1. Context
The pandemic nature of coronavirus disease 2019 (COVID-19) has taken a heavy toll in the world, with almost 1.8 million deaths within a year. The subtle flu-like nature of the infection has deceived the population in terms of severity and mortality. Although there is a vaccination drive worldwide, the emerging new strains of COVID-19 has put the success of this drive at risk (1).
The clinical manifestation of the disease has been heterogeneous. The reports have been varied in terms of respiratory symptoms, dermatological symptoms, gastrointestinal symptoms, hematological disturbances, and neurological breakdowns. Furthermore, patients with comorbidities such as diabetes, hypertension, and cardiac disease have been found to be at a significantly higher risk of hospital admissions, severe disease, and mortality (2). The disease originated in China, and according to the statement of the Centers for Disease Control and Prevention (CDC), 2.3% of COVID-19 mortality was seen in critical patients (3).
Diabetes is one of the commonest occurring lifestyle diseases worldwide, which is associated with multi-system damage in the long run. It is one of the arms of the complex branch of metabolic syndrome X, which carries with itself numerous other chronic conditions. The susceptibility of COVID-19 infection increases in patients with diabetes as their immune functions is significantly decreased. Literature reports a diabetes prevalence of 7.87% - 20% in COVID-19 patients (4-7). Studies have shown that it is one of the predictors of poor prognosis and mortality among the patients with COVID-19 (8). Moreover, the association of diabetes with adverse outcomes has also been observed in other human coronaviruses like severe acute respiratory syndrome (SARS) and Middle East respiratory syndrome (MERS) (9).
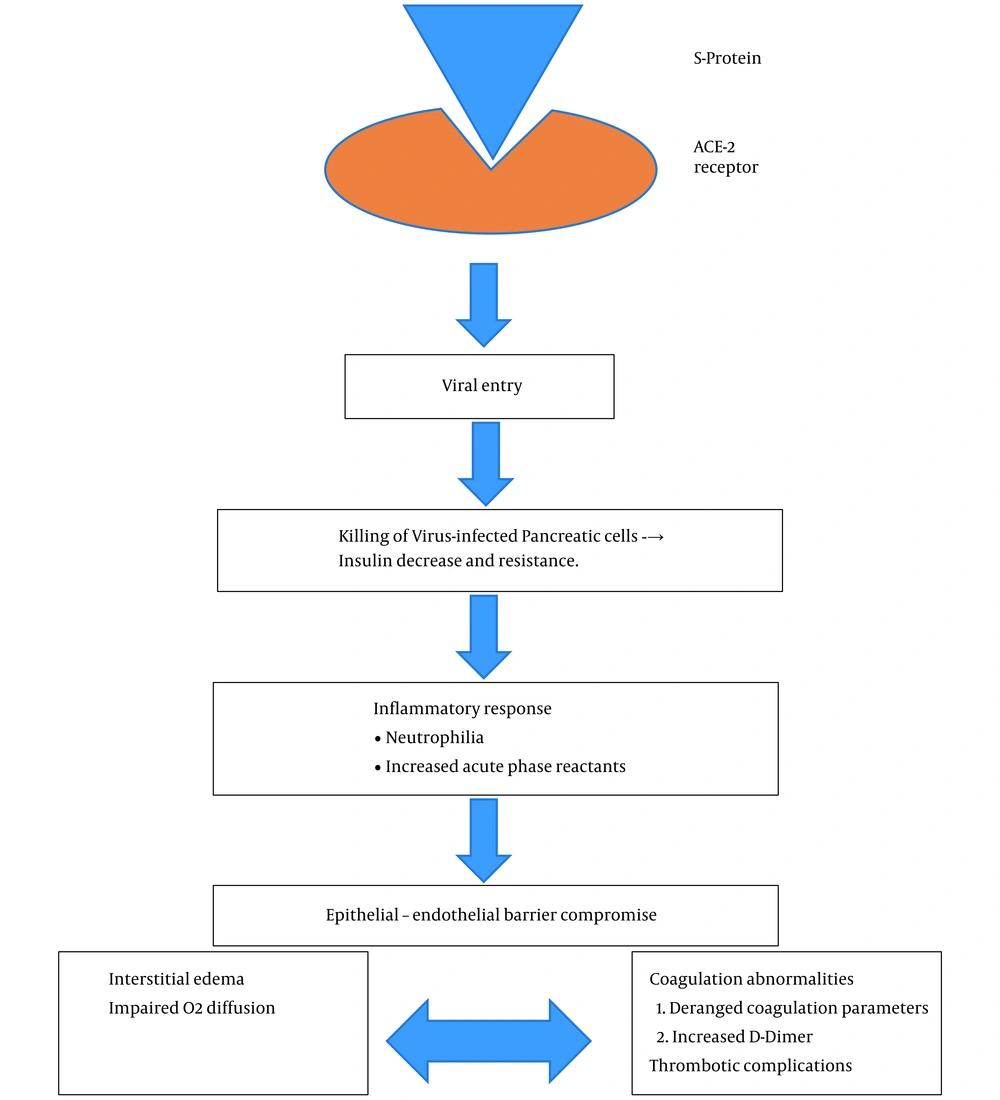
Various underlying mechanisms have been proposed for the substantially increased risk of infection in diabetes as follows: (1) Increased inflammatory markers; (2) hypercoagulability; (3) unregulated inflammatory responses to viral infection; (4) decreased “viral clearance” due to low immunity; and (5) presence of other comorbidities like cardiovascular diseases (2, 9). Figure 1 shows the pathophysiological association of COVID-19 with diabetes.
