1. Background
The global outbreak of SARS-CoV-2 viral infection (COVID-19 pandemic) had devastating health, social, and economic effects (1-3). The first case was reported on 8 December 2019 in Wuhan City, Hubei Province, China. On 11 March 2020, it was considered a pandemic by the World Health Organization (WHO).
With the increasing spread all over the world, a worse fate awaited people with various underlying diseases. Two-thirds of patients with other comorbid conditions needed intensive care unit (ICU) admission or died (4). Meanwhile, patients with cardiovascular disease (CVD) have a worse prognosis (5). It appears that there is sophisticated interaction between COVID-19 and cardiovascular system. In fact, cardiovascular risk factors (such as diabetes mellitus, systemic hypertension, and CVD) were preexisting findings in patients with COVID-19 infection (6-9). In hospitalized patients, lines of evidence of myocardial injury have been reported in 20 - 40% of cases (6, 10). In addition, acute coronary syndrome, venous thromboembolism, and arrhythmias may occur in victims (11). Regarding the prognosis of these patients, myocardial damage is associated with a remarkably worse prognosis (12). Proper and timely assessment of the viral-induced heart damage by biomarkers of high-sensitivity cardiac troponin I (cTnI) and NT-proBNP is mandatory for early identification of this complication, which may predict the prognosis of affected patients (5).
2. Objectives
The purpose of this study was to describe the prognostic comparison of SARS-CoV-2 viral infection in patients with and without underlying heart disease in our center.
3. Methods
This descriptive-analytical study was conducted from April to June 2020 at Imam Khomeini Hospital of Tehran University of Medical Science with the ethical code of IR.TUMS.VCR.REC.1399.355. In this study, 549 patients hospitalized with the initial diagnosis of SARS-CoV-2 were examined. After obtaining written consent, the patients with relevant symptoms, lung computed tomography (CT) scan, and positive polymerase chain reaction (PCR) test consistent with SARS-CoV-2 infection were enrolled in the study. Patients who did not consent to participate in the study or were discharged during hospitalization with personal consent were excluded. Finally, 400 diagnosed patients with SARS-CoV-2 infection were enrolled in the study and divided into 4 groups: (1) without any underlying disease (n = 100), (2) with a history of CVD alone without other underlying diseases (n = 100), (3) a history of underlying diseases, except CVD (n = 100), (4) with both previous CVD and other underlying non-CVDs (n = 100).
The checklist (including demographic and anthropometric information of patients) was completed. Patient prognosis as the length of hospitalization (in the ward or ICU), appropriate response to treatment based on the percentage of blood oxygen saturation (Spo2), the requirement for supplemental oxygen (with reserve bag mask), transferred or not transferred to ICU intubation, life-threatening complication (acute kidney injury [acute kidney injury (AKI), pulmonary thromboembolism (PTE), acute myocardial infarction (AMI)) and its type, and death or discharge were registered and recorded in the checklist.
Finally, the obtained data were assessed using SPSS version 25 (SPSS Inc, Chicago, Ill, USA), and the relationship between SARS-CoV-2 viral infection in patients with and without underlying heart disease was analyzed. P-values less than 0.05 were considered statistically significant.
4. Results
A total of 400 SARS-CoV-2 patients agreed to participate in the study. Patients were divided into 4 groups as without the underlying disease (n = 100), with a previous CVD (n = 100), non-CVD (n = 100), with both previous CVD and non-CVD (n = 100). The frequency of socio-demographic and anthropometric characteristics of patients is displayed in Table 1 by groups.
