1. Background
An adequate amount of vitamin D is essential for the maintenance of good bone and skeletal health as well as for calcium homeostasis (1). Vitamin D deficiency can be an important risk factor for osteomalacia and osteoporosis in adults. It is associated with secondary hyperparathyroidism and enhanced bone turnover, eventually leading to bone loss (2). It is extensively prevalent in the Indian population, and the importance of understanding the effects of vitamin D deficiency is well documented in several studies (3, 4).
Bone cells express vitamin D receptors, thereby denoting an important role of vitamin D in bone remodeling (5). The amplitude of bone remodeling can be determined using the bone formation marker N-terminal propeptide of type I procollagen (PINP) and the bone resorption marker serum carboxy-terminal telopeptide of type- I collagen (sCTX). Serum 25(OH)D concentrations are considered to be the best functional indicator of vitamin D status (6, 7). The relationship between serum 25(OH)D levels with bone turnover markers and bone mineral density (BMD) in women residing in different geographical areas is debatable. A recent study has shown a negative correlation of only the bone formation marker with vitamin D (8). In contrast, another study has shown a negative correlation of both bone resorption and formation markers with vitamin D levels, post supplementation (9). A majority of studies did not find any correlation between either bone markers and vitamin D (10-12). A study on Saudi nationals found a positive correlation of BMD with vitamin D (13), whereas another study on the same population observed no correlation between them (14).
2. Objectives
Considering the controversial results regarding the relationship between vitamin D levels and BMD in different populations, the present study evaluated the vitamin D status and bone health in healthy Indian women. The aim was to determine whether vitamin D insufficiency or deficiency affects bone turnover and BMD by considering the relationship between bone turnover markers and BMD in vitamin D sufficient, insufficient, and deficient subjects.
3. Methods
3.1. Study Design
This cross-sectional observational study was conducted at the Department of Molecular Immunodiagnostics of the National Institute for Research in Reproductive Health Institute, and the study protocol was approved by the Institutional Ethics Committee for Clinical Studies. Camps were organized near the study site for creating awareness about the importance of vitamin D and osteoporosis, encouraging women to participate in the study. A total of 310 healthy Indian women, aged 25 - 65 years, were recruited in the study after obtaining written informed consent from them. Women on bone compromising drugs, women with hepatic or renal disorders, metabolic bone disease, hypercortisolism, sterility, oligomenorrhea, diabetes, and malignancy, as well as pregnant and lactating women, were excluded. The clinical history of the participants and their demographic data, including age, height, weight, body mass index (BMI), menopausal status, and menopause duration, were recorded. Menopause was defined as the natural cessation of menstruation for more than 12 months. To reduce pre-analytical variations during the estimation of bone markers, a fasting blood sample was collected from all the participants between 0900 - 1000 hrs, serum was separated, and aliquots were frozen at -80ºC for further analysis. Serum Ca, and P levels were estimated in an auto-analyzer using reagents from ERBA (Mannheim, Germany). Serum levels of 25(OH) vitamin D (Calbiotech, USA; Sensitivity: 0.67 ng/mL), parathyroid hormone (PTH) (Biomerica Inc., USA; Sensitivity: 0.67 ng/mL), Serum Crosslaps, CTX-I (Immunodiagnostic Systems, UK; Sensitivity: 0.020ng/mL), and PINP (Elabscience, USA; Sensitivity: 9.38 pg/mL) were estimated using commercial ELISA kits. The study cohort was classified based on menopausal status as premenopausal or postmenopausal, and each group was further classified based on serum vitamin D levels as sufficient (≥ 30 ng/mL), insufficient (16 - 29 ng/mL), and deficient (≤ 15 ng/mL).
3.2. Bone Mineral Density
All the participants underwent BMD measurement at the lumbar spine, hip, and its sub-regions with a Lunar iDXA densitometer (GE, Healthcare) by a trained operator. Quality control procedures were carried out following the manufacturer’s recommendations, and results were expressed as T-score. The instrument variation was determined regularly with a weekly calibration procedure using a phantom supplied by the manufacturer. According to the World Health Organization (WHO) criteria, BMD values were stratified as normal, osteopenic, and osteoporotic based on T-score.
3.3. Statistical Analysis
All statistical analyses were performed with Graph Pad Prism version 5.01. Means ± standard deviation (SD) were used to express standard descriptive statistics. The non-parametric test used as the data did not show Gaussian distribution. Differences in the median were investigated using either the Mann-Whitney test or the Kruskal-Wallis test with Dunn’s post hoc analysis. Spearman’s rank correlation was calculated to indicate the strength of the relation between the two parameters. A p-value of less than 0.05 was considered statistically significant.