After an individual is infected with COVID-19, the body initiates an innate immune response releasing a plethora of cytokines and inflammatory markers, thereby altering endothelial cells structure and function, altered insulin delivery, and insulin resistance (10). Chen et al. (4) further added the occurrence of liver damage impairing one’s ability to utilize glucose, causing a surge in the blood glucose levels. Molecular studies have shown that angiotensin converting enzyme 2 (ACE2) (11) is also one of the targets of coronavirus, leading to islet cell damage of the pancreas (12). This hyperglycemia is exaggerated in diabetics causing more oxidative stress, vascular damage, and multi-organ damage, including heart and kidneys.
The present meta-analysis aimed to determine the association of diabetes with mortality in COVID-19 patients. Our results may help in providing substantial evidence, thus laying a platform for prevention and control of diabetes in the growing pandemic of COVID-19.
2. Methods
To enlist the articles investigating diabetes-associated mortality in patients with COVID-19, searching the databases was conducted on December 24, 2020. The final search was updated on December 31, 2020. All the related studies were retrieved in their summary format for screening.
2.1. Search Strategy
The articles were searched on Pubmed, Embase, MEDLINE, Web of Science, Google Scholar, and DOAJ databases.
The keywords and their MeSH words were used in the search, which included diabetes (all fields), diabetes mellitus (MeSH), diabetes insipidus (MeSH), outcome (all field), admitted (all field), hospitalization (all field), severity (all field), mortality (all field), COVID-19, SARS-CoV-2 (MeSH), severe acute respiratory syndrome coronavirus 2 (MeSH), NCOV (MeSH), and 2019 NCOV (MeSH).
The search was done in the advanced mode using the Boolean operator “OR” and “AND”. The final advanced search builder showed: ((((((((((((Diabetes) OR (diabetes mellitus)) OR (diabetes insipidus)) AND (outcome)) OR (admitted)) OR (hospitalization)) OR (severity)) AND (mortality)) AND (COVID-19)) OR (SARS-CoV-2)) OR (Severe Acute Respiratory Syndrome Coronavirus 2)) OR (NCOV)) OR (2019 NCOV).
We screened the free full-text of articles written in English language and conducted on humans.
2.2. Inclusion Criteria
We included case-control/cohort studies conducted on COVID-19 patients, as well as the studies representing the data of COVID-19 mortality and diabetes-associated mortality.
2.3. Exclusion Criteria
The following cases were excluded from the study: systematic reviews, editorials, meta-analysis, and review articles (narrative); studies from which the odds risk for diabetes-associated mortality could not be determined; duplicate studies reporting same results; and studies with a 50% matching data from a single center.
2.4. Reviewing Process
The studies were independently reviewed by two authors (P.G and M.G). Any discrepancy was resolved after discussing the issue with two other authors (K.G and N.K). The full texts of all eligible articles were retrieved and assessed. The full-text of all included studies was available in open access, and in cases where only abstract was available, the article was not included.
2.5. Outcomes
The primary outcome of the study was (1) the incidence of mortality in patients with COVID-19 and (2) pooled diabetes-associated mortality risk in patients with COVID-19.
2.6. Data Extraction and Quality Assessment
The data extraction was done independently by two authors (KG and BG). A Microsoft Excel sheet was prepared to record the outcomes of the included studies. The extracted parameters from the studies included the last name of author (s), year of publication, location, total patients, age, gender, total mortality, proportion of patients with diabetes, and diabetes-associated mortality. The quality of the articles was adjudged by Newcastle-Ottawa Quality Assessment scale (NOS). This tool scores the studies on the basis of patient selection, patient comparability, and outcomes with a score ranging from 0 to 9. The total score of > 7 is considered as good quality (13).
2.7. Statistical Analysis
Statistical analysis was performed using the Stata (version 13). The most common measures of effect used for dichotomous data were the odds ratio (OR). The OR of mortality in diabetes mellitus patients was calculated with 95% confidence interval (CI). The studies were weighted as per the sample size to reflect the value of its evidence. It is important to recognize the smaller studies as they contribute less to the estimates of overall effect.
Random-effects models was used to synthesize data for the relevant outcomes with the assumption that distribution of effects exists, resulting in heterogeneity among study results, known as τ2. Heterogeneity was evaluated using I2 statistic. This is desirable because of the strong assumption that the effect of interest is frequently untenable in all studies.
