1. Context
Dietary clinical trials (DCTs) have successfully been expanded over the three last decades to determine dietary requirements and supply strong evidence for the development of dietary guidelines (1). The DCTs, defined as “controlled modifications in dietary intakes or habits in humans to evaluate their potential effects on selected outcomes”, determine causal relationships and provide strong evidence for real clinical practice (2, 3).
Since observational study designs in the field of nutrition science could not reach a consensus on optimal nutrition and provide reliable evidence for years, well-designed DCTs should be performed to compensate current back warded state of nutrition science (4, 5). Several global dietary guidelines and successful population-based strategies [e.g., iodized-salt programs for iodine deficiencies (6), or folic acid fortifications to prevent neural tube defects (7)] have been derived from the DCTs. Furthermore, evidence-based medicine (EBM) has been used DCTs to develop public strategies and guidelines (8).
In contrast to pharmaceutical trials, the process of designing DCTs is not straightforward and not established (9, 10). DCTs encounter critical challenges in terms of study design and methodology, tinny effect sizes, high heterogeneity of the responses, limited translatability of the observed effect size (11). Some practical challenges of the translatability of the DCT findings can be limited due to the inadequacy of the outcome measures, intervention duration to high dropout rate, and contrast between study groups. Other contributing factors include low adherence and high variability between the circumstances (12). Furthermore, DCTs are suffering from the lack of good reporting of the study design, implementation and results that makes DCTs mostly impractical to translate into public health and clinical practice.
Here, we aimed to discuss the limitations of different types of study designs for DCTs, and to provide some practical solutions to reduce the limitations. Furthermore, we discussed how different natures of nutrition interventions vs. pharmaceutical interventions make DCTs difficult to translate into public health and evidence-based practices. The review could be a practical guide for researchers in the field of healthcare and gives new hints in dietary clinical trials.
2. Evidence Acquisition
2.1. Limitations of Dietary Clinical Trials for Translation Into Practice
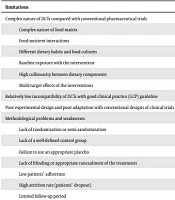
Many aspects of pharmaceutical and dietary trials are common. DCTs investigating the effects of a single nutrient (e.g., vitamin C) or bioactive food compounds (e.g., sulforaphane) or even a medical food formulation (e.g., medium-chain triglycerides; MCTs) are more similar in design and conduct to drug intervention trials. However, a huge number of DCTs evaluating the benefits of foods (e.g., fruits, grains) and dietary patterns (e.g., Mediterranean diet) or change of dietary behaviors (e.g., decrease fast food eating) have fundamental differences with conventional clinical trials. In other words, the adaptation of DCTs with the good clinical practice (GCP), as proposed by the International Council for Harmonization of Technical Requirements for Pharmaceuticals for Human Use (ICH), is complicated and challenging (13). Box 1 provides a summary of common limitations of DCTs for translation into clinical practice.