4. Results
4.1. Anthropometric Data
The basic anthropometric data of the study cohort are given in Table 1. The mean age of the women was 41.54 ± 11.55 years, and their mean BMI was 24.75 ± 4.78 kg/m2. The mean BMD at the spine was 1.02 ± 0.166 g/cm2, and the mean BMD at the femoral neck (FN) was 0.876 ± 0.123 g/cm2. All the women had normal levels of Ca and P, while the mean 25(OH)D level was 18.95 ± 15.35 ng/mL. Moreover, the mean PTH, CTX, and PINP levels were 59.23 ± 23.37 pg/mL, 0.457 ± 0.299 ng/mL, and 37.35 ± 16.24 ng/mL, respectively.
| Characteristics | Cohort (N = 310) | Premenopausal (N = 215) | Postmenopausal (N = 95) |
|---|---|---|---|
| Age, years | 41.54 ± 11.55 | 34.32 ± 8.57 | 52.10 ± 5.69 |
| BMI, kg/m2 | 24.75 ± 4.78 | 23.92 ± 4.33 | 25.66 ± 4.55 |
| BMD Spine, g/cm2 | 1.02 ± 0.166 | 1.089 ± 0.132 | 0.913 ± 0.146 |
| BMD FN, g/cm2 | 0.876 ± 0.123 | 0.907 ± 0.114 | 0.808 ± 0.116 |
| T-score spine | -1.38 ± 1.33 | -0.81 ± 1.06 | -2.31 ± 1.17 |
| T-score FN | -1.12 ± 0.88 | -0.93 ± 0.8 | -1.65 ± 0.81 |
| Calcium, mg/dL | 9.24 ± 0.25 | 9.18 ± 1.02 | 9.19 ± 0.94 |
| Phosphorous, mg/dL | 5.17 ± 1.35 | 5.06 ± 1.39 | 5.08 ± 1.12 |
| Vitamin D, ng/mL | 18.95 ± 15.35 | 18.71 ± 17.09 | 41.63 ± 16.82 |
| PTH, pg/mL | 57.45 ± 17.76 | 55.50 ± 13.46 | 58.19 ± 19.27 |
| sCTX, ng/mL | 0.457 ± 0.299 | 0.401 ± 0.234 | 0.515 ± 0.263 |
| PINP, ng/mL | 37.35 ± 16.24 | 38.91 ± 15.81 | 59.19 ± 19.27 |
Abbreviations: BMD, bone mineral density; BMI, body mass index; FN, femoral neck; PINP, N-terminal propeptide of type I procollagen; PTH, parathyroid hormone; sCTX, serum carboxy-terminal telopeptide of type-I collagen.
aValues are expressed as mean ± SD.
4.2. Association of Vitamin D with Various Parameters
The study cohort was also classified based on vitamin D levels as sufficient (≥ 30 ng/mL), insufficient (16 - 29 ng/mL), and deficient (≤ 15 ng/mL). According to the WHO recommendation, the postmenopausal women were classified as normal, osteopenic, and osteoporotic based on their T-scores, whereas the premenopausal women were classified as normal and with low bone mass based on their Z-score (Table 2).
| Characteristics | Values | Premenopausal (N = 215) | Postmenopausal (N = 95) | ||||
|---|---|---|---|---|---|---|---|
| Normal BMD | Low BMD | Normal | Osteopenia | Osteoporosis | |||
| Sufficient | 46 (14.84) | 29 (90.62) | 3 (9.38) | 1 (7.14) | 4 (28.57) | 9 (64.29) | |
| Insufficient | 97 (31.29) | 53 (94.64) | 3 (5.36) | 2 (4.87) | 17 (41.46) | 22 (53.65) | |
| Deficient | 167 (53.87) | 121 (95.27) | 6 (4.73) | 6 (15) | 17 (42.5) | 17 (42.5) | |
aValues are expressed as No. (%).
bFrequency in parenthesis: the premenopausal women classified as normal (Z-score > -2.0) and low BMD (Z-score ≤ -2.0); the postmenopausal women classified as normal (T-score > -1.0), osteopenic (T-score -1.0 to -2.5), and osteoporotic (T-score ≤ -2.5); sufficient (vitamin D ≥ 30 ng/mL), insufficient (vitamin D 16 - 29 ng/mL), and deficient (vitamin D ≤ 15 ng/mL).
