1. Background
Chronic kidney disease (CKD) is a globally growing health concern incurring great costs to healthcare systems due to its morbidity and mortality (1). That CKD is usually present in subjects suffering from multiple comorbidities increases the challenge of identifying significant prognostic predictors among this often complicated population. Also, CKD importance is featured by its role in increasing the risk of cardiovascular disease (CVD) (2). It is a fact that overt disease is only the tip of the CKD ‘iceberg’ (3), and there are effective measures that prevent its progression to end-stage renal disease (ESRD) (4). Therefore, studying CKD and understanding the dynamics that drive its progression and influence its outcomes is not only a national priority but also a global challenge.
Among many indicated abnormalities, suboptimal thyroid function has also been identified as a potential player in the development and outcome of CKD (5-7). It is considerably associated with a higher risk of CKD without considering other conventional risk factors, among which age might affect this association (8). Thyroid hormone affects the entire body, including both the renal and cardiovascular systems. While the thyroid hormone influences the growth and function of the kidney, it is also affected by the kidney, as it is involved in the clearance of iodine, thyroid-stimulating hormone (TSH), and thyrotropin-releasing hormone (TRH), besides the deiodination of T4 to produce T3, being biologically a more active form. Subjects that suffer from acute kidney injury present with low T4, T3, and TSH levels, known as a euthyroid sick syndrome (9, 10). However, when the insult to the kidney occurs chronically, as is the case in CKD, these changes are less evident.
Despite the growing prevalence of CKD and the postulated detrimental effects of even subclinical thyroid dysfunction on its outcome, there is little data on the prevalence of subclinical hypothyroidism (SCH) in subjects with CKD. That thyroid and kidney function can affect each other, with thyroid hormones necessary for kidney growth, development, and electrolyte homeostasis, and the kidney responsible for thyroid hormone metabolism and elimination, makes it very difficult to establish a causal link between thyroid and kidney dysfunction. However, looking at the prevalence of the concurrence of these conditions may be a step towards better recognizing a more at-risk population subgroup. To this end, we examined data from the Tehran thyroid study (TTS) to determine the prevalence of SCH in subjects with CKD.
2. Methods
2.1. Study Population
The present study was conducted within the framework of the TTS, which is a prospective population-based cohort study performed on the residents of district-13 of Tehran (11). The study aimed to evaluate the natural course and prevalence of thyroid diseases and their long-term consequences related to CVD, cardiovascular mortality, and all-cause mortality in the iodine-sufficient urban population of Tehran. Details of the study methods have previously been described (12). In brief, 5,786 persons (2,376 men and 3,410 women), aged 20 years or over, were evaluated in the TTS cross-sectional phase, from March 1999 to December 2001. The study participants were followed every three years for 12 years in a prospective follow-up study.
For this study, we included subjects who had measurements of FT4 and TSH (N = 1,368, missing = 4), and participants were divided into two groups: those with eGFR > 60 mL/min/1.73 m2 and those with eGFR ≤ 60 mL/min/1.73 m2 at baseline. We excluded seven subjects with high FT4 and TSH or low FT4 and TSH and 145 pregnant women.
In each phase, a detailed interview was performed by a trained interviewer. Demographic data and information regarding smoking habits, thyroid surgery, history of radioiodine ablation, and detailed medication use (anti-thyroid, thyroxin, or any medication interfering with thyroid function tests, or anti-diabetic agents and lipid-lowering agents) were obtained. The study protocol was approved by the Ethics Committee of the Research Institute for Endocrine Sciences of Shahid Beheshti University of Medical Sciences. Written informed consent was obtained from all subjects.
2.2. Laboratory Measurements
All participants gave fasting blood samples drawn between 7:00 and 9:00 a.m. into vacutainer tubes at each reassessment. Details of biochemical measurements were completely presented previously (13). Serum creatinine was measured at baseline and after six years of follow-up, according to the standard colorimetric Jaffe_Kinetic reaction method. Both intra- and inter-assay CVs were below 3.1%; all analyses were performed using commercial kits (Pars Azmoon Inc., Tehran, Iran). We used the modification of diet in renal disease (MDRD) equation to express eGFR in mL/min/1.73 m2 of body surface area (14). The abbreviated MDRD study equation is as follows:
Jostel’s TSH index (TSHI) was examined by online SPINA Thyr 3.4.2.548 application (15).