A meta-analysis was conducted for the included studies in terms of subjects involved, interventions, and outcomes to provide a meaningful summary. Forest plots were made, which visually showed effect estimates of the included studies. Funnel plots and Egger’s test were used to evaluate potential publication bias. A two tailed P < 0.05 was considered as statistically significant.
3. Results
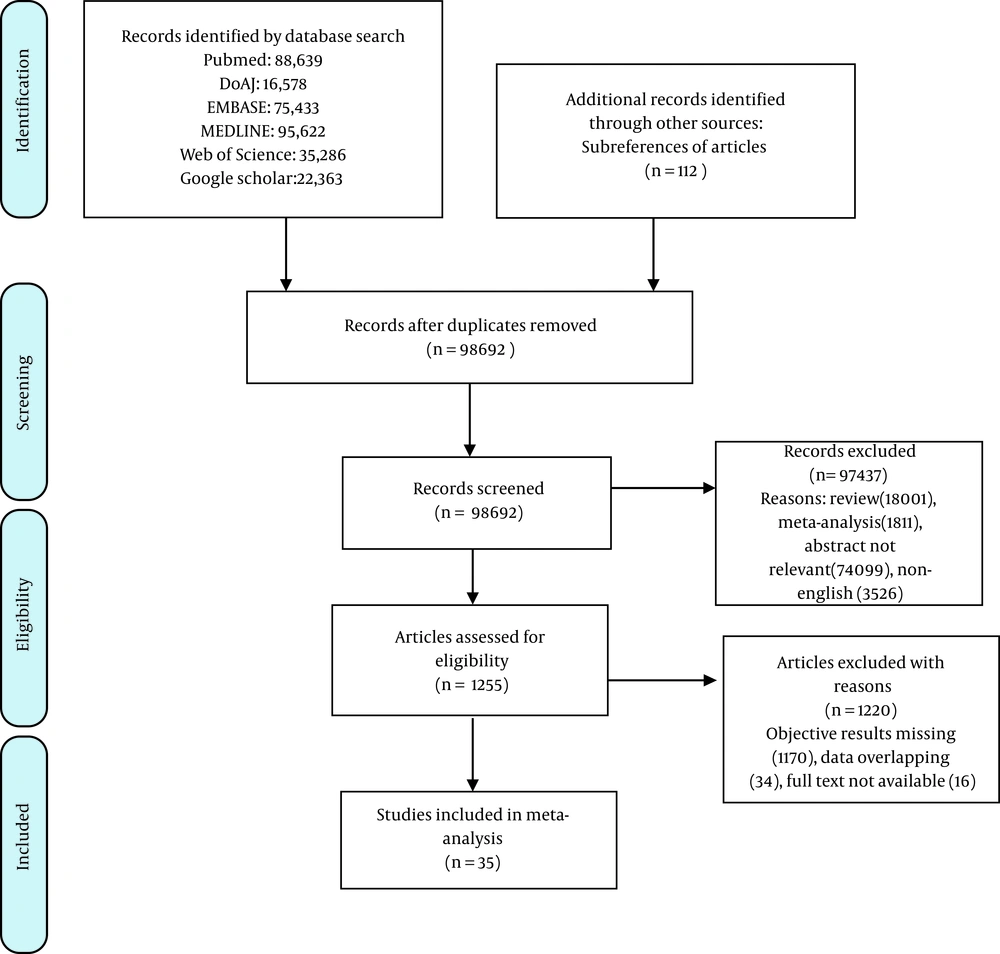
The flow of studies in our meta-analysis is depicted in Figure 2. A total of 35 studies with 25,934 patients were finally included for meta-analysis (9, 14-47). Among them, 23 studies were conducted in China (20-23, 25-31, 33, 34, 38-47), and 12 were conducted outside China (9, 14-19, 24, 32, 35-37). The study characteristics are shown in Table 1.