In the study cohort, 59.08% of the premenopausal women had vitamin D deficiency, of which 62.99% had low bone mass. However, the prevalence of vitamin D deficiency in the postmenopausal women was less (42.12%) compared to the premenopausal women (Table 2).
We further checked whether BMD at the spine and FN, biochemical parameters serum Ca and P, PTH, and bone turnover markers PINP and sCTX varied between the sufficient, insufficient, and deficient groups of premenopausal and postmenopausal women, and the results are summarized in Tables 3 and 4.
| Parameters | Sufficient (N = 32) | Insufficient (N = 56) | Deficient (N = 127) | P Value |
|---|---|---|---|---|
| Age, years | 42 (36 - 46) | 34 (30 - 41)b | 33 (25 - 39)b | 0.0001 |
| BMD spine, g/cm2 | 1.056 (0.88 - 1.292) | 1.075 (0.845 - 1.428) | 1.052 (0.837 - 1.571) | 0.645 |
| BMD FN, g/cm2 | 0.876 (0.685 - 1.113) | 0.923 (0.687 - 1.206) | 0.886 (0.648 - 1.291) | 0.678 |
| Calcium, mg/dL | 8.4 (7.5 - 10.5) | 8.7 (6.2 - 11.7) | 9.5 (7.1 - 11.4)b, c | 0.001 |
| Phosphorous, mg/dL | 4.65 (3.6 - 6.72) | 5.26 (3.4 - 11.44) | 4.61(2.5 - 12.84)d | 0.003 |
| PTH, pg/mL | 53.68 (21.04 - 60.45) | 64.29 (39.68 - 107.2) | 64.89 (42.69 - 112.6) | 0.038 |
| PINP, pg/mL | 27.62 (6.04 - 67.22) | 36.82 (13.94 - 83.6) | 36.25 (12.94 - 95.1) | 0.034 |
| sCTX, ng/mL | 0.444 (0.069 - 1.093) | 0.433 (0.153 - 0.945) | 0.326 (0.016 - 1.157)d, e | 0.002 |
Abbreviations: BMD, bone mineral density; FN, femoral neck, PINP, N-terminal propeptide of type I procollagen; PTH, parathyroid hormone; sCTX, serum carboxy-terminal telopeptide of type-I collagen.
aValues are expressed as median (range).
bP < 0.001 compared to sufficient.
cP < 0.001 compared to insufficient.
dP < 0.05 compared to insufficient.
eP < 0.05 compared to sufficient.
| Parameters | Sufficient (N = 14) | Insufficient (N = 41) | Deficient (N = 40) | P Value |
|---|---|---|---|---|
| Age, years | 53 (44 - 61) | 52 (41 - 65) | 53 (42 - 63) | 0.849 |
| BMD spine, g/cm2 | 0.889 (0.640 - 1.099) | 0.890 (0.576 - 1.286) | 0.911 (0.672 - 1.296) | 0.868 |
| BMD FN, g/cm2 | 0.817 (0.652 - 1.032) | 0.815 (0.567 - 0.956) | 0.774 (0.612 - 1.194) | 0.461 |
| Calcium, mg/dL | 9.4 (7.2 - 11.1) | 9.1 (7.7 - 11.3) | 8.55 (8 - 10.6) | 0.147 |
| Phosphorous, mg/dL | 5.06 (4.06 - 6.17) | 5.04 (2.8 - 9.39) | 4.87 (3.3 - 8.12) | 0.482 |
| PTH, pg/mL | 44.43 (19.04 - 70.99) | 52.41 (23.01 - 106.8) | 60.01 (30.98 - 112.1) | 0.032 |
| PINP, pg/mL | 36.95 (22.20 - 53.08) | 31.24(10.90 - 51.6) | 34.7 (21.68 - 72.76)b | 0.028 |
| sCTX, ng/mL | 0.759 (0.308 - 0.953) | 0.434 (0.03 - 0.940)c | 0.444 (0.06 - 1.05) | 0.029 |
Abbreviations: BMD, bone mineral density; FN, femoral neck, PINP, N-terminal propeptide of type I procollagen; PTH, parathyroid hormone; sCTX, serum carboxy-terminal telopeptide type-I collagen.
aValues are expressed as median (range).
bP < 0.05 compared to insufficient.
cP < 0.05 compared to sufficient.
The vitamin D status did not affect BMD at the spine or FN in both premenopausal and postmenopausal women. However, levels of Ca and P varied significantly among the three groups in the premenopausal women, as compared to the postmenopausal women. Moreover, levels of intact PTH and PINP were significantly different among the three groups in the premenopausal and postmenopausal women, but post hoc test results were non-significant. In addition, sCTX levels varied significantly between the three groups in both premenopausal and postmenopausal women.