| Variables | The Type of Underlying Disease | Total | |||
|---|---|---|---|---|---|
| Group 1 a (n = 100) | Group 2 b (n = 100) | Group 3 c (n = 100) | Group 4 d (n = 100) | ||
| Gender | |||||
| Female | 27 | 48 | 33 | 35 | 143 (35.8) |
| Male | 73 | 52 | 67 | 65 | 257 (64.2) |
| Hospitalization in the ICU | |||||
| No | 94 | 80 | 75 | 72 | 321 (80.3) |
| Yes | 6 | 20 | 25 | 28 | 79 (19.7) |
| Reservoir bag mask | |||||
| No | 72 | 51 | 36 | 41 | 200 (50) |
| Yes | 28 | 49 | 64 | 59 | 200 (50) |
| Intubation | |||||
| No | 97 | 90 | 90 | 86 | 363 (90.8) |
| Yes | 3 | 10 | 10 | 14 | 37 (9.2) |
| Type of life-threatening complication | |||||
| Pneumonia | 7 | 13 | 18 | 18 | 56 (14) |
| ARDS | 1 | 6 | 7 | 4 | 18 (4.5) |
| Sepsis | 0 | 3 | 5 | 12 | 20 (5) |
| AKI | 3 | 9 | 5 | 14 | 31 (7.8) |
| Myocarditis | 1 | 1 | 0 | 5 | 7 (1.8) |
| DVT | 4 | 2 | 4 | 5 | 15 (3.8) |
| Seizure | 0 | 0 | 2 | 0 | 2 (0.5) |
| QT | 0 | 1 | 3 | 3 | 7 (1.8) |
| Myocardial infarction | 1 | 0 | 1 | 0 | 2 (0.5) |
| Heart failure | 0 | 0 | 4 | 9 | 4 (1) |
| Pyelonephritis | 0 | 0 | 1 | 0 | 1 (0.25) |
| No | 85 | 73 | 60 | 64 | 282 (70.5) |
The Frequency (Percentage) of Socio-demographic and Anthropometric Characteristics of Patients
Further, 64.2% (257) of patients were male, and 19.7% (79) of patients required ICU admission. Half of the patients needed the reservoir bag mask. In 9.2% of patients, intubation was performed. During hospitalization, the patients developed pneumonia (14%), AKI (7.8%), sepsis (5%), acute respiratory distress syndrome (ARDS; 4.5%), deep vein thrombosis (DVT; 3.8%), myocarditis (1.8%), QT prolongation (1.8%), heart failure (1%), seizure (0.5%), myocardial infarction (0.5%), and pyelonephritis (0.25%).
The means and SDs of age, length of hospitalization, and length of hospitalization in ICU variables are presented in Table 2 separately for each group.
| Variables | The Type of Underlying Disease | Total | P-Value | |||
|---|---|---|---|---|---|---|
| Group 1 (n = 100) | Group 2 (n = 100) | Group 3 (n = 100) | Group 4 (n = 100) | |||
| Age a | 50.92 ± 15.69 | 53.87 ± 14.76 | 63.46 ± 13.79 | 63.80 ± 12.87 | 58.01 ± 15.36 | < 0.001 |
| Length of hospitalization b | 4.17 ± 3.25 | 5.53 ± 4.85 | 6.12 ± 4.23 | 7.69 ± 7.96 | 5.88 ± 5.50 | < 0.001 |
| Length of hospitalization in the ICU4 | 4.83 ± 3.97 | 7.05 ± 4.57 | 4.88 ± 3.07 | 10.89 ± 9.76 | 7.56 ± 6.99 | 0.027 |
The Means and SDs of Age, Length of Hospitalization, and Length of Hospitalization in the Intensive Care Unit
The total mean age was 58.01 ± 15.36 years. On average, each patient was hospitalized for 5.88 days. In patients who transferred to the ICU, the mean length of hospitalization was 7.56 days. The mean differences in age, length of hospitalization, and length of hospitalization in the ICU variables were statistically significant between the study groups. Multiple comparisons of these variables are displayed in Table 3. The highest mean difference in the age variable is between group 1 and group 4 (P < 0.001), and the lowest mean difference was between group 3 and group 4. In the length of hospitalization variable, the mean difference between group 1 and groups 3 and 4 was statistically significant. The mean length of hospitalization in the ICU in group 4 was 6 days longer than group 3, and this difference was statistically significant.