| Study | Location | N, Study Design | Diabetes population, No. (%) | Age, y | Males, % | Total Mortality | Diabetes Mortality | Diabetes as mortality risk (OR with 95% CI) |
|---|---|---|---|---|---|---|---|---|
| Alshukry et al. (14) | Jaber Al-Ahmad Hospital, Kuwait. | 417, R | 97 (23.3) | 47 (32, 60) | 262 (63) | 60 | 24 | (OR 2.59; 95% CI, 1.46 - 4.62) |
| Borobia et al. (15) | La Paz University of Spain | 2226, P | 381 (17.1) | 61 (46, 78) | 1074 (48.2) | 460 | 157 | (OR 3.57; 95% CI, 2.81 - 4.53) |
| Alguwaihes et al. (16) | Riyadh, Saudi Arabia | 439, R | 300 (68.34) | 55 | 300 (68.3) | 77 | 61 | (HR: 1.2, 95% CI: 0.7–2.3) |
| Yadaw et al. (Development Set) (17) | Mount Sinai Hospital System, New York | 3841, P | 608 (15.8) | 56.2 ± 19 | 2125 (55.3) | 313 | 90 | (OR 2.35; 95% CI, 1.80 - 3.5) |
| Morrison et al. (18) | Five hospital health systems in Michigan | 81, R | 37 (45.7) | 64 (58, 71) | 56 (69.1) | 35 | 14 | (OR 0.67; 95% CI, 0.27 - 1.6) |
| Bhatti et al. (19) | Dubai | 103, OC | 26 (25.24) | 54 | 69 (66.9) | 5 | 1 | (OR: 0.73, 95% CI, 0.078 - 6.84) |
| Cao et al. (20) | Zhongnan Hospital | 102, R | 11 (10.8) | 54 (37, 67) | 53 (52) | 17 | 6 | (OR 8.73; 95% CI, 2.28 - 33.46) |
| Chen et al. (21) | Wuhan (China) | 1105, R | 138 (12.49) | 56.3 | 597 (54) | 217 | 49 | (HR: 7.96; 95% CI 2.25 - 28.24) |
| Chen et al. (22) | The Central Hospital of Wuhan | 660, R | 114 (17.3) | 55 (34, 68) | 295 (44.7) | 82 | 21 | (OR 1.80; 95% CI, 1.04 - 3.09) |
| Chen et al. (23) | Tongji hospital | 274, R | 47 (17) | 62 (44, 70) | 171 (62) | 113 | 24 | (OR 1.62; 95% CI, 0.86 - 3.04) |
| Chung et al. (24) | Yeungnam University Hospital in Daegu, South Korea | 110, R | 29 (26.4) | 56.9 ± 17.0 | 48 (43.6) | 6 | 5 | (OR 16.67; 95% CI, 1.86 - 149.66) |
| Deng et al. (25) | Hankou and Cai-dian branch of Tongji Hospital | 225, R | 26 (11.6) | death: 69 (62, 74); recovered: 40 (33, 57) | 124 (55.1) | 109 | 17 | (OR 2.20; 95% CI, 0.93 - 5.16) |
| Du et al. (26) | Wuhan Pulmonary Hospital | 179, P | 33 (18.4) | 57.6 ± 13.7 | 97 (54.2) | 21 | 6 | (OR 1.94; 95% CI, 0.69 - 5.45) |
| Fu et al. (27) | Union Hospital of Huazhong University of Science and Technology | 200, P | 137 (68.5) | 39 (19.5%) cases older than 70 years | 99 (49.5) | 34 | 26 | (OR 1.61; 95% CI, 0.68 - 3.79) |
| Gu et al. (28) | Mainland China | 275, CC | 72 (26.2) | 66.4 ± 14.5 | 173 (62.9) | 94 | 26 | (OR 1.12; 95% CI, 0.64 - 1.97) |
| Guan et al. (29) | 575 hospitals in China | 1590, R | 130 (8.2) | 48.9 ± 16.3 | 904 (57.3) | 50 | 13 | (OR 4.27; 95% CI, 2.21 - 8.26) |
| Guo et al. (30) | Wuhan Union hospital | 174, R | 37 (21.1) | 59 (49, 67) | 76 (43.7) | 9 | 4 | (OR 3.20; 95% CI, 0.81 - 12.58) |
| He et al. (31) | The Sino-French New City Branch of Tongji hospital | 54, R | 13 (24.1) | 68 (59, 74) | 34 (63) | 26 | 8 | (OR 2.04; 95% CI, 0.57 - 7.33) |
| Paranjpe et al. (32) | 5 major hospitals of New York city | 2199, OC | 583 (26.5) | 65 (54, 76) | 1293 (58.8) | 310 | 105 | (OR 2.09; 95% CI, 1.56 - 2.81) |
| Li et al. (33) | Wuhan (China) | 199, R | 76 (38.19) | 63 | 89 (44.7) | 18 | 11 | OR: 10.816; 1.895 - 61.741) |
