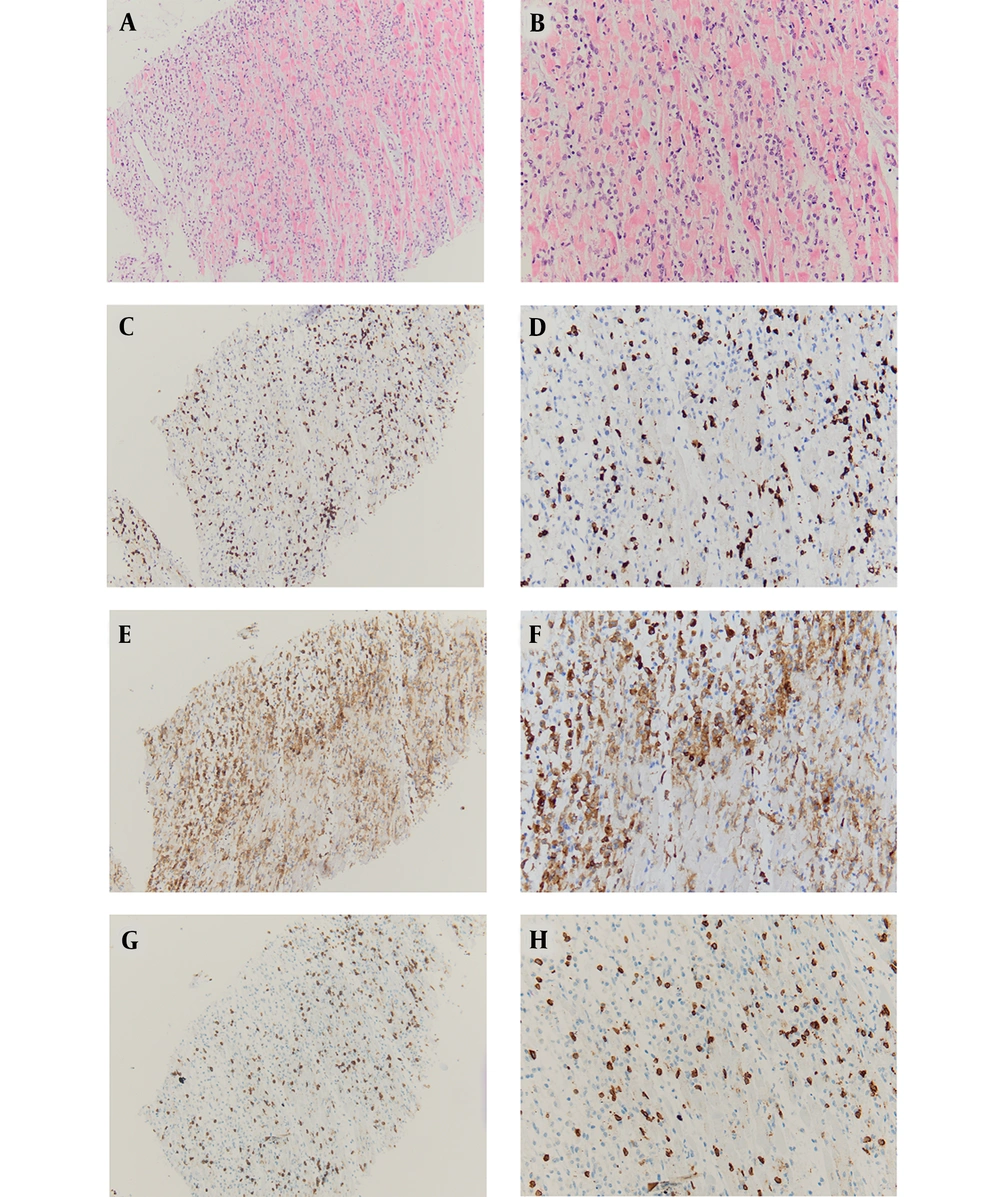
Myocardial biopsy of case 1 suggested inflammatory cell infiltration, myocardial cell degeneration, and non-ischemic necrosis. Myocardial histopathology was characterized by T lymphocyte infiltration (Figure 1).
A, HE-10X: Cardiomyocyte degeneration, necrosis, atrophy, interstitial lymphocyte infiltration, 10x; B, HE-20X: Cardiomyocyte degeneration, necrosis, atrophy, interstitial lymphocyte infiltration, 20x; C, CD3-10X: T lymphocyte marker, distributed among cardiomyocytes, 10x; D, CD3-20X: T lymphocyte marker, distributed among cardiomyocytes, 20x; E, CD4-10X: Helper T lymphocyte subtype marker, distributed among cardiomyocytes, 10x; F, CD4-20X: Helper T lymphocyte subtype marker, distributed among cardiomyocytes, 20x; G, CD8-10X: Cytotoxic T lymphocyte subtype marker, distributed among cardiomyocytes, 10x; H, CD8-20X: Cytotoxic T lymphocyte subtype marker, distributed among cardiomyocytes, 20x
3. Discussion
The application of ICIs has provided a new hope for tumor treatment. However, the side effects have remained unnoticed. Studies revealed that the incidence of myocarditis after immunotherapy was 0.06 - 1% (6). Immunotherapy-associated fulminant myocarditis was rare but progressed rapidly, resulting in high mortality and poor prognosis (1-3).
