1. Background
The emergence of the coronavirus named severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) uniquely challenged health care systems. The high infectivity and transmissibility of this virus, even in the asymptomatic stage, along with its relatively low virulence, resulted in its rapid transmission beyond borders and the emergence of a pandemic.
The first case of this disease was diagnosed on December 8th, 2019, in Hubei province, China (1). Since then, it rapidly spread to other countries worldwide (2). The dominant symptom of COVID-19 is respiratory system involvement, ranging from a mild pseudo-influenza disease to a deadly acute respiratory syndrome.
Similar to other respiratory infections, the presence of cardiovascular diseases (CVD) and their risk factors can increase susceptibility to COVID-19. Moreover, COVID-19 can worsen underlying CVDs and precipitate new cardiac complications. Sufficient knowledge and understanding of the relationship between CVDs, such as arrhythmia, and COVID-19 are highly required for the effective management of this disease.
Given the newly-emerged nature of COVID-19 and our limited knowledge about its complications and treatment, this study aims to investigate hospitalized COVID-19 patients in terms of arrhythmia. Arrhythmia is one of the CVD complications of COVID-19. Bradyarrhythmia and tachyarrhythmia are the two recognized arrhythmias in COVID-19, with a prevalence of 16.7% so far (3).
Arrhythmia is more prevalent in patients requiring ICU hospitalization compared to those not needing ICU treatments (44% vs. 8.9%).
The arrhythmogenic effects of COVID-19 are anticipated, and they can influence the disease outcome. This is particularly important in patients at risk of cardiac arrhythmia, either secondary due to conditions and medication or inherited syndromes such as prolonged QT syndrome, Brugada syndrome, short QT syndrome, and catecholaminergic polymorphic tachyarrhythmia, and can challenge the treatment of the COVID-19 pandemic.
Since these patients are prone to COVID-19-related proarrhythmic effects such as fever, stress, electrolyte disturbances, and antiviral medication administration, prolonged acquired QT is particularly more prevalent in older patients because provocative causes are more common in this age group (4, 5).
Drugs are among the causes of arrhythmia during COVID-19. Many drugs are capable of blocking cardiac potassium currents and disturbing ventricular repolarization, resulting in a prolonged QT interval and, hence, increasing the risk of malignant arrhythmia. Moreover, many drugs can disrupt drug metabolism.
For instance, by inhibiting the P34A4 cytochrome, the plasma level of drugs prolonging the QT interval increases, thereby increasing the risk of arrhythmia. Chloroquine, an anti-malaria drug with a wide range of antiviral effects, is used in COVID-19 treatment. This drug interferes with the final glycosylation of ACE2 and, due to its association with quinidine, can result in QT prolongation and QT-related malignant arrhythmia (6, 7).
Chloroquine has modest impacts on prolonged QT in patients without prolonged QT syndrome. As both hydroxychloroquine and chloroquine are metabolized by P3A4, combining COVID-19 treatments with other antiviral drugs such as ritonavir plus lopinavir (both inhibiting P3A4 cytochrome), azithromycin (a macrolide antibiotic with antibacterial, antiviral, and P3A4 inhibition effects), or remdesivir (with QT-prolonging effects) can enhance the risk of arrhythmia due to a prolonged QT interval (8, 9).
The reason could be the increased plasma level of the drug and, consequently, prolonged QT. On the other hand, the majority of hospitalized COVID-19 patients have a fever (10). Although the relationship between fever and arrhythmia has not been well documented, sepsis is a risk factor for acquired prolonged QT, and septic shock is one of the clinical symptoms of COVID-19, so fever could also be included as one of the arrhythmia causes (10, 11).
Moreover, COVID-19 accompanied by diarrhea (which can result in hypokalemia affecting the QT interval) is among the other reasons for arrhythmia during COVID-19. Given the new emergence of COVID-19, further investigations are undoubtedly required.
2. Objectives
The present study is also designed to investigate the causes of arrhythmia during COVID-19 disease.
3. Methods
In this retrospective observational study, we included 288 consecutive COVID-19 patients who were admitted to the emergency department from June 15 to November 15, 2020. It was carried out in a referral academic hospital after approval from the Ethics Committee of Iran University of Medical Science. Patients with a history of old arrhythmia, including atrial fibrillation, flutter, and atrial tachycardia, were excluded. The study was performed in accordance with the principles of the Declaration of Helsinki and was reviewed and approved by the Ethics Committee of Iran University of Medical Sciences (ID: IR.IUMS.REC.1399.006), and written informed consent was provided by all patients participating in the study.
Electrocardiography was performed within the first 24 hours of hospitalization, and hematological biomarkers such as hemoglobin, RBC, WBC, platelet count, RDW, PDW, and troponin were measured. If these tests were done more than once a day, the lowest number was archived.