| Variables | I | J | Mean Difference (I-J) | SE | P-Value |
|---|---|---|---|---|---|
| Age a | Group 4 | Group 1 | 12.88 | 2.025 | < 0.001 |
| Group 2 | 9.93 | 2.025 | < 0.001 | ||
| Group 3 | 0.34 | 2.025 | 0.998 | ||
| Group 3 | Group 1 | 12.54 | 2.025 | < 0.001 | |
| Group 2 | 9.59 | 2.025 | < 0.001 | ||
| Group 2 | Group 1 | 2.95 | 2.025 | 0.465 | |
| Length of hospitalization | Group 4 | Group 1 | 3.52 | 0.760 | < 0.001 |
| Group 2 | 2.16 | 0.760 | 0.193 | ||
| Group 3 | 1.57 | 0.760 | > 0.999 | ||
| Group 3 | Group 1 | 1.95 | 0.760 | 0.001 | |
| Group 2 | 0.59 | 0.760 | 0.344 | ||
| Group 2 | Group 1 | 1.36 | 0.760 | 0.359 | |
| Length of hospitalization in the ICU2 b | Group 4 | Group 1 | 6.06 | 2.974 | 0.068 |
| Group 2 | 3.84 | 1.935 | 0.309 | ||
| Group 3 | 6.01 | 1.819 | 0.005 | ||
| Group 3 | Group 1 | 0.047 | 3.005 | > 0.999 | |
| Group 2 | -2.17 | 1.983 | 0.114 | ||
| Group 2 | Group 1 | 2.21 | 3.077 | > 0.999 |
Multiple Comparisons of Age, Length of Hospitalization, and Length of Hospitalization in the Intensive Care Unit Variables
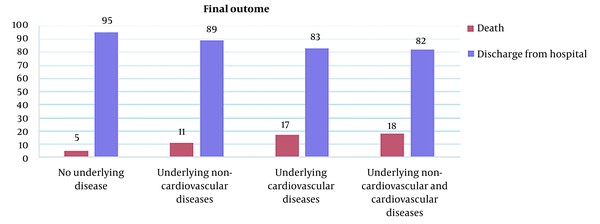
Figure 1 shows the number of deaths and discharges in each group.
The association of final outcomes and socio-demographic and anthropometric features was analyzed using a logistic regression model (Table 4), with a univariable model as the crude odds ratio (OR) and a forward selection model as the adjusted OR.
| Variables | Crude OR (95% CI) | P-Value | Adjusted OR (95% CI) | P-Value |
|---|---|---|---|---|
| Age | 1.038 (1.017 - 1.060) | < 0.001 | 1.048 (1.008 - 1.09) | 0.019 |
| Length of hospitalization | 1.112 (1.062 - .166) | < 0.001 | 0.812 (0.726 - 0.907) | < 0.001 |
| Length of hospitalization in the ICU | 1.287 (1.192 - 1.391) | < 0.001 | - | - |
| The type of underlying disease | ||||
| Group 1 | Base | - | - | - |
| Group 2 | 2.348 (0.785 - 7.026) | 0.127 | - | - |
| Group 3 | 3.982 (1.376 - 11.007) | 0.010 | - | - |
| Group 4 | 4.171 (1.483 - 11.728) | 0.007 | - | - |
| Gender | ||||
| Male | 0.928 (0.505 - 1.706) | 0.810 | - | - |
| Female | Base | - | - | - |
| Hospitalization in the ICU | ||||
| No | Base | - | Base | - |
| Yes | 46.733(20.38 - 107.14) | < 0.001 | 19.33 (5.26 - 71.01) | < 0.001 |
| Reservoir bag mask | ||||
| No | Base | - | - | - |
| Yes | 20.737 (6.33 - 67.85) | < 0.001 | - | - |
| Intubation | ||||
| No | Base | - | Base | - |
| Yes | 88.60 (33.13 - 236.94) | < 0.001 | 72.43 (12.66 - 414.22) | < 0.001 |
| Type of life-threatening complication | ||||
| Pneumonia | 9.86 (5.09 - 19.11) | < 0.001 | 13.29 (3.61 - 48.91) | < 0.001 |
| ARDS | 8.09 (3.04 - 21.52) | < 0.001 | - | - |
| Sepsis | 13.11 (5.05 - 34.04) | < 0.001 | - | - |
| AKI | 2.15 (0.87 - 5.29) | 0.094 | - | - |
| Myocarditis | 9.81 (2.13 - 45.22) | 0.003 | 94.68 (6.74 - 1329.04) | < 0.001 |
| DVT | 6.78 (2.34 - 19.61) | < 0.001 | - | - |
| Seizure | 6.96 (0.43 - 113.04) | 0.173 | - | - |
| QT | 9.81 (2.13 - 45.22) | 0.003 | 104.48 (13.12 - 832.15) | < 0.001 |
| Myocardial infarction | 0 | NA | - | - |
| Heart failure | 2.3 (0.235 - 22.60) | 0.473 | - | - |
| Pyelonephritis | 0 | NA | - | - |
| No | Base | - |
Factors Associated with COVID-19 Patients’ Final Outcomes