2.3. Terms Definition
Hyperthyroidism was defined as TSH < 0.32 mU/L and FT4 > 19.95 pmol/L, hypothyroidism as TSH > 5.06 mU/L and FT4 < 11.71 pmol/L, subclinical hyperthyroidism as TSH < 0.3 mU/L and normal FT4, and subclinical hypothyroidism as TSH > 5.06 mU/L and normal FT4 (normal FT4, 11.71 pmol/L< FT4 < 19.95 pmol/L) (16). Those with TPOAb+ were men with TPOAb > 32 IU/mL and women with TPOAb > 35 IU/mL®. Jostel's TSHI was calculated as ln TSH (mIU/L) + 0.1345 × fT4 (pmol/L) (17). Finally, eGFR ≥ 60 mL/min/1.73 m2 was defined as not having CKD and eGFR< 60 mL/min/1.73 m2 as having CKD corresponding to stages 3 – 5 of CKD based on the Kidney Disease Outcomes and Quality Initiative guidelines (17). Patients were classified based on their eGFR levels using the national kidney foundation guidelines.
2.4. Statistical Analysis
The data on covariates with normal and skewed distribution are expressed as means (SD) and medians (interquartile range), respectively, and percentages for categorically distributed variables. Com-parison of baseline characteristics between participants was done by student’s t-test for continuous variables, chi-square test for categorical variables, and Mann-Whitney test for skewed variables. A multivariate Logistic regression analysis was used to calculate the odds ratio (OR) of hypothyroid-ism. Statistical significance was indicated with a P-value of less than 0.05.
3. Results
A total of 5,626 individuals (2,371 males and 3,255 females) with a mean age ± SD of 40.6 ± 14.3 years were recruited in this study. Of them 823 (24.4%) with mean Sd age, 54 ± 11.1 years had CKD. No individuals had eGFR< 15%.
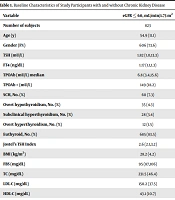
Table 1 shows the study characteristics of participants with and without CKD. Individuals with CKD were older (54.9 versus 38.2 years, P < 0.001) and had a higher prevalence of diabetes (21.3% versus 8.8%, P < 0.001) and higher BMI (28.2 versus 26.3 kg/m2, P < 0.001) than those without CKD. The serum TSH level was higher (1.82 mU/L versus 1.57 mU/L, P = 0.004) and FT4 was lower (1.17 ng/dL versus 1.22 ng/dL, P < 0.001) in CKD cases. The median TPOAb level was higher in the CKD group than in the non-CKD group; all of these differences were statistically significant with trivial clinical significance. A total of 149 (18.2%) and 631 (13.2%) individuals were TPOAb-positive in those with and without CKD, respectively. Jostel’s TSH index was 2.6 (2.1 - 3.2) and 2.5 (2.1 - 3.1) in those with and without CKD, respectively, which was statistically significant but not clinically significant.