The prevalence and type of incident sustained arrhythmias were evaluated. Our primary aim was to explore the incidence and predictors of any new-onset arrhythmias in hospitalized COVID-19 patients. The secondary aim was to determine the correlation between new-onset arrhythmias and both baseline characteristics and in-hospital mortality.
3.1. Statistic and Analysis
Data analysis was performed using SPSS software for Windows (Statistical Product and Service Solutions, version 20.0, SPSS Inc., Chicago, IL, USA). Statistical analysis was presented as mean ± SD. The χ2 test was used for analysis of categorical data, and the Student t-test or Mann-Whitney U test were used for continuous data. Univariable and multivariable risk ratios (RR) for both incident arrhythmias and the outcomes of interest were calculated using logistic regression models and presented as RR with their 95% confidence intervals (CI). All independent variables with a P-value < 0.1 for the association with the response variable in univariable analysis were tested in the multivariable model. A P-value less than 0.05 was considered statistically significant.
4. Results
In this study, 288 consecutive patients diagnosed with COVID-19 were enrolled. Cardiac arrhythmia was detected in 68 patients (23.6%). Arrhythmia was categorized as atrial fibrillation (11.8%), ventricular tachycardia (4.2%), premature ventricular contraction (2.7%), and paroxysmal supraventricular tachycardia (2.4%). Underlying diseases in patients included DM (22%), IHD (15.2%), HTN (18%), and COPD (7.9%) (Table 1).
There were 26 women and 42 men in the Arrhythmia group and 92 women and 128 men in the No Arrhythmia group. Gender was not significantly different between the groups (P = 0.71). The mean age in the Arrhythmia group was 70.6 ± 13.8, and in the No Arrhythmia group, it was 65.6 ± 15.8 (P = 0.105) (Table 1).
| Variables | Values |
|---|---|
| Age | 66.7 ± 14.1 |
| Sex (M/F) | |
| Ward | (170/118) |
| (Ward/ ICU) | (220/68) |
| Underlying disease | |
| DM | 64 (22) |
| IHD | 44 (15.2) |
| HTN | 52 (18) |
| COPD | 23 (7.9) |
| Arrhythmia | |
| Atrial fibrillation | 34 (11.8) |
| Ventricular tachycardia | 12 (4.2) |
| Premature ventricular contraction | 8 (2.7) |
| Paroxysmal supraventricular tachycardia (PSVT) | 7 (2.4) |
Abbreviations: DM, Diabetes; IHD, Ischemic Heart Disease; HTN, Hypertension.
a Values are presented as No. (%) or mean ± SD.
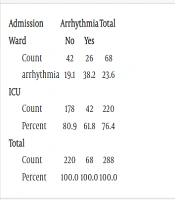
Sixty-eight patients were admitted to the ward, and 220 patients were admitted to the ICU. Arrhythmia in ICU patients (61.8%) was significantly higher than in ward patients (38.2%) (P = 0.022) (Table 2).
| Admission | Arrhythmia | Total | |
|---|---|---|---|
| Ward | No | Yes | |
| Count | 42 | 26 | 68 |
| arrhythmia | 19.1 | 38.2 | 23.6 |
| ICU | |||
| Count | 178 | 42 | 220 |
| Percent | 80.9 | 61.8 | 76.4 |
| Total | |||
| Count | 220 | 68 | 288 |
| Percent | 100.0 | 100.0 | 100.0 |
a Values are expressed as (%).
Among lab tests, including CPK, LDH, Troponin, D-DIMER, Cr, AST, ALT, HB, MCV, RDW, Lymphocyte, PMN, PLT, MPV, BS, K, FBS, CRP, and Ferritin, none were prognostic factors for arrhythmia in COVID-19 patients (P > 0.05) (Table 3).