| Luo et al. (34) | Eastern Campus of Renmin Hospital, Wuhan University | 403, R | 57 (14.1) | 56 (39, 68) | 193 (47.9) | 100 | 25 | (OR 2.82; 95% CI, 1.58 - 5.05) |
| Mirani et al. (35) | Italy | 385, OC | 90 (23.38) | 66 | 257 (66.7) | 102 | 38 | (OR: 2.638, 95% CI, 1.597 - 4.356) |
| Moon et al. (9) | South Korea | 5307, P | 770 (14.51) | 56 | 2043 (38.5%) | 211 | 94 | (OR: 5.253, 95% CI, 3.957 - 6.973) |
| Nikpouraghdam et al. (36) | Baqiyatallah hospital in Tehran, Iran | 2964, R | 113 (3.8) | 56 (46, 65) | 1955 (66.0) | 239 | 11 | (OR 1.24; 95% CI, 0.66 - 2.34) |
| Shahriarirad et al. (37) | Five hospitals affiliated by the Shiraz University | 113, R | 16 (14.2) | 53.75 | 71 (62.8) | 9 | 2 | (OR 1.84; 95% CI, 0.35 - 9.75) |
| Shi et al. (38) | Renmin Hospital and Zhongnan Hospital of Wuhan University | 306, R | 153 (50.0) | No diabetes: 65 (56, 72); diabetes:64 (56, 72) | 150 (49.0) | 47 | 31 | (OR 2.18; 95% CI, 1.13 - 4.17) |
| Wang et al. (39) | Zhongnan Hospital of Wuhan University and Xishui People’s Hospital | 107, R | 11 (10.3) | 51 (36, 65) | 57 (53.3) | 19 | 5 | (OR 4.88; 95% CI, 1.31 - 18.18) |
| Wang et al. (40) | China | 663, R | 67 (10.11) | 58 | 321 (48.4%) | 25 | 3 | Poor therapeutic effect: (OR 2.99; 95% CI, 1.07 - 8.66) |
| Wang et al. (41) | Hankou Hospital | 121, R | 25 (20.7) | 59 (46, 67) | 66 (54.6) | 26 | 7 | (OR 1.58; 95% CI, 0.58 - 4.31) |
| Wang et al. (42) | Renmin Hospital of Wuhan University | 339, OC | 54 (16.0) | 69 (65, 76) | 166 (49.0) | 65 | 11 | (OR 1.09; 95% CI, 0.53 - 2.26) |
| Yan et al. (43) | Tongji hospital | 193, OC | 48 (24.9) | 64 (49, 73) | 114 (59.1) | 108 | 39 | (OR 4.77; 95% CI, 2.16 - 10.57) |
| Yang et al. (44) | Wuhan Jin Yin-tan hospital | 52 R | 9 (17) | 59.7 ± 13.3 | 35 (67) | 32 | 7 | (OR 2.52; 95% CI, 0.47 - 13.58) |
| Zhang et al. (45) | Wuhan No.1 Hospital | 48 R | 10 (20.8) | 70.6 ± 13.4 | 33 (68.8) | 17 | 5 | (OR 2.17; 95% CI, 0.53 - 8.93) |
| Zhang et al. (46) | Zhongnan Hospital and No.7 hospital of Wuhan | 289, R | 27 (9.3) | 57 (22, 88) | 154 (53.3) | 49 | 7 | (OR 1.83; 95% CI, 0.73 - 4.61) |
| Zhou et al. (47) | Jin Yin-tan hospital and Pulmonary Hospital | 191, R | 36 (19) | 56 (46, 67) | 119 (62) | 54 | 17 | (OR 2.85; 95% CI, 1.35 - 6.05) |
Abbreviations: CC, case control; OC, observational cross-sectional; P, prospective cohort; R, retrospective observational cohort.
The pooled prevalence of diabetes mellitus in patients with COVID-19 was 16.8% (n = 4381). The overall mortality seen in all the studies was 12.81% (n = 3159), and diabetes-associated mortality was 22.14% (n = 970).
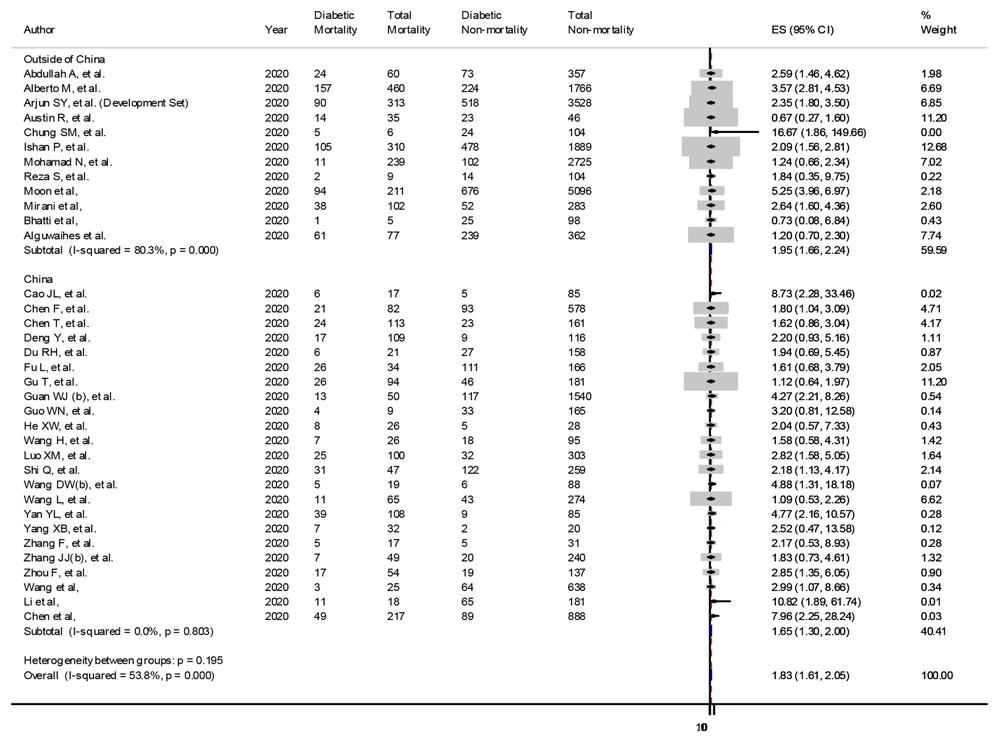
Among the 12 studies conducted outside China, seven studies (9, 14, 15, 17, 24, 32, 35) reported a significantly increased risk of diabetes-associated mortality, but five studies (16, 18, 19, 36, 37) failed to show a statistical association of diabetes with mortality. The 12 studies carried a weightage of 59.59% with an OR of 1.95 (95% CI 1.66 - 2.24) for mortality in diabetes with COVID-19. The heterogeneity in the studies was significantly high (I2 = 80.3%, P < 0.0001), but all the studies were of high quality as per the NOS scale. The studies value was adjusted as per their weightage, as shown in the forest plot (Figure 3).