The risk of death was higher in elderly patients than in younger patients (crude OR = 1.038 and adjusted OR = 1.048). Mortality was lower in patients with long hospitalization (adjusted OR = 0.812). Mortality in the underlying non-CVD and CVD group was 4.17 (crude OR) times higher than the group without the disease. This rate was 3.98 (crude OR) times higher in the underlying CVD group than in the baseline. The adjusted OR of patients transferred to the ICU was 19.33. Also, the adjusted OR for patients who were intubated was 72.43. The mortality rate for patients who got pneumonia during hospitalization was 13.29 times higher. The mortality rate for patients who got pneumonia, myocarditis, and QT during hospitalization was 13.29, 94.68, and 104.48 times higher than the baseline, respectively.
5. Discussion
The present study is a descriptive-analytical investigation of patients with COVID-19, reviewing the prognosis in patients with and without underlying CVDs in Imam Khomeini Hospital, Tehran, Iran.
Regarding COVID-19, a truly relevant illness is probably any form of heart disease. This is because first, the prevalence of cardio-cerebrovascular disorders is much greater in patients with COVID-19 compared to the general population (13), and second, patients with hypertension, cardio-cerebrovascular disorders, or diabetes are more expected to stay in the ICU or expire because of SARS-CoV-2 (12-14). The overall rates of hypertension, cardio-cerebrovascular complications, and diabetes were approximately 2-fold, 3-fold, and 2-fold, respectively, higher in ICU cases than in non-ICU inpatients (13). Recently, the Chinese Center for Disease Control and Prevention reported the results of a study with the largest sample size to date in the field of COVID-19 in China, indicating that the overall mortality rate was 2.3%, and in comparison, the number of deaths was 10.5% in cases with underlying CVD (8).
In the present study, the number of deaths in COVID-19 cases with underlying non-CVD and CVD was 4.17 times higher than that of COVID-19 patients without any underlying disorders. The mortality rate was 3.98 times higher in patients with underlying CVD than the baseline.
On the other hand, as a minimum, 8.0% of COVID-19 cases suffered any type of acute heart injury, but additional investigations revealed that the occurrence of cardiac injury is about 13-fold greater for patients requiring ICU than non-ICU inpatients (13, 15, 16).
A recent investigation indicated that during the COVID-19 pandemic, patients with underlying CVD, coronary heart disease, hypertension, viral illness, cardiomyopathy, or other comorbid disorders are more likely to suffer myocardial damage (17). Myocardial cell injury occurs through several mechanisms, including destabilized coronary plaque, systemic inflammatory reactions, virus-related damage, and serious hypoxia (6, 17, 18).
5.1. Conclusions
Underlying cardiovascular complications are possibly associated with COVID-19 prognosis. Predicting the effect of common complications on COVID-19 prognosis support health professionals and authorities to consider appropriate facilities and top approaches to managing COVID-19 in hospitals. Further investigations with longer durations and greater sample populations are highly recommended.