| limitations |
|---|
| Complex nature of DCTs compared with conventional pharmaceutical trials |
| Complex nature of food matrix |
| Food-nutrient interactions |
| Different dietary habits and food cultures |
| Baseline exposure with the intervention |
| High collinearity between dietary components |
| Multi-target effects of the interventions |
| Relatively low incompatibility of DCTs with good clinical practice guideline |
| Poor experimental design and poor adaptation with conventional designs of clinical trials |
| Methodological problems and weaknesses |
| Lack of randomization or semi-randomization |
| Lack of a well-defined control group |
| Failure to use an appropriate placebo |
| Lack of blinding or appropriate concealment of the treatments |
| Low patients’ adherence |
| High attrition rate (patients’ dropout) |
| Limited follow-up period |
| Insufficient sample size |
| Poor definition of outcome |
| Poor reporting of the trials |
| Lack of clear statement of the study hypothesis |
| Lack of reporting study population characteristics, inclusion and exclusion criteria |
| Lack of reporting of study design |
| Inadequate statistical method section |
| Limited translatability of the observed effect size due to high heterogeneity of study population |
| High susceptibility to confounding variables (e.g., ethnicity, genotype, and physiological state e.g., pregnancy or lactation, and sub-clinical nutritional deficiencies) |
DCTs are more susceptible to confounding variables and design difficulties compared to pharmaceutical trials (2, 14). On the other hand, the magnitude of the treatment effects raised by the DCTs are tended to be small for most clinical outcomes, and patient’s adherence, and dropout rate may be unpredictable (16, 17). Several diet-related factors, including the nature of the habitual diet that may change the chemical form of the nutrient and food matrix, interactions between nutrients and food additives (e.g., aspartame, benzoic acid, sodium benzoate, monosodium glutamate), food processing methods (cooking vs. frying), and intestinal and systemic factors markedly affect the absorption and bioavailability of food ingredients, can limit the translatability of the observed effect size (18). Other factors like ethnicity, genotype, and physiological state (e.g., pregnancy or lactation), and sub-clinical nutritional deficiencies can also confound the treatment effect of intervention trials. DCTs have also been criticized for some inaccurate treatment effects (e.g., over-estimated effect size), caused by selection bias (e.g., selection of high-risk populations rather representative of the target population), invalid control experiment, semi-randomization rather than full-randomization, lack of a well-formulated placebo, and unblinding of either patients or researchers that may badly affect the clinical outcomes (17). For example, a randomized controlled trial that investigated the effects of vitamin K2 supplementation on glucose homeostasis reported that patients with type 2 diabetes mellitus (T2DM) who received 180 µg MK-7 twice daily, had significantly lower fasting plasma glucose (FPG) and hemoglobin A1C (HbA1C) after 12 weeks of trial, compared to control group. Effect sizes for FPG and HbA1C were -0.68 mmol/L and -0.36%, respectively (19). Similarly, a meta-analysis of pharmaceutical trials to evaluate the efficacy of dapagliflozin in patients with T2DM reported that the overall effect sizes of HbA1C and FPG were -0.52% and -1.13 mmol/L, respectively (P-value < 0.001 for both) (20). In contrast, in a dietary clinical trial that investigated the effects of beetroot powder (5 g/d for 24 weeks) on FPG and HbA1C in patients with T2DM, no significant difference was observed between intervention and control groups for glycemic parameters (21).
The inadequacy of the outcome measures and insufficient intervention duration to high dropout rate, low adherence, the variability of circumstances, or insufficient contrast between study groups also limit the translatability of the findings of DCTs (12).
To sum up, some believe that since nutrition interventions contain several interacting components, difficulties of delivering or receiving the interventions, and high variability of the measured outcomes, DCTs may need to be considered complex interventions and adopt appropriate methods as well.
2.2. Common Challenges of Dietary Clinical Trials
2.2.1. Complex Nature of Nutrition Interventions
The pharmacological clinical trials are based on the assessment of pharmaceutical active ingredients or the placebo. However, DCTs interventions most likely consider complex ingredients, extracts or multi-ingredient food products, or even full or partial dietary modifications, rather than well-defined single molecular compounds (3). Drugs are isolated chemical compounds, with well-known physical characteristics and pharmacokinetic dynamics that target a particular pathway or influence specific pathologic conditions (14). Nutrition, in contrast, has a ubiquitous nature, with a heterogeneous mixture of nutrients and bioactive components, which may exhibit several off-target properties in the body (8, 14). Although food supplements (e.g., vitamin C, β-carotene) are isolated substances and classified as drugs (22), whole diet is categorized as “complex interventions” (10), mainly due to the multifaceted nature of interventions (2, 23). High collinearity between foods, nutrients, and bioactive components, or even synergistic and antagonistic properties of nutraceuticals, can obscure the relationship between food and their effects on the outcome of interest (14). Furthermore, dietary components may exert different functional effects, which cannot be captured; therefore, may result in misleading (22). Keeping the balance between micronutrients and bioactive compounds across the intervention and control groups is also difficult due to different nutrients composition of foods (24). Different methods of food processing, cooking, and food storage conditions can also change the results (24). Lack of consistency between food products due to their dependency on genotype of foods, condition of growth and harvesting, transport and storage, makes more complexity in terms of nutrition interventions (25).