In 23 studies conducted in China, 13 studies (20-22, 24, 29, 33, 34, 38-40, 43) reported a significantly increased risk of diabetes-associated mortality, but 12 studies (23, 25-28, 30, 31, 41, 42, 44-46) only found an increased risk, which failed to cross the statistical boundaries. The 23 Chinese studies carried a weightage of 40.41% with an OR of 1.65 (95% CI 1.3 - 2) for mortality in diabetes with COVID-19. The studies had a low heterogeneity (I2 = 0.0%, P = 0.803), and all the studies were of high quality as per the NOS scale. The studies’ values were adjusted as per their weightage. The forest plot has been presented in Figure 3.
The pooled analysis of 35 studies showed that diabetes mellitus had a significantly higher mortality rate (22.14% vs. 12.81%, P < 0.05) with higher odds of death (pooled OR 1.83, 95% CI: 1.61 - 2.05). The subgroup analyses in different locations showed similar results (P = 0.195) (Figure 3).
3.1. Publication Bias
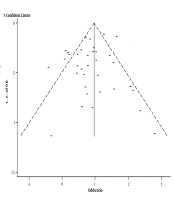
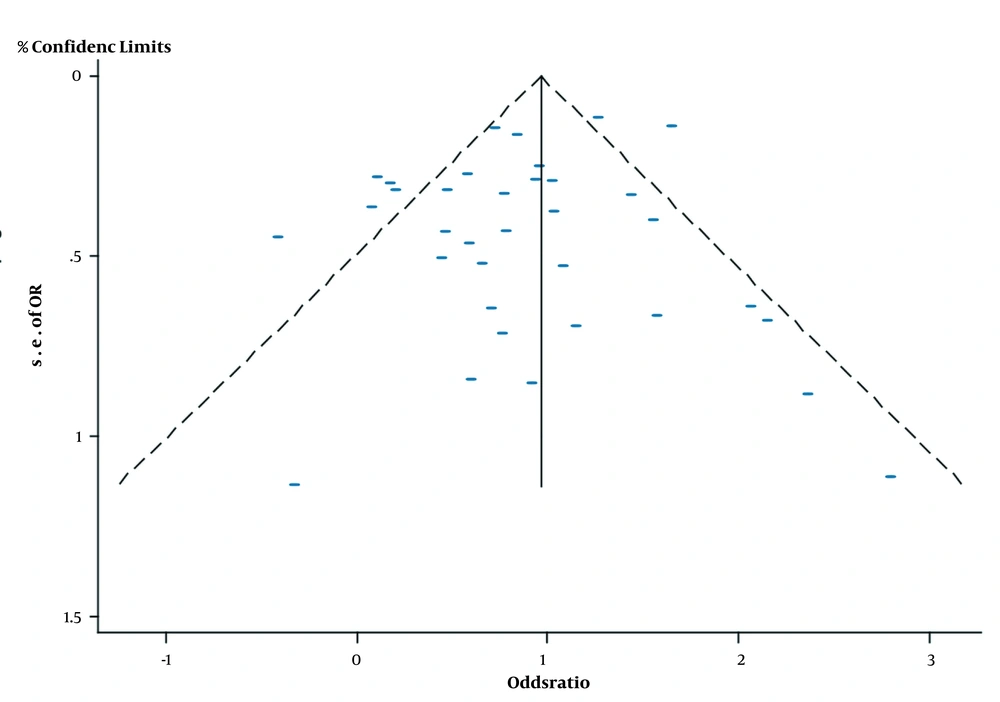
The NOS scale showed that all studies were of good quality, and no study remarkably affected the pooled mortality (Table 2). The funnel plot was symmetric, with Egger’s test showing a P-value of 0.268, thereby indicating a low risk of publication bias (Figure 4).