| Lab | N | Mean ± Std. Deviation |
|---|---|---|
| CPK | ||
| Yes | 13 | 394.15 ± 569.10 |
| No | 12 | 209.67 ± 194.4 |
| LDH | ||
| Yes | 21 | 671.71 ± 359.6 |
| No | 29 | 768.41 ± 369.3 |
| Troponin | ||
| Yes | 17 | 78.3971 ± 147.38 |
| No | 38 | 2.0233 ± 4.43 |
| D.DIMER | ||
| Yes | 7 | 1.85 ± 5.3 |
| No | 29 | 2.52 ± 2.8 |
| Cr | ||
| Yes | 34 | 1.38 ± .8 |
| No | 106 | 1.53 ± 1.3 |
| AST | ||
| Yes | 24 | 48.67 ± 42.3 |
| No | 38 | 58.95 ± 47.3 |
| ALT | ||
| Yes | 23 | 37.57 ± 34.8 |
| No | 38 | 46.13 ± 52.7 |
| HB | ||
| Yes | 34 | 12.14 ± 2.7 |
| No | 104 | 12.24 ± 2.9 |
| MCV | ||
| Yes | 34 | 89.37 ± 9.8 |
| No | 104 | 85.86 ± 10.4 |
| RDW1 | ||
| Yes | 1 | 18.50 ± 1.3 |
| No | 75 | 15.26 ± 2.7 |
| Lymphocyte | ||
| Yes | 34 | 21.11 ± 21.9 |
| No | 100 | 16.03 ± 10.5 |
| PMN1 | ||
| Yes | 34 | 70.53 ± 24.9 |
| No | 100 | 76.39 ± 13.5 |
| PLT | ||
| Yes | 34 | 212.56 ± 99.0 |
| No | 101 | 233.78 ± 99.7 |
| MPV1 | ||
| Yes | 1 | 11.80 ± 1.3 |
| No | 69 | 10.19 ± 1.16 |
| BS | ||
| Yes | 25 | 150.88 ± 75.3 |
| No | 47 | 175.43 ± 96.8 |
| K1 | ||
| Yes | 34 | 4.26 ± .6 |
| No | 81 | 4.40 ± .7 |
| FBs | ||
| Yes | 11 | 164.18 ± 47.3 |
| No | 41 | 150.63 ± 74.7 |
| CRP | ||
| Yes | 28 | 71.286 ± 36.5 |
| No | 59 | 79.325 ± 76.9 |
| Ferritin | ||
| Yes | 6 | 430.67 ± 525.2 |
| No | 15 | 361.20 ± 313.3 |
5. Discussion
Since the risk of arrhythmia increases with the enhancement of the systemic inflammatory response in infectious diseases (12), significant myocardial inflammation and cardiac arrest have been reported along with ventricular arrhythmia in COVID-19 patients (13-16). In patients with severe infection, hyperinflammatory responses or cytokine storms can result in multi-organ disorders (17). Recent studies have shown that increased cytokines (especially interleukin-26) can elevate the risk of arrhythmia through various mechanisms, including HERG blocking and prolonging the QT interval (18-20).
Besides cardiac failure and infarction, arrhythmia is one of the three major risks of viral infections in myocarditis, inflammatory effects, and increased sympathetic system provocation. In a report from Wuhan, China, 16.7% of hospitalized patients and 44.4% of ICU patients with a definite COVID-19 diagnosis had cardiac arrhythmia (21). Similarly, in our study, cardiac arrhythmia was detected in 23.6% of hospitalized patients and 61.8% of ICU patients with COVID-19. Arrhythmia was categorized as atrial fibrillation (11.8%), ventricular tachycardia (4.2%), premature ventricular contraction (2.7%), and paroxysmal supraventricular tachycardia (2.4%).
The evidence on ICU patients has shown that only half of the patients with arrhythmia experienced cardiac damage (increased troponin) despite the high frequency of arrhythmia (50% of cases). This indicates that factors other than myocardial injuries are involved in arrhythmia. For instance, pharmacologic treatments can increase prolonged QT (22). Prolonged QT has been observed in patients with high CRP, which is associated with IL-6 concentration (21).
Therefore, the inflammatory response of the body in COVID-19 not only affects the lungs but is also involved in cardiovascular complications such as arrhythmia due to prolonged QT, highlighting the need for more attention to this issue.
Among the drug regimens used, more concerns are focused on chloroquine/hydroxychloroquine with or without azithromycin, which can increase the risk of prolonged QT and, hence, torsades de pointes arrhythmia (a form of polymorphic ventricular tachycardia) (23, 24). The majority of drugs leading to prolonged QT operate by blocking potassium ether-a-go-go related gene (HERG) channels (25).
In previous studies, other risk factors for arrhythmia include drug interference, female sex, structural cardiac deficits, genetic polymorphisms, electrolyte disorders, bradycardia, and hepatic diseases (26, 27). In contrast, in our study, sex, age, and levels of CPK, LDH, troponin, D-dimer, Cr, AST, ALT, Hb, MCV, RDW, lymphocytes, PMN, PLT, MPV, BS, K, FBS, CRP, and ferritin were not prognostic factors for arrhythmia in COVID-19 patients. A study in 2012 showed that a 5-day treatment with azithromycin had a significant association with cardiac death. Azithromycin is a macrolide antibiotic that enhances the QT interval and the risk of torsades de pointes (28, 29).
5.1. Conclusions
Arrhythmia was observed in 23.6% of patients with COVID-19 and in 61.8% of ICU patients with COVID-19. Cardiac arrhythmias were not associated with sex, age, or lab tests.