| Variable | eGFR ≤ 60, mL/min/1.73 m2 | eGFR > 60, mL/min/1.73 m2 | P-Value b |
|---|---|---|---|
| Number of subjects | 823 | 4803 | |
| Age (y) | 54.9 (11.1) | 38.2 (13.3) | < 0.001 |
| Female, No. (%) | 606 (73.6) | 2649 (55.2) | < 0.001 |
| TSH (mU/L) | 1.82 (1.0,13.3) | 1.57 (0.9,2.5) | 0.004 |
| FT4 (ng/dL) | 1.17 (1.1,1.3) | 1.22 (1.1,1.3) | < 0.001 |
| TPOAb (IU/mL) | 6.8 (3.4,15.6) | 5.6 (3.3,11.6) | < 0.001 |
| TPOAb +, No. (%) | 149 (18.2) | 631 (13.2) | |
| SCH, No. (%) | 60 (7.3) | 251 (5.2) | < 0.001 |
| Overt hypothyroidism, No. (%) | 35 (4.3) | 77 (1.6) | < 0.001 |
| Subclinical hyperthyroidism, No. (%) | 28 (3.4) | 182 (3.8) | < 0.001 |
| Overt hyperthyroidism, No. (%) | 12 (1.5) | 110 (2.3) | < 0.001 |
| Euthyroid, No. (%) | 685 (83.5) | 4169 (87.1) | < 0.001 |
| Jostel’s TSH Index | 2.6 (2.1,3.2) | 2.5 (2.1,3.1) | < 0.001 |
| BMI (kg/m2) | 28.2 (4.2) | 26.3 (4.61) | < 0.001 |
| FBS (mg/dL) | 95 (87,106) | 89 (83,96) | < 0.001 |
| TC (mg/dL) | 231.5 (46.4) | 196.1 (43.1) | < 0.001 |
| LDL-C (mg/dL) | 150.2 (37.5) | 123.6 (34.4) | < 0.001 |
| HDL-C (mg/dL) | 43.1 (10.7) | 41.4 (10.9) | < 0.001 |
| TG (mg/dl) | 171 (122,236) | 129 (88,192) | < 0.001 |
| Smoking (%) c | 47 (5.8) | 592 (12.5) | < 0.001 |
| DM (%) | 170 (21.3) | 388 (8.8) | < 0.001 |
| On diabetic therapy (%) | 642 (7.8) | 115 (2.4) | < 0.001 |
| On LT4 therapy (%) | 24 (2.9) | 111 (2.3) | 0.3 |
| On lipid-lowering therapy (%) | 46 (5.6) | 79 (1.7) | < 0.001 |
Abbreviations: F, female; FT4, free thyroxine; TSH, thyroid stimulating hormone; TPOAb, thyroid peroxidase antibody; SCH, subclinical hypothyroidism; FBS, fasting blood sugar; BMI, body mass index; TC, total cholesterol; HDL-C, high-density lipoprotein-cholesterol; LDL-C, low-density lipoprotein-cholesterol; TG, triglycerides; DM, diabetes mellitus; LT4, levothyroxine.
aValues are presented as mean (SD) or median (Q25, 75) or No. (%).
bP-value < 0.05 is significant.
cSmoking status was categorized into two groups: (1) current smokers (who smoke cigarettes or pipe water daily or occasionally), (2) former smokers (those who used to smoke) or never smokers.
The prevalence of SCH was 7.3% in CKD individuals, which was 5.2% in individuals without CKD (P < 0.001). The prevalence of overt hypothyroidism was 4.3% in CKD individuals, which was higher than in those without CKD (1.6%) (P < 0.001). Subclinical and overt hyperthyroidism prevalence was also compared between the groups, with the CKD group exhibiting a 3.4% prevalence for subclinical and 1.5% for overt hyperthyroidism, while the non-CKD group showed a 3.8% subclinical and 2.3% overt hyperthyroidism. Looking at metabolic markers, the CKD group had higher serum FBS levels, total cholesterol levels, LDL-C levels, TG levels, and HDL-C levels.
The study population was further divided into two subgroups: Subjects with CKD and SCH and subjects with CKD without SCH (Table 2). Although there were no differences in metabolic parameters, e.g., TC, LDL-C, HDL-C, TG, and prevalence of DM between the two groups, more individuals with CKD and SCH were on lipid-lowering therapy (12.1% vs. 5.2%). Besides, TSH was higher and FT4 was lower in those with CKD and SCH than in those with CKD without SCH. Also, TPOAb was present in 56.7% of cases in the CKD and SCH group versus 15.2% in the CKD and no SCH group (P < 0.001). Jostel’s index was higher in those with CKD and SCH than in those with CKD without SCH (3.9 vs. 2.5, P < 0.001).