| Study | Selection | Comparability | Exposure | Total Score (0 - 9) | |||||
|---|---|---|---|---|---|---|---|---|---|
| Is the Case Definition Adequate? (⋆) | Representativeness of the Cases (⋆) | Selection of Controls (⋆) | Definition of Controls (⋆) | Comparability of CASES and controls on the Basis of the Design or Analysis (⋆⋆) | Ascertainment of Exposure (⋆) | Same Method of Ascertainment for Cases and Controls (⋆) | Non-Response Rate (Dropouts) (⋆) | ||
| Alshukry et al. (14) | ⋆ | ⋆ | ⋆ | ⋆ | ⋆⋆ | ⋆ | ⋆ | ⋆ | 9 |
| Borobia et al. (15) | ⋆ | ⋆ | ⋆ | ⋆ | ⋆⋆ | ⋆ | ⋆ | 8 | |
| Alguwaihes et al. (16) | ⋆ | ⋆ | ⋆ | ⋆ | ⋆⋆ | ⋆ | ⋆ | 8 | |
| Yadaw et al. (17) | ⋆ | ⋆ | ⋆ | ⋆ | ⋆⋆ | ⋆ | 7 | ||
| Morrison et al. (18) | ⋆ | ⋆ | ⋆ | ⋆ | ⋆⋆ | ⋆ | 7 | ||
| Bhatti et al. (19) | ⋆ | ⋆ | ⋆ | ⋆ | ⋆⋆ | ⋆ | 7 | ||
| Cao et al. (20) | ⋆ | ⋆ | ⋆ | ⋆ | ⋆⋆ | ⋆ | 7 | ||
| Chen et al. (21) | ⋆ | ⋆ | ⋆ | ⋆ | ⋆⋆ | ⋆ | ⋆ | 8 | |
| Chen et al. (22) | ⋆ | ⋆ | ⋆ | ⋆ | ⋆⋆ | ⋆ | ⋆ | ⋆ | 9 |
| Chen et al. (23) | ⋆ | ⋆ | ⋆ | ⋆ | ⋆⋆ | ⋆ | ⋆ | ⋆ | 9 |
| Chung et al. (24) | ⋆ | ⋆ | ⋆ | ⋆ | ⋆⋆ | ⋆ | ⋆ | 8 | |
| Deng et al. (25) | ⋆ | ⋆ | ⋆ | ⋆ | ⋆⋆ | ⋆ | ⋆ | ⋆ | 9 |
| Du et al. (26) | ⋆ | ⋆ | ⋆ | ⋆ | ⋆⋆ | ⋆ | ⋆ | 8 | |
| Fu et al. (27) | ⋆ | ⋆ | ⋆ | ⋆ | ⋆ | ⋆ | ⋆ | 7 | |
| Gu et al. (28) | ⋆ | ⋆ | ⋆ | ⋆ | ⋆⋆ | ⋆ | ⋆ | 8 | |
| Guan et al. (29) | ⋆ | ⋆ | ⋆ | ⋆ | ⋆⋆ | ⋆ | ⋆ | ⋆ | 9 |
| Guo et al. (30) | ⋆ | ⋆ | ⋆ | ⋆ | ⋆⋆ | ⋆ | ⋆ | 8 | |
| He et al. (31) | ⋆ | ⋆ | ⋆ | ⋆ | ⋆ | ⋆ | ⋆ | 7 | |
| Paranjpe et al. (32) | ⋆ | ⋆ | ⋆ | ⋆ | ⋆⋆ | ⋆ | 7 | ||
| Li et al. (33) | ⋆ | ⋆ | ⋆ | ⋆ | ⋆⋆ | ⋆ | 7 | ||
| Luo et al. (34) | ⋆ | ⋆ | ⋆ | ⋆ | ⋆⋆ | ⋆ | ⋆ | ⋆ | 9 |
| Mirani et al. (35) | ⋆ | ⋆ | ⋆ | ⋆ | ⋆⋆ | ⋆ | 7 | ||
| Moon et al. (9) | ⋆ | ⋆ | ⋆ | ⋆ | ⋆⋆ | ⋆ | 7 | ||
| Nikpouraghdam et al. (36) | ⋆ | ⋆ | ⋆ | ⋆ | ⋆⋆ | ⋆ | 7 | ||
| Shahriarirad et al. (37) | ⋆ | ⋆ | ⋆ | ⋆ | ⋆⋆ | ⋆ | ⋆ | ⋆ | 9 |
| Shi et al. (38) | ⋆ | ⋆ | ⋆ | ⋆ | ⋆⋆ | ⋆ | ⋆ | ⋆ | 9 |
| Wang et al. (39) | ⋆ | ⋆ | ⋆ | ⋆ | ⋆⋆ | ⋆ | ⋆ | ⋆ | 9 |
| Wang et al. (40) | ⋆ | ⋆ | ⋆ | ⋆ | ⋆⋆ | ⋆ | 7 | ||
| Wang et al. (41) | ⋆ | ⋆ | ⋆ | ⋆ | ⋆⋆ | ⋆ | 7 | ||
| Wang et al. (42) | ⋆ | ⋆ | ⋆ | ⋆ | ⋆⋆ | ⋆ | ⋆ | ⋆ | 9 |
| Yan et al. (43) | ⋆ | ⋆ | ⋆ | ⋆ | ⋆⋆ | ⋆ | 7 | ||
| Yang et al. (44) | ⋆ | ⋆ | ⋆ | ⋆ | ⋆⋆ | ⋆ | 7 | ||
| Zhang et al. (45) | ⋆ | ⋆ | ⋆ | ⋆ | ⋆⋆ | ⋆ | ⋆ | 8 | |
| Zhang et al. (46) | ⋆ | ⋆ | ⋆ | ⋆ | ⋆⋆ | ⋆ | ⋆ | ⋆ | 9 |
| Zhou et al. (47) | ⋆ | ⋆ | ⋆ | ⋆ | ⋆⋆ | ⋆ | ⋆ | ⋆ | 9 |
4. Discussion
This is one of the first meta-analysis which used all the studies published in 2020, thereby encompassing a large sample size for evaluation. The results confirmed that the prevalence of diabetes was high (16.8%), and it carries an increased risk of mortality in patients with COVID-19 (OR = 1.83, 95% CI: 1.61 - 2.05). This was in line with the recent review by Pinedo-Torres et al. (48) who showed a prevalence of 100.42 per 1000 COVID-19 patients (95% CI: 77.85 - 125.26, I2 = 67.94%) in SARS-Cov-2.