Diversity of food cultures and behaviors can also alter the true effectiveness of an intervention (2, 14). Owing to these diversities, DCTs are subjected to a large inter- and intra-individual variability in response to the same dietary intervention. Taken together, comprehensive knowledge regarding the food matrixes, composition of foods and their synergistic-antagonistic effects, is critical to achieving a better understanding of food and its physiological effects.
2.2.2. Baseline Exposure and Baseline Dietary Status
One of the major challenges of DCTs is a dietary background and baseline exposure to the food being investigated (24). Lack of enough information about the background dietary intake and baseline status of the participants limits the generalizability of findings (24). In case of nutrient supplementation, baseline dietary status (e.g., nutrient deficiency vs. adequacy) will affect the effectiveness of the intervention (24). In addition, background intake of the food component may affect the outcome in the control group, especially when the supplement is being used in a greater amount available from foods rather than in pharmacological quantities (14). The importance of baseline nutritional status (malnourished vs. normal-nourished) of the study participants will be more highlighted in NCTs investigating the treatment effect of the intervention on hard-clinical endpoints and survival, especially in critically ill patients (26, 27).
Altogether, interventions that aim to change the existing diet are required to assess initial dietary intakes for both screenings the participants in case of inclusion and exclusion criteria and baseline measurement against which dietary changes resulting from the intervention (28). Although different methods (29) are available to assess baseline dietary exposure and dietary status, it is difficult to accurately determine because of drawbacks of the assessment methodologies, incomplete nutrient databases, change of nutrients composition of foods, and temporal changes in exposure (8). Baseline dietary status can be assessed using common methods, including weighed food record (e.g., 7-day food record as the gold standard), estimated food record, diet history, 24-hour recall, food frequency questionnaires (FFQs), or digital assessment methods, internal nutrient databases and image analysis method to automatically estimate intakes (29). Chemical assessments of the nutrients or bioactive food components in biological samples can also be applied to determine previous exposure with the intervention or dietary status of the patients in case of the nutrient (30); these methods are more valid and reliable, based on the objective assessing methods and they are free of self-reported biases (30). The most common nutritional biomarkers used in the field of nutrition research are plasma carotenoids, vitamin C and ferric reducing antioxidant power (for assessment of fruit and vegetable intakes) (31), serum creatine (for assessment of meat and fish intakes) (32), urinary or plasma daidzein or genistein (for assessment of legumes intakes and soy-products) (33), plasma docosahexaenoic acid (DHA), erythrocyte DHA, and platelet DHA (for assessment of dietary intakes of w3-fatty acids) (34).
Other methods, e.g. randomization (either group, block or stratified randomization), conducting sub-group analysis according to the baseline status of intakes, statistical adjustment based on the baseline status are recommended to minimize confounding effects of previous exposure or baseline dietary status (8, 35).
Considering potential effect of baseline dietary status prior to study design is preferred rather the use of post-intervention-adjusting methods; e.g., a good suggestion is stratified randomization, within which a separate block of dietary status (e.g. high vs. low intakes) is defined and the participants will be assigned to the appropriate block. Subsequent to the block assignment, simple randomization within each block will categorize the subjects into either the intervention or the control group (35). This method, however, requires a greater sample size than simple randomization (36).
The use of statistical methods for adjustment of the baseline dietary status is appropriate for small sample sizes, including less than 500 participants. Most common statistical methods used to adjust this issue are analysis of covariance (ANCOVA), multivariate ANCOVA, or both (35). An important limitation of such methods, which makes complicated the interpretation of the finding, is that imbalanced covariates may result in unanticipated interaction effects, such as unequal slopes among subgroups of covariates (37). Failure to adjust or lacking data on baseline nutritional variables (especially those related to the treatment) may lead to over- or under-estimation of the effect size (14).
2.2.3. Randomization and Blinding
Randomization is the principal component of a well-designed clinical trial. It is commonly described as the systematic tendency of factors related to the design, conduction, analysis, and findings’ interpretation of a study, in treatment assignments. Randomization provides researchers with the opportunity to perform comparable groups (35); the use of probability theory requires the likelihood of chance, as a source for the measurement of difference in final outcome, to be expressed (35). Appropriate randomization reduces the likelihood of bias from inaccurate effect size (i.e., over- or under-estimated treatment effects) (38).
Although randomization can effectively capture allocation bias, it also counteracts with preference effects (e.g., for the participants who are assigned an intervention that is other than their favorite), and can decrease potential generalizability of the study results (5) because dietary interventions are not randomly assigned in real life. The preferences of the participants, as possibly related to study outcomes, might influence the estimated effect size. In other words, not only the effect of preferences can be substantial but it may also appear larger and more significant than the direct treatment effect (39).
In case of lifestyle intervention studies like DCTs, where intervention may be affected by subject’s presences, the use of randomized preference designs can partially overcome this problem (39). Successful and complete blinding, as blinding of participants, caregivers and outcome assessors about the intervention, is usually difficult to achieve and not feasible in lifestyle interventions (40). Blinding of treatment in DRCTs cannot always be achieved because interventions for dietary habits and nutritional counseling cannot be blinded (15).
2.2.4. Placebo
A placebo is defined as an intervention applied in a control group to mimic all other aspects of the main treatment except its bioactive compound; comparing the main treatment and placebo groups allows the identification of the components responsible for the physiological alteration made in the treatment (41, 42). Although the use of placebo controls is relatively straightforward when intervention of interest is a nutrient or nutraceutical compound, applying an appropriate placebo-controlled group in DCTs is a major obstacle when foods are considered interventions (43). Lack of ability to adequate masking of intervention or inappropriate choice of placebo food or diet still remains the major challenging issues in DCTs (14, 43, 44). Besides the components of foods, the tastes, colors, physical textures, and labels of the food may exert placebo-like effects, and therefore, a placebo should be analogous for all aspects of the intervention (25).
Several attempts have been, therefore, made to develop appropriate placebo; newly developed placebos have been ranged from simple to more complicated ones. For example, a formulation was developed for Aronia juice (as a polyphenol-rich food) by mixing various nutrients, artificial colors and flavors with water; this placebo provided physicochemical properties (i.e., taste, color, smell, texture, and nutrient composition) as same as the treatment, while it lacked polyphenols (42). Another good example of food placebo is nitrate-depleted beetroot juice, first developed by Gilchrist et al., to assess the effect of nitrate of beetroot juice on reaction time; this placebo was as same as beetroot juice for all physicochemical properties, but its nitrate content was ~ 0.002 mmol/250 mL compared to original beetroot juice contained 7.5 mmol/250 mL (45). The use of placebo term in DCTs, evaluating change of dietary habits or patterns, is not as common as food and nutrient interventions, however, social scientists have provided some important placebo-related insights on dietary habits, food choice, and nutrition (25). In these cases, different terminology such as belief, expectancy, conditioning and context are considered as equal as placebo in pharmaceutical or food-nutrient clinical trials (25); different complicated placebo-like models have been developed for these complex interventions that discussing them is out of the scope of this review and was previously discussed (25).
2.2.5. Patient’s Adherence and Its Assessment
According to the WHO, “adherence” or “compliance” is defined by the extent to which the behavior-taking medication, following a diet, and/or executing lifestyle changes, adapts with the recommendations of the healthcare provided (46).
Low adherence of the patients, reported as 30 to 50%, is an important challenge for DCTs, in cases of diet/lifestyle modification and long-term interventions (9, 47). In DCTs, particularly those involved difficult-to-implement diets (e.g., a very low-carbohydrate diet, very low energy diets, sodium-restricted diets) or strict elimination diets, or those that take years to complete, compliance of the study participants with diet or food intervention is an extremely difficult issue.
Psychological, cultural, environmental, and behavioral factors influence one’s adherence to nutrition interventions (48). Once dietary changes are intervention of interest, the habitual dietary practices of the study population need to be considered to deal with the cultural obstacles to eating the "proper" foods or diet (49). Different methods, including educational tools (e.g., video, booklet), individual and group sessions with a dietitian to barrier identification and problem-solving, reminders (e.g., telephone follow-up), motivational interviewing, promotion of self-monitoring and feedback diary, stress management and goal setting, or contract with the involvement of a family member or friend are available to increase adherence to interventions (50, 51). Developing skills in dish preparation, raising awareness regarding the high variety of foods, and adapting the recipes to the cultural preferences are general approaches for enhancement of the adherence to more complicated dietary patterns such as vegetarian diets (52).
To improve the adherence rate in DCTs, researchers deserve to acquire knowledge related to predictors of adherence to both medical and lifestyle modification interventions [for more details, see (47, 53)]. A controlled-feeding design is also considered the gold standard to maximize participant adherence (14).
Measuring adherence of the study population helps indicate the extent to which intervention of interest has achieved its specific aims. Considering the assumption that a nutritional marker in biological samples (e.g., plasma, urine, saliva, hair) is tightly related to its dietary intakes, nutritional biomarkers can be used as measures of patients’ adherence in DCTs to clearly define the compliance with a new dietary regimen or food intervention (54). Both biological and functional nutritional biomarkers can be useful in the reflection of the dietary intake, nutrients’ metabolism, and the possible influence on the disease progression. The assessment of dietary food intake would require the use of biological biomarkers, whereas the measurement of function or biological response would be related to biomarkers of effect/function (30, 55). These biomarkers reflect not only the intake but also the metabolism of nutrients and, possibly, effects on disease processes (30).
2.2.6. Measurement of Efficacy
Evaluation of the efficacy of the nutrition intervention on the patient’s response needs a valid measure of change from baseline to endpoint (over the study period) or accurate diagnosis of a clinical outcome at the end of the study (e.g., cardiovascular events). In case of interventions that evaluate change of an objective biomedical marker (e.g., weight, plasma lipids), the measurement and analysis are relatively straightforward, whereas if DCT requires measurement of change in self-reported diets or dietary behavior, the task is complex due to many possible biases (28). Furthermore, in contrast to drug, targeting an individual and specific pathway or function in the human body, dietary components have functional roles in several physiological pathways; definition and measurement of a specific outcome, therefore, may be impossible or even misleading somewhere. For example, if off-target effects of dietary intervention are not captured, an inaccurate picture of its true effects would be drawn (54).
2.2.7. Attrition Rate
High attrition rate (reported average ~ 20 - 49%) is one of the main concerns of DCTs, especially in case of long-term interventions (9); it is a serious threat to internal and external validity of the study, and lead to inadequate power of the observed effect size which can greatly affect the strength of a trial's findings (56-58). For example, an attrition rate of 49.3% was reported in a 12-month randomized dietary intervention trial, investigated the effects of dairy foods on cardiometabolic health in overweight subjects (9); similarly, a dropout rate of 25.9% was reported in a 68-week dietary trial investigated the effects of low-fat diets on cardiovascular disease risk (59). Likewise, an attrition rate of 42.4% occurred in a 12-month randomized trial that assessed the effects of high-protein, low-fat diet in obese adults with T2DM (60).
Therefore, making a reasonable estimation of expected dropout rates is critical for DCTs. Under- and over-estimation of the expected dropout rates may result in false results. Underestimation increases the risk of negative outcomes and overestimation leads to magnified sample sizes, raising concerns in both ethical and economic issues (61). In particular, the parameters related to the attrition in dietary clinical trials have not been completely defined and understood, and researchers seem to estimate dropout rates mainly based on the rule of thumb and personal experience. Referring to previously published trials on similar dietary interventions or specific food-based interventions can provide some helpful hints for researchers to reliably estimate the expected dropout rates.