| Variable | CKD with SCH (N = 60) | CKD with No SCH (N = 763) | P-Valueb |
|---|---|---|---|
| Age (y) | 53.6 (11.4) | 55.1 (11.1) | 0.3 |
| Female, No. (%) | 52 (86.7) | 551 (72.5) | 0.01 |
| TSH (mU/L) | 6.7 (5.6,9.1) | 1.6 (0.9,2.7) | < 0.001 |
| FT4 (ng/dL) | 1.1 (0.9,1.2) | 1.2 (1.1,1.3) | < 0.001 |
| TPOAb (IU/mL) | 62.7 (4.6,275.2) | 6.6 (3.9,13.3) | < 0.001 |
| Positive TPOAb, No. (%) | 34 (56.7) | 115 (15.2) | < 0.001 |
| Jostel’s TSH Index | 3.9 (3.6,4.1) | 2.5 (2.1,3.1) | < 0.001 |
| BMI (kg/m2) | 28.8 (4.2) | 28.2 (4.2) | 0.3 |
| FBS (mg/dL) | 94.5 (85,116.7) | 95 (87,106) | 0.9 |
| TC (mg/dL) | 240.5 (56.3) | 203.5 (45.3) | 0.2 |
| LDL-C (mg/dL) | 157.6 (42.7) | 149.4 (36.9) | 0.1 |
| HDL-C (mg/dL) | 41.7 (9.2) | 43.1 (10.8) | 0.3 |
| TG (mg/dL) | 205 (118.5,282) | 169 (121.3,232.8) | 0.08 |
| Smoking, No. (%) c | 3 (5.2) | 44 (5.8) | 1 |
| Diabetes Mellitus, No. (%) | 16 (28.6) | 153 (20.7) | 0.7 |
| On diabetic therapy, No. (%) | 4 (6.9) | 60 (7.9) | 1 |
| On LT4 therapy, No. (%) | 4 (6.9) | 20 (2.6) | 0.08 |
| On lipid lowering therapy, No. (%) | 7 (12.1) | 39 (5.2) | 0.03 |
Abbreviations: CKD, chronic kidney disease; F, female; FT4, free thyroxine; TSH, thyroid-stimulating hormone; TPOAb, thyroid peroxidase antibody; SCH, subclinical hypothyroidism; FBS, fasting blood sugar; BMI, body mass index; TC, total cholesterol; HDL-C, high-density lipoprotein-cholesterol; LDL-C, low-density lipoprotein-cholesterol; TG, triglycerides; DM, diabetes mellitus; LT4, levothyroxine; N/A, not applicable.
aValues are presented as mean (SD) or median (Q25, 75) or No. (%).
bP-value < 0.05 is significant.
cSmoking status was categorized into two groups: (1) current smokers (who smoke cigarettes or pipe water daily or occasionally), (2) former smokers (those who used to smoke) or never smokers.
The risk of SCH was higher in the CKD group than in the non-CKD group after adjusting for age, sex, smoking, TPOAb positivity, and BMI (OR 1.28, 95% CI 0.89 - 1.83) but not significantly (Table 3). Looking at the effect of SCH on cardiovascular outcomes in the CKD population, we found 29 cases of first CVD event in those with SCH and 579 cases in those without. In terms of CVD and all-cause mortality, we had nine cases in the SCH group but found 200 cases in those without SCH. Due to the small number of events in the CKD with SCH group, we were unable to determine the effect of SCH on the outcome in our CKD population.
| Odds Ratio | 95% CI | P Value | |
|---|---|---|---|
| eGFR > 60 (mL/min/1.73 m2) | 1.28 | 0.89 - 1.83 | 0.1 |
| Age | 0.99 | 0.98 - 1.00 | 0.04 |
| Sex (male) | 1.81 | 1.34 - 2.44 | 0.001 |
| Smoking | 0.57 | 0.33 - 0.99 | 0.04 |
| TPOAb | 7.62 | 5.96 - 9.75 | 0.001 |
| BMI | 0.98 | 0.96 - 1.01 | 0.2 |
a Reference groups are e-GFR < 60, males, and nonsmokers.
4. Discussion
Comparison of the CKD and non-CKD groups revealed a statistically significantly higher prevalence of hypothyroidism in the CKD group, as well as TPOAb positivity; however, after adjusting for confounding factors, the significant differences no longer remained. The CKD group was also older and consisted of subjects with higher BMI, FBS, and lipid levels, as well as a higher prevalence of diabetes. Within the CKD group, comparing those with SCH and those without SCH showed no differences in metabolic parameters, except for higher TSHI and using lipid-lowering drugs.