Among the 35 included studies, the increased OR in 17 studies failed to reach a statistical significance (16, 18, 19, 25-28, 30, 31, 36, 37, 41, 42, 44-46). This might be due to the adjustment with respect to other comorbidities.
The findings have been concordant with the meta-analysis of Shang et al. (2), reporting an OR of 2.21 for diabetes-associated mortality.
Another study accounted diabetes for a high mortality in COVID-19, that is, 40% (49).
Also another meta-analysis by Huang et al. (50), coroborated with our study reporting an RR of 2.12 for diabetes associated mortality.
Diabetes is one of the comorbid conditions found in the elderly patients getting infected with COVID-19. Its prevalence among COVID-19 patients has been on an increase in the last few months (9). One of the meta-analysis portrayed that a pooled prevalence of diabetes mellitus among the patients with COVID-19 was 14.34% (51). The underlying factors in diabetes mellitus that may affect the mortality remain wide. Carrasco-Sanchez et al. (52), in a retrospective multicenter review, showed that hyperglycemia was an independent risk factor for mortality irrespective of the pre-existing diabetes (HR = 1.5, 95% CI: 1.31 - 1.73 for blood glucose > 180 mg/dL). The debate on the anti-diabetic medications conducive for use in COVID-19 among a plethora of oral hypoglycemics was solved by Cheng et al. (53), where the use of metformin showed no significantly increased risk of 28-days mortality [adjusted HR 1.65, 95% CI: 0.71 - 3.86, P = 0.247), which might be associated with reduction in inflammation. However, the use of metformin was associated with an increased risk of acidosis and derangement of kidney functions, which increased hospital admissions. Li et al. (33) compared the clinical characteristics of diabetic and nondiabetic COVID-19 patients. Compared to nondiabetics, diabetics were older, with higher levels of D-dimer, glycemic levels, and blood urea at admission and lower levels of oxygen saturation, lymphocyte counts, and plasma albumin; these parameters were labeled as important in conferring a higher mortality (P < 0.05). The possible role of hypercoagulability, as an underlying mechanism has been reported in the studies by Cheng et al. (53) and Chen et al. (21).
The results of the current study and few previous meta-analysis studies showed that diabetes can increase the risk of mortality in COVID-19 patients, thus warranting a strict glycemic control, regular insulin infusions, pre-admissions HbA1c check, and compliance to the anti-diabetic medications till the pandemic is controlled. This has been regularly enforced by expert panels from American Diabetes Association (9), Chinese Diabetes Society (54), the US CDC, Korean Diabetes Association (9), and the European Society of Endocrinology (55).
Since we are entering into the phase of vaccination, the control of inflammatory surge under the background of weakened immunity (as seen in diabetes) needs further research. Although vaccination drive has started, the current clinical management for the diabetes should be continuously advised to the susceptible population.
This study had some limitations. First, the confounding variables such as duration of diabetes, ongoing medications, blood glucose levels, and HbA1c levels were not assessed. Second, due to an initial surge in cases in China, it is possible that few studies have reported overlapping patients. Third, the study included only hospitalized patients with a high case fatality, but the routine patients with diabetes who went unnoticed could not be taken into account. This might have led to the overestimation of diabetes-associated mortality in studies. Fourth, the study design in most of the studies was retrospective case-control, which might cause an attrition bias. Future prospective and randomized studies must be conducted to annul the confounding bias. Last, the research articles that were not free were excluded from the study.
5. Conclusions
In conclusion, the results of meta-analysis studies published worldwide during 2020 showed that the presence of diabetes is associated with a significantly increased risk of mortality in patients admitted to the hospital with COVID-19. The pooled OR for diabetes-associated mortality was 1.83, which was significantly higher than the overall COVID-19 mortality. Thus, considering the vulnerability for adverse outcomes, the subpopulation of diabetes must be continuously monitored for glycemic levels, coagulation abnormalities, and inflammatory surge in the event of COVID-19 admission. Besides, hospitals must stick to a strict diabetes management protocol for better care of COVID-19 patients.