1. Background
Thyroid hormones are essential in the growth and maintenance of hair follicles (1, 2). Therefore, thyroid disorders can cause hair loss in the scalp, eyebrows, and other body parts (3, 4). Hypothyroidism can be seen in any age group, predominantly in middle-aged and older adults. It can change many metabolic processes in the body (5, 6). Although hypothyroidism and severe or prolonged hyperthyroidism can cause hair loss, previous studies have shown that changes in the hair cycle do not cause hair loss in patients with thyroid disorders. It occurs probably due to a disturbance in the quality of the new hair (7, 8).
Patients with hypothyroidism experience dry, coarse, slow-growing hair with diffuse and partial alopecia of the scalp and lateral eyebrows. As the higher percentage of hair in hypothyroidism in the telogen phase may be reversed by treatment (1, 9), early diagnosis of hair loss and subsequent treatment of the underlying etiology has clinical importance for the patients. There are many challenges in the early diagnosis of hair loss in patients with hypothyroidism. Common diagnostic methods that assess hair loss may be invasive (e.g., biopsy) or non-invasive (e.g., hair count and pull test).
Differentiating inflammatory versus non-inflammatory processes can have clinical significance since each has a different diagnostic/therapeutic approach. The biopsy is the gold standard for the diagnosis of inflammation. However, incisional biopsies are invasive and costly procedures. Therefore, it seems that dermoscopy, as a non-invasive and inexpensive tool, can be used by experienced dermatologists (10). Dermoscopy provides a better assessment of the epidermis, upper dermis, superficial vascular structures, and pigmentations with 10 to 50 times magnification by connecting a light source to a set of lenses. In addition to pigmented lesions, it is used to evaluate non-pigmented lesions, inflammatory processes, and alopecia (8, 11, 12).
Numerous studies have evaluated the relationship between thyroid disorders and hair loss (1, 7, 9). Also, few studies have examined dermoscopic findings in patients with certain types of alopecia (8, 11, 12). Still, no study has evaluated the dermoscopic results in patients with hypothyroidism and hair loss.
2. Objectives
This study aimed to determine the frequency of dermoscopic findings in hypothyroidism and compare it with euthyroidism. The result of this study can be used for early diagnosis and possible treatments in this group.
3. Methods
3.1. Study Design
This analytic cross-sectional study was performed on 82 patients with hypothyroidism referred to Razi and Besat dermatology clinics of Guilan University of Medical Sciences, Iran. Hypothyroid patients with the chief hair loss complaints were enrolled and compared with an equal number of euthyroid individuals with androgenetic hair loss. The diagnosis of hair loss was based on patient complaints and dermatologist physical examination. An expert endocrinologist diagnosed subclinical and overt hypothyroidism. Subclinical hypothyroidism was indicated by TSH levels of 4.5 - 10 mg/dL and normal free T4. Besides, overt hypothyroidism was noted by TSH of more than 10 and any level of free T4. They were treated with the appropriate dose of levothyroxine. In the euthyroid group, androgenetic alopecia was indicated as the probable etiology for patients referred to dermatology clinics with the chief complaint of alopecia.
Exclusion criteria were the history of scalp trauma, receiving drugs such as topical/systemic steroids, biologic, chemotherapeutic, immunosuppressive, and anticonvulsant (valproic acid/carbamazepine/phenytoin) agents over the past six months, and vasoactive, calcium channel blockers, beta-blockers, nitroglycerin, and retinoids in the past month. Also, we excluded patients with pregnancy, inflammatory diseases such as psoriasis and seborrheic dermatitis, Lichen Planopilaris, Discoid Lupus Erythematosus, hypothyroidism, and euthyroidism who had hair loss due to conditions other than hypothyroidism and androgenic hair loss, abnormal laboratory results (e.g., anemia), and self-reported stress.
Written informed consent was obtained from patients, and ethical approval was obtained from the Ethics Committee of the Vice-Chancellery for Research of Guilan University of Medical Sciences (code: IR.GUMS.REC.1400.075). In addition to the routine self-reported chronic diseases, all patients underwent routine laboratory tests, including liver function tests, lipid profile, fasting blood sugar, blood sugar, and hemoglobin A1C before enrollment. Hormonal assessments were done for girls with irregular menstruation to rule out polycystic ovarian syndrome. Therefore, we did not include patients with abnormal results.
After recording the demographic and clinical characteristics of patients, including sex, age, hypothyroidism duration, and hair loss duration in a checklist, all patients were subjected to hair dermoscopy by a single experienced dermatologist based on five categories of possible hair abnormalities, including hair shaft disorders, follicular opening disorders, scales, pigmentation disorders, and vascular disorders. The hairline and one spot in every of the four head quadrants were evaluated by hair dermoscopy (Table 1). To determine head quadrants, we divided the head into four sections based on the perpendicular division of sagittal to coronal lines.
| Category | Findings |
|---|---|
| Hair shaft disorders | Coiled and twisted (pili torti-like), Regrowing, Vellus hair, Tufting, Corkscrew, Comma |
| Follicular opening disorders | White dots (Pinpoint, Fibrotic); Yellow dots (Large, Small); Black dots, Red dots |
| Scale disorders | Diffuse, Scattered, Perifollicular (tubular) |
| Pigmentation disorders | Honeycombing, Interfollicular scattered brown/black pigments, Peripilar dark halo, Pinpoint speckled brown-black |
| Vascular disorders | Thick arborizing, per follicular concentric, Interfollicular red loops, Plexiform and telangiectatic |
Categories of Dermoscopic Findings
3.2. Statistical Analysis
Data were analyzed using SPSS version 16 software (Chicago, IL, USA). Descriptive characteristics of qualitative variables were reported by frequency and percentage. The Shapiro-Wilk test was used to determine the normality of data distribution. Continuous variables were demonstrated as mean ± SD. The t test was used to compare quantitative variables, and the chi-square test to compare categorical variables. We used multiple logistic regression to detect predictors.
4. Results
This study evaluated 164 patients (82 hypothyroid patients with hair loss complaints and an equal number of euthyroid individuals with androgenetic hair loss). The mean age was 35.55 ± 13.67 years. No significant difference was detected between the groups in terms of age, sex, and hair loss duration (< 0.668, 0.599, and 0.951, respectively) (Table 2).
Demographic Characteristics of Patients in Two Groups
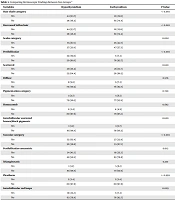
The presence of hair shaft abnormalities significantly differed between the groups (P < 0.001). More specifically, the decrease in vellus hair (P < 0.001) was significantly different between the two groups. Both groups' most common pigmentation disorders