1. Context
The endocrine system plays a key role in the rhythmicity of the menstrual cycle setting (1, 2). Menstrual disorders are one of the most common gynecological complaints of women (3), a common problem that continues from menarche to menopause (4). As known, the menstrual cycle includes the ovarian and the endometrial cycles (5) in which the cyclic response to the hormone production from the hypothalamus, pituitary, and ovaries (HPO) axis leads to the occurrence of menstrual cycle (6). Failure to conceive in a menstrual cycle is followed by the shedding of the endometrium (5). In different stages of life, women sometimes experience various changes in their menstrual cycle (7).
Data available on the subject of women’s reproductive health show menstrual disorders to be a fundamental problem (8, 9). The prevalence of irregular menstrual cycles in women varies from 14.2% to 27% in different regions of the world (10, 11).
Abnormal uterine bleeding in reproductive age women defined as an irregularity in frequency, the volume of flow, duration, and regularity of menstrual cycle in the absence of pregnancy (12). According to the International Federation of Gynecology and Obstetrics (FIGO), structural and non-structural causes lead to abnormal uterine bleeding, the non-structural causes include ovulatory, coagulopathy, endometrial, iatrogenic factors and not otherwise classified (13).
In the reproductive axis, gonadotropin-releasing hormone (GnRH) pulse secretion has a key functional role, and disrupting GnRH activities is associated with endocrine disorders (14). It is well known that in chronic disorders, any changes in ovarian steroid secretion results in menstrual abnormality (15). The menstrual cycle pattern also reflects the endogenous endocrine health environment (2), and irregular menstrual cycles may be a marker of chronic conditions such as diabetes, and polycystic ovary syndrome (PCOS) (16, 17). Menstrual cycle irregularity can impair women’s health status and different aspects of their social life and leads to an increase in the economic burden of health care (18).
Currently, although the same studies focus on a menstrual abnormality in different endocrine disorders, there is no comprehensive study that has searched and summarized these studies. This study aimed to review and summarize the features of menstrual disturbances in some endocrine diseases.
2. Evidence Acquisition
A comprehensive literature search was conducted in Google Scholar, PubMed [including Medline], Web of Science, and Scopus databases for retrieving studies published in English language up to December 2019 on menstrual disturbances and endocrine disorders. Further, a manual search in the references list of the included studies and other relevant reviews were used to maximize the identification of eligible studies. Different types of studies (review, observational, experimental) relevant to the subjects were reviewed.
The following terms, alone or in combination, were used as keywords for the search: “Polycystic Ovary Syndrome” OR “PCOS” OR ”Cushing’s syndrome” OR “Hypercortisolism” OR “Hypothyroidism” OR “Hyperparathyroidism” OR “Thyrotoxicosis” OR “Hyperthyroidism” OR ”Hyperprolactinemia” OR “Prolactin Hypersecretion Syndrome” AND “Menstrual Cycle” OR “Uterine Hemorrhage” OR “Uterine Bleeding” OR ”Menstruation” OR “Menstruation Disorder” OR “Irregular Menstruation” OR “Polymenorrhea” OR “Hypomenorrhea” OR ”Retrograde Menstruation” OR “Menstruation Disturbance” OR “Menorrhagia” OR “Heavy Menstrual Bleeding” OR “Oligomenorrhea” OR “Amenorrhea”.
3. Results
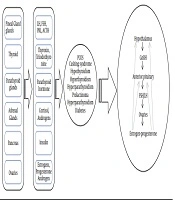
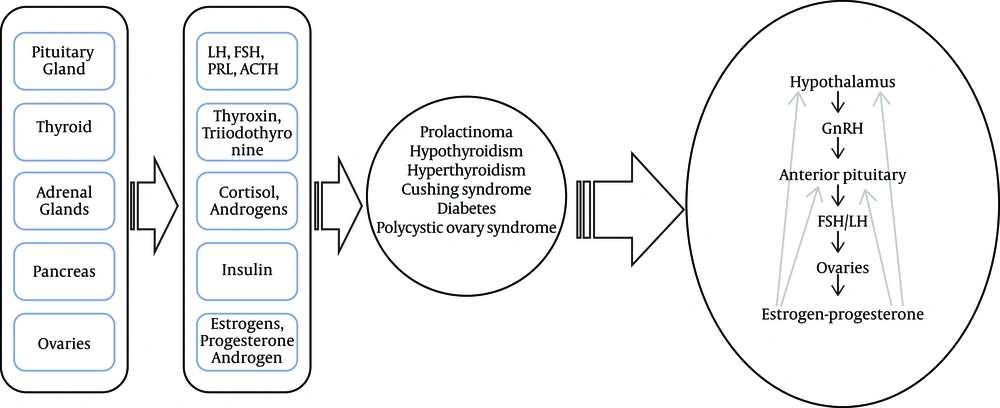
In this review, the results were presented as six subheadings, including polycystic ovary syndrome, hyperprolactinemia, thyroid disorders, diabetes, Cushing’s syndrome, and other endocrine disorders. Table 1 summarizes the hormonal changes and results of menstrual disturbances that were observed in various endocrine disorders. Figure 1 shows the schematic relationship between the endocrine disorders and the hypothalamic-pituitary-gonadal axis.
| Type of disorder/ref | Possible Mechanism | Type of Menstrual Disturbance |
|---|---|---|
| PCOS (19, 20) | Increased LH and androgen secretion, prolonged exposure to estrogen | Long and irregular menstrual cycles, secondary amenorrhea, oligomenorrhea |
| Diabetes (21-23) | Increased LH/FSH ration and androgens, decreased gonadotrophin levels, hyperinsulinemia | Oligomenorrhea, long cycles, long and heavy menstruation |
| Hyperprolactinemia (24, 25) | Inhibition of LH GnRH and maybe FSH | Amenorrhea, oligomenorrhea, bleeding between cycles, polymenorrhea, hypermenorrhea and menometrorrhagia. |
| Hypothyroidism (26, 27) | Deceased SHBG, estradiol (E2), testosterone levels | Irregular menses, heavy bleeding, oligomenorrhea, amenorrhea, breakthrough bleeding |
| Thyrotoxicosis (27) | Increased estradiol, testosterone levels and androgen conversion to estrone | Oligomenorrhea, amenorrhea |
| Cushing’s syndrome (28, 29) | Ovarian damage, androgens, hypercortisolism | Amenorrhea, oligomenorrhea, polymenorrhea |
Different factors lead to variation in the secretion of hormones that influence the menstrual cycles. It has been shown that the menstrual cycle is highly varied by genetic, aging, lifestyle, environmental, and socioeconomic factors (30, 31). For instance, by each one year increase in age (from 25 - 45 years), the mean cycle length is decreased by 0.19 days. Moreover, obese women with body mass index (BMI) higher than 35 experience 14% higher mean variation of the menstrual cycle (32). Psychological factors may affect the menstrual cycle per se or through various medications (33). Among various disorders, endocrine disorders highly influence the menstrual cycle properties through several hormonal pathways. Sometimes, the first manifestation of endocrine disorders may be menstrual disturbances. Here, we summarized the effect of the most common endocriopathy on menstrual cycles.
3.1. Menstrual Cycle Disturbances in Women with Polycystic Ovary Syndrome (PCOS)
Irregular bleeding with ovulatory dysfunction in women at childbearing age is commonly caused by PCOS (34). Recently, PCOS has been recognized as the most common problem in endocrine practice (35). It should be noted that PCOS symptoms include numerous small cysts with enlarged ovaries, irregular menstrual cycles, hirsutism, alopecia, and acne (36, 37); one of the clinical manifestations of PCOS is an abnormal menstrual cycle (from amenorrhea to menorrhagia) (38). Strowitzki et al. (39) investigated 118 German women with PCOS and showed that the degree of menstrual cycle irregularity indicated the degree of the endocrine disorder in these patients. Maslyanskaya et al. (40) reported that PCOS is an under-recognized cause of abnormal uterine bleeding (AUB) in teenagers admitted to health care institution. In the Tehrani et al. (37) study conducted on Iranian women, the subclinical menstrual dysfunction was diagnosed among 11.3% of women with hirsutism (26).
In the Balen et al. (41) study, 1,741 women with PCOS were investigated, approximately 30% of whom had a regular menstrual cycle, nearly 50% had oligomenorrhoea, and nearly 20% were amenorrhoeic; consequently, the majority of women with PCOS had abnormal menstrual cycles, and the most frequent pattern was infrequent menstruation associated with anovulation. Owing to the prolonged exposure to estrogen and unaffected by the inhibitory effects of progesterone, the mean thickness of the endometrium was raised in women with PCOS (42, 43); thus, oligoovulation or anovulation is the well-known clinical changes in the menstrual cycle of PCOS patients (44).
A study by Elting et al. (45) showed that in the ovarian aging process, women with PCOS experience regular menstrual cycles. The most recent recommendation for the management of PCOS is behavioral, dietary, and exercise interventions (lifestyle), combined oral contraceptive pills (COCPs), anti-androgen pharmacological agents, and metformin (43). According to evidence, the effect of lifestyle interventions on the menstrual cycle of PCOS women is unknown (46). Data from a randomized trial study conducted on 68 obese PCOS women suggested that behavioral modifications can improve menstrual regularity in PCOS women (47). Teede et al. (48), in a meta-analyses study, reported that for the regulation of the menstrual cycle in women with PCOS, combined oral contraceptive therapy was more effective than the metformin therapy (P = 0.03); however, abnormal menstrual bleeding is also one of the side effects of oral contraceptive use (49). The literature reports that long-term (more than 6 months) treatment with metformin can increase the rates of achieving normal menstrual cycles in women with PCOS (50, 51). Metrorrhagia is one of the side effects of spironolactone, an anti-androgen agent, which is a prescribed medication for women with PCOS (52).
3.2. Menstrual Cycle Disturbances in Women with Diabetes
In 2017, the International Diabetes Federation atlas showed that 219.3 million women live with diabetes (53). Nowadays, diabetes is a global health problem (54). According to the current evidence, adolescent and adult females with diabetes may suffer from menstrual cycle irregularity (55, 56). Also, evidence shows the casual relationship between variations in the levels of sex hormones and the risk of type 2 diabetes mellitus (57, 58). The results of the Nurses’ Health study II indicate that women with menstrual cycle length ≥ 40 days are more susceptible to develop diabetes mellitus type 2 (16). Griffin et al. reported that nearly 25% - 30% of women at childbearing age with diabetes suffer from menstrual abnormalities (59). Strotmeyer et al. (21) had assessed menstrual cycle differences in 143 women with diabetes, 186 of their sisters without diabetes, and 156 women controls, and the results showed that women with type 1 diabetes experience some menstrual problems such as heavy menstruation, menstrual irregularity, menstruation lasting longer than 6 days, and cycle length over 31 days more than that were observed in their sisters or controls. The most common menstrual abnormalities among women with diabetes are secondary amenorrhea and oligomenorrhea (60).
In summary, there is little evidence about the natural history of menstruation among women with diabetes during the reproductive lifespan (21). Also, the literature on the effect of diabetes treatments on the menstrual function among women with diabetes is limited; the luteinizing hormone (LH) correlates with serum levels of glucose, and in good serum glucose control results in optimum menstrual function (61).
3.3. Menstrual Cycle Disturbances in Women with Hyperprolactinemia
Another disorder related to menstrual problems is hyperprolactinemia (62). Hyperprolactinemia may occur due to physiological adaptation, or it may be pathological or induced by pharmacological medication (63). In a normal menstrual cycle, prolactin level decreases in the follicular phase and rises in luteal phases (64). Prolactin also stimulates progesterone synthesis in follicles granulosa cells (64). Prolactin-releasing factors include thyrotropin-releasing hormone (TRH), serotonin, opioids, and other neuropeptides and neurotransmitters (64). The mean prevalence of prolactinemia in women is 30 per 100,000, the peak prevalence of which is seen between 25 to 34 years of life (65). The link between hyperprolactinemia and menstrual irregularity is well established (24). Prolactin hormones are produced by pituitary lactotrophs and play a role in neuroendocrine regulation. Increased production of prolactin impairs women’s reproduction function (66). Hyperprolactinemia is one of the causes of amenorrhea (absence of menstrual bleeding for three consecutive cycles) (67), and the mechanism suggested for amenorrhea is the suppression of the pulsatile GnRH (68). Also, increasing the level of prolactin can suppress the secretion of luteinizing hormone (LH) and inhibit ovulation in the menstrual cycle (69).
Different patterns of menstrual disorders such as oligomenorrhea, bleeding between cycles, polymenorrhea, hypermenorrhea, and menometrorrhagia have been seen in women with hyperprolactinemia (25). The first line of treatment for hyperprolactinemia is the use of dopamine antagonist (DA) medications, and it has been shown that Cabergoline has been more efficacious than bromocriptine (70, 71). In most patients (approximately 80%), DA induced a normal menstrual cycle with ovulation (72). Rasmussen et al. (73) reported that bromocriptine had no adverse effect on pregnancy outcomes of those women with hyperprolactinemic whose prolactin level and menstruation were normal (13 to 108 months after breastfeeding period). Kulshreshtha et al. (24) conducted a study to investigate the menstrual cycle in patients with hyperprolactinemia; their results showed that restoration of the regular menstrual cycle takes three to five months and they found no relationship of time to onset of menstrual cycles regularity with menstrual cycle length, duration of menstrual irregularity, or initial prolactin level in patients with prolactinomia.
3.4. Menstrual Cycle Disturbances in Women with Thyroid Disease
There are global variations in the incidence and prevalence of thyroid disorders (74) that the incidence of them in women is higher than in men (75, 76). Thyroid dysfunction, such as hyperthyroidism and hypothyroidism affect the reproductive health of women (77). The hypothalamic-pituitary-thyroid axis and the hypothalamic-pituitary-gonadal axis work together, and any dysfunction in the thyroid can affect the serum sex steroid levels, sex hormone-binding globulin (SHBG), gonadotropin-releasing hormone (GnRH), and prolactin (26). Thyroid dysfunction can affect the normal menstrual cycle, sometimes presenting as AUB (78). Changes in menstrual cycle length and amount of bleeding often occur in women with hypothyroidism (27). In general, hypothyroidism causes different menstrual conditions such as irregular menses, heavy bleeding, oligomenorrhea, amenorrhea, breakthrough bleeding (26). Kakuno et al. (79) report a higher prevalence of menstrual disturbance only in severe thyroid disease. The most common menstrual manifestations in hyperthyroidism and hypothyroidism are oligomenorrhea and polymenorrhea, respectively (80).
The levothyroxine (LT4) therapy is the standard treatment option for hypothyroidism (81). Jamilian et al. (82) reported that in euthyroid women, menstrual irregularity is improved by taking levothyroxine. Levothyroxine therapy may lead to elevated levels of TSH in mid-cycle (83). The average dose of radioiodine (I-131), R-I131, which is used for treating hyperthyroidism therapy is lower than cancer treatment, indicating that its gonadal side effects are not significant (84).
3.5. Menstrual Cycle Disturbances in Cushing’s Syndrome
Cushing’s syndrome (CS) is a rare endocrine disorder (85), with an incidence rate of estimated to be 1 - 3 per million (86). Excess production of glucocorticoids may result in Cushing’s syndrome (87). The related clinical signs of hypercortisolism in CS include obesity, hirsutism, hypertension, protein wasting signs, menstrual irregularity (oligoamenorrhea, amenorrhea). Menstrual irregularity is the most common complaint in Cushing’s syndrome (28). Clinical and biochemical signs of hyperandrogensim are more commonly manifested in women with CS (88). One of the most common clinical features of CS in female patients is menstrual irregularity (80%) (89). In the European Registry on CS, in 390 female patients, 56% had menstrual irregularity (90). Similarly, Bolland et al. (91) observed that 35.5% of women with CS in a New Zealand nationwide survey, presented with menstrual irregularity. Lado-Abeal et al. (28) found that in 45 female patients with CS, only 20% of them had a normal menstrual cycle, while 31.1%, 33.3%, and 8.8% experienced oligomenorrhea, amenorrhea, and polymenorrhea, respectively. Overall in this study, 80% of the participants had menstrual irregularity, a manifestation most closely related to the level of serum cortisol rather than androgen level. The clinical management of this disorder includes steroidogenesis inhibitors, glucocorticoid receptor antagonist, ACTH-lowering agents, and radiation techniques, and bilateral adrenalectomy (86). The literature on CS treatment and improving menstrual cycle irregularity problems is scarce.
3.6. Other Endocrine Causes of Menstrual Disturbances
Other endocrinopathies may affect the menstrual cycles. The metabolic bone diseases (MBD) like primary hyperparathyroidism (PHPT) is another endocrine disorder, which is defined as inappropriately plasma levels of parathyroid hormone (PTH) and is more frequently diagnosed in women aged > 50 years (92); as a result, there are no sufficient data regarding the relationship between this disorder and menstrual cycle during reproductive age. Nevertheless, it should be noted that PTH levels in normal women do not vary despite the fluctuation of estrogen (93).
Non-classical congenital adrenal hyperplasia (NCCAH) is another disorder that may influence the menstrual cycle; the most common manifestations of menstrual disorder in these patients are primary or secondary amenorrhea and oligomenorrhea (94).
Acromegaly is a rare disease with possible manifestation on menstrual cycle due to the higher levels of prolactin and estrogen deficiency. Kaltsas et al. (95) study showed that 62% of patients with acromegaly experienced amenorrhea, 15% had oligomenorrhea and 4% had polymenorrhea. Data from the multi-center acromegaly registry reported that hypogonadism was observed in more than half of women with acromegaly (96). Also, PCOS is reported as a frequent manifestation of women with active acromegaly (97).
Addison’s disease is a rare adrenal disorder characterized by low secretion of adrenocortical hormones (98). Female patients with Addison’s disease may experience menstrual irregularity, but there is no sufficient data regarding the type of menstrual disturbance that is mostly observed in this rare disorder (99).
4. Conclusions
Endocrine glands (pituitary, thyroid, pancreas, adrenal, and ovaries) play a functional role in the endocrine regulation of a women’s menstrual cycle. As a result, endocrine disorders are the triggers of onset of menstrual disturbance across the reproductive lifespan of women. Oligomenorrhea is the most common menstrual disturbance in endocrine disorders. Further studies are highly needed for better clarification of the underlying pathways of the association between endocrine disorders and the menstrual cycle.