Recent phase II clinical trials showed a therapeutic effect of pembrolizumab in thymoma (4, 5). The chemotherapy regimen of pembrolizumab combined with albumin-bound Paclitaxel was promoted worldwide (4). However, the thymus participated in the maturation of T lymphocytes, and patients with thymoma were more likely to have autoimmune diseases such as MG, nephritic nephrotic syndrome, rheumatoid arthritis, and lupus erythematosus (4). Studies also indicated that the incidence of irAEs in patients with thymoma after immunotherapy was significantly higher (4, 5). Cases of immunotherapy-associated myocarditis were reported previously with favorable prognoses. However, there were no detailed case reports of myocarditis after immunotherapy in patients with thymoma.
Case 1 had a history of PM before receiving immunotherapy, suggesting that it was already autoimmune dysfunction before the patient received immunotherapy. Studies have found that patients with thymoma are more likely to develop autoimmune diseases after receiving immunotherapy, with PM and myocarditis being the most common complications (5). The pathological feature of PM is that the CD8+ T lymphocytes invade non-necrotic muscle fibers, where T lymphocyte-mediated immune responses play an important role (7). Myocardial histopathology of myocarditis caused by immunotherapy is also characterized by T lymphocyte infiltration, suggesting that there may be an intersection of autoimmune damage. A retrospective study, published in 2018, found that myositis and myocarditis were co-morbidities when referring to irAEs, implying similar or shared pathogenesis (3). In the present three cases, moreover, PM was diagnosed since CK was significantly elevated. Accordingly, for patients with previous autoimmune diseases, especially those with a history of PM, immunotherapy might have been risky due to the higher probability of developing immune-associated myocarditis. Most of the current clinical trials have excluded cases with previous autoimmune diseases, leading to the unclear safety endpoints of immunotherapy in those patients (5).
Programmed cell death protein 1 gene is mainly expressed on the surface of activated T cells. When it binds to PD-L1 on the surface of tumor cells, it down-regulates the activity of cytotoxic T cells, so that tumor cells avoid immune surveillance (4). Immune checkpoint inhibitors (e.g., pembrolizumab) enhance the immune surveillance and immune clearance of T cells by inhibiting the binding of PD-1 to PD-L1 (8). The mechanism of ICI-associated acute myocarditis is still unclear (9). In mice, PD-1 functions in the myocardial immune response and inhibits inflammatorily cardiomyocyte injury (10). On the contrary, dilated cardiomyopathy in PD-1 gene-deficient mice explains the effect of PD-1 on protecting cardiomyocytes due to the production of autoimmune antibodies against troponin I (TNI) (11, 12).
Previous studies found T lymphocytes- and/or macrophages-infiltrated myocardial tissue in patients with immunotherapy-associated myocarditis (1, 2), which was consistent with the result of myocardial biopsy in our study. It indicated similar pathogenic and pathological characteristics in thymoma, compared with other tumors.
At present, glucocorticoid pulse therapy is the main treatment of ICI-associated myocarditis (3). Mahmood et al. summarized 35 cases of immunotherapy-associated myocarditis and found that high-dose glucocorticoid pulse therapy reduced the incidence of major adverse cardiac events (13). In addition, the IVIG can block autoimmune antibodies and inhibit autoimmune reactions, which has been widely used in treating viral myocarditis and rheumatic immune diseases. However, cases 1 and 3 in our study did not recover after glucocorticoid plus IVIG therapy. Mahmood et al. also reported two cases treated with IVIG with one death and one survival (13). Salem et al. reported a successful case of fulminant myocarditis after nivolumab (a PD-1 inhibitor) with Abatacept (a CTLA-4 agonist) in a patient with terminal lung cancer (6). Esfahani et al. reported a case of melanoma complicating fulminant myocarditis after immunotherapy, who was treated with alemtuzumab (14). Abatacept is mainly recommended for rheumatoid arthritis and Alemtuzumab for chronic lymphocytic leukemia and multiple sclerosis. There was no evidence of their safety and effectiveness. Frigeri et al. reported a case of early use of extra-corporeal membrane oxygenation (ECMO) and IABP on fulminant myocarditis after immunotherapy (15). In our study, case 3 received ECMO and IABP only after repeated cardiac arrest and cardiopulmonary resuscitation. However, it was recommended that further studies should be carried out to determine whether early use of heart-assist devices such as IABP, Impella, or ECMO helped in the treatment.