The rapid increase in the prevalence of CKD worldwide, as well as its high treatment costs, makes CKD an increasingly important public health concern. As known, 13.1% of US adults are affected by CKD (18), and although the prevalence rates vary greatly across the globe, from 3.0 to 21.8% (1), they are all similarly on the rise. Besides, CKD is disproportionately more prevalent in developing nations, as its main risk factors, i.e., diabetes (19), hypertension (20), obesity (21), population growth and increasing age (22, 23), are increasing in these nations. The latest reported age-adjusted prevalence of CKD in Iran is 14.9% and the overall prevalence is 15.1% (24, 25), making CKD an even greater concern than before. In this regard, SCH is a purely biochemical diagnosis that has been associated with a multitude of clinical manifestations including abnormal lipid metabolism (26), cardiac dysfunction (27), and increased risk of atherosclerosis and coronary heart disease (28), all of which are conditions associated with CKD and its poor outcome.
Looking at thyroid function tests within this population, subjects with normal eGFR were more likely to be euthyroid than those classified as having CKD, but we did not find a higher prevalence of overt and subclinical hypothyroidism in the CKD group after adjustment for confounders. There are few studies that have examined the prevalence of hypothyroidism among CKD patients not requiring dialysis. The findings of the current study are in contrast with some studies. Lo et al. examined the NHANSE data to show a steady rise in the prevalence of hypothyroidism (overt and subclinical) with declining GFR (29, 30). One in five CKD persons with eGFR < 60 mL/min/1.73 m2 had evidence of hypothyroidism. The study showed an increased prevalence of hypothyroidism with reduced eGFR, which was independent of gender, age, and ethnicity but TPOAb titer or positivity was not considered a confounder to be adjusted for. Chonchol et al. reported a 17.9% prevalence of SCH after adjustment for age, gender, and metabolic profile (FPG, total cholesterol, and TG) in their cross-sectional observational study, compared to a 7% prevalence in subjects with eGFR ≥ 90 ml/min/1.73 m2. The findings of the study also showed increase in risk of SCH with decreasing level of eGFR. (31). Similarly, this study did not consider TPOAb as a confounder and the results might change after adjustment for TPOA positivity. We also calculated Jostel’s TSH Index (TSHI), which showed that the higher TSHI level in the CKD group was not due to TSH deficiency in all subgroups that were studied; this suggests that there is a reduced response to TSH within the functional tissue of the thyroid gland.
The main concern in these subjects is the concurrence of two conditions with known morbid effects on the cardiovascular system: SCH and CKD. As shown, SCH has been associated with CVD risk factors such as older age (32), high total cholesterol (TC), LDL-C (33, 34), hypertension (35), and elevated CRP (35), as well as diastolic dysfunction (36), ischemic heart disease (37), and congestive heart failure (38). Clinical trials have also shown that hormone replacement therapy in SCH subjects reduces CV risk factors (39, 40) and improves myocardial structure and contractility (41). We observed 12 cases in the CKD group with SCH (among 60 cases) that developed their first CVD event during the three-year follow-up period. Although this seems to be slightly greater than the percentage of subjects that had the first CVD event in the CKD without SCH group (12.6% versus 11.3%), the numbers are still too small to enable us to make an informed comparison of CVD outcome between these two groups.
The current study has some limitations. First, our definition of kidney function was based on estimated GFR and calculated using serum creatinine values, where the ideal would be to specifically measure clearance. Second, serum creatinine values have been a matter of debate recently, with many nephrologists seeking a standardized creatinine measurement. While this matter has largely been resolved in many countries, this study was unable to ascertain with certainty that serum creatinine was measured using an internationally standardized kit. However, all the creatinine samples were measured using the same kit (Pars Azmoon), which is one of the most commonly used assays for creatinine across Iran. Lastly, as mentioned above, the present study was unable to look at the effect of SCH in CKD on CVD and overall outcome, due to the small number of events in this relatively small subgroup of the TTS cohort. As the number of years of follow-up increases, we hope to be able to better study these effects.
4.1. Conclusions
The prevalence of SCH was comparable between subjects with CKD and those without in this urban Middle-Eastern cohort. In the present study population, the concurrence of CKD and SCH with female gender and older age may explain potentially worse outcomes; however, we do not currently have sufficient prospective follow-up data on CVD outcomes and mortality to explore this further.