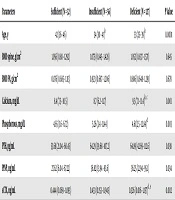
4.3. Correlation of Vitamin D Levels with Study Parameters
Vitamin D levels were correlated with age, BMI, BMD, biochemical parameters, and bone turnover markers, as depicted in Table 5. Interestingly, sCTX positively correlated with vitamin D in the premenopausal (Spearman’s r = -0.228, P < 0.01) and postmenopausal (Spearman’s r = -0.244, P < 0.05) women.
Abbreviations: BMI, body mass index; BMD, bone mineral density; FN, femoral neck, PINP, N-terminal propeptide of type I procollagen; PTH, parathyroid hormone; sCTX, serum carboxy-terminal telopeptide of type-I collagen.
aValues expressed as Spearman’s correlation coefficient.
bP value < 0.001.
cP value < 0.05.
5. Discussion
Vitamin D deficiency is widespread in India, with its prevalence ranging from 70% - 100% (15). The cut-off for vitamin D sufficiency in our study was considered as serum 25(OH) vitamin levels ≥ 30 ng/mL and deficiency as ≤ 15 ng/mL. The reason is that PTH levels fall down considerably at this threshold value of vitamin D, as indicated in many cross-sectional studies (16). Vitamin D deficiency was about 1.4 times more prevalent among the premenopausal women compared to postmenopausal women. This is consistent with other Indian studies, where women at reproductive age have a higher prevalence of vitamin D deficiency (17-19). This could be attributed to low dietary calcium and vitamin D intake, limited exposure to sunlight, socio-cultural factors, geographical factors, level of pollution, clothing, pigmentation of the skin, and time spent outdoors, which affect the production of subcutaneous vitamin D (20).
Vitamin D levels did not show any correlation with BMD in our study. There has been a lack of consensus on the relationship of vitamin D with BMD. Few studies have reported the association of vitamin D with BMD in the south Asian population (21-23). However, other studies have indicated a lack of association between vitamin D and BMD in the same population (18, 24, 25). The disparity can be attributed to differences in ethnicity, age group, and differences in cut-off limits to define vitamin D deficiency and insufficiency.
Calcium levels had an inverse correlation with vitamin D, whereas serum phosphorous was positively correlated with vitamin D in premenopausal women. Serum calcium increases due to enhanced bone resorption, and demineralization carried out using PTH in the vitamin D deficiency state (26). Although the exact mechanism of phosphorous release and reclamation is not clear in various organs, the positive effect is due to phosphate regulation carried out with vitamin D, PTH, and fibroblast growth factor 23 (FGF23), collectively called calciotropic and phosphpotropic hormone, by modulating intestinal phosphate absorption and renal phosphate reabsorption (27).
A significant negative correlation was observed between vitamin D and PINP. An increase in PTH levels leads to an increase in PINP levels, indicating that bone remodeling requires an adequate amount of PTH (8, 28). Hence, the increase in PINP levels is due to the increase in PTH levels. Unlike other studies, which showed either negative (8) or no (29) correlation of CTX with vitamin D, we observed a positive correlation between sCTX and vitamin D with a marked effect observed in premenopausal women. This is in line with a study carried out in Tehran on healthy adolescents (30). CTX levels were high in females less than 20 years, decreased between the age range of 20 - 49 years, and increased afterward (31). A cross-sectional study on Indian women indicated that hypovitaminosis D existed in about 88% of women at reproductive age (20 - 49 years) (17). Low sCTX and vitamin D in the premenopausal women of our cohort may justify the strong positive association between them. Surprisingly, we found a higher level of vitamin D in the postmenopausal women compared to the premenopausal ones. This could be due to the intake of vitamin D supplements, which was not noted. Hence, a positive association of vitamin D with sCTX was observed.
There were some limitations to the study. Firstly, the confounding factors for bone turnover markers were not adjusted during analysis. Secondly, we could not determine whether the participants, especially the postmenopausal women, were on vitamin D supplementation, which could be the reason for the lower prevalence of vitamin D deficiency in postmenopausal women.
Nevertheless, this observational study reveals that although vitamin D does not affect BMD directly, it influences the bone remodeling process. In our study, vitamin D was observed to have a positive correlation with CTX and a negative correlation with PINP. Bone turnover markers varied across the three groups classified based on vitamin D levels based on. Thus, vitamin D levels may be a predisposing risk factor for osteoporosis and should be considered along with BMD measurements for therapeutic purposes.