The dropout rates may be unequal (called differential dropout or attrition) between treatment arms and tend to be increased in a control group due to their dissatisfaction (62, 63). If applicable, dropout rates during the trial need to be analyzed and compared between intervention and control groups to provide information about the feasibility and safety of the intervention.
Similar to other complex interventional trials that report different dropout and compliance rates among females and males (61), DCTs may also be subject to this issue. Thus, when planning respective studies, it is important to note the exceptional and special affairs related to each individual trial.
2.2.8. Timing and Duration of Interventions
In terms of duration, DCTs may include short-term and acute studies evaluating the postprandial effects of specific dietary interventions, and long-term and chronic investigations assessing risk markers or incidence of disease, which could last for months or years (64)). For example, a follow-up duration of 12 to 24-months was reported to be essential to assess potential efficacy of a low-carbohydrate and a low-fat diet on glycemic parameters in patients with T2DM (65, 66).
Some dietary interventions may require longer administration over several weeks for a significant effect to be exerted. The intestinal effect of probiotics and the changes induced by stanols or sterols on cholesterol metabolism are noteworthy examples. It might also take months or years to get to a stage at which the key effects could be observed, which can be exemplified by the bone density response to calcium supplementation. Prolonged interventions can be financially costly and result in higher dropout rates, and reduced compliance and logistical hurdles (67).
Taken together, standard study durations must endorse the efficacy, and if applicable, the sustainability of effect (67). Thus, intervention duration must be balanced. It must be adequately concise to pursue high compliance, appear cost-efficient, and prevent significant dropout rates, and lengthy enough to guarantee the objective biological efficacy. To reach and maintain an optimal intervention duration, different factors, including the availability of the resources, acceptability of the dietary treatments, and restrictions over a prolonged period, and the required time for the achievement of stable effects on endpoint/outcome measures (i.e., to capture the most feasible significant biological effect) should be taken into consideration (67).
2.2.9. Sample Size and the Fragility of Statistically Significant Results
Standard and appropriate sample size is defined as the minimum number of participants for the detection of a scientific effect. It most commonly depends on the primary study outcome, and is an important element in the study design (67). In general, pharmaceutical compounds tend to show more beneficial effects as compared to micro-compounds of nutrition interventions, where the implications are usually close to the “noise” threshold of biological variability (68). Some practical guides for conducting sample size planning for statistical power, with the types of data and hypotheses commonly encountered in nutrition research are available (69).
2.2.10. Logistical Problems
There are several logistical challenges that make DCTs more difficult than other intervention studies. Researchers most likely face important obstacles while conducting DCTs. Some of these challenges include designing complex dietary menus, the procurement, storage, and intervention delivery to the participant. Participants may also have reciprocal difficulties with receiving the intervention, storage, preparation, and protocol compliance (24). In some DCTs such as ‘controlled-feeding design’, all required foods and beverages are provided to the participants. The diet administered to the placebo group is inert in nature and is distinguished with the intervention by the absence of the active component being investigated (43, 70), face with serious logistical challenges. These studies can be challenging in terms of designing controlled diets through specific software, meeting the food and paper supply requirements, providing equipment for food preparation and storage, employing trained staff to carry out the stages related to study design, and daily intervention preparation and delivery (70).
To identify and successfully overcome such logistical challenges, development of pilot trial, defined as “a small-scale test of the methods and procedures to be used on a larger scale” (71), may be helpful (64).
3. Conclusions
The promising future perspective of DCTs includes the establishment of nutritional interventions that reduce the relative risk of overall mortality by 5 - 10% among the general population rather than merely high-risk patients. These will not be achieved unless DCTs employ larger sample sizes and longer follow-up duration, build a relationship with disease and mortality registries, and dedicate diligence efforts to maximize participants’ adherence. DCTs are required to be focused either on dietary patterns and diet behaviors alongside socioeconomic factors, affecting lifestyle modifications and adherence to the interventions. Although the quality of the study design, rationale, and statistical analysis of the nutritional interventions are assessed based on similar criteria with drug trials, innovative study designs will certainly help overcome common obstacles and achieve the optimum quality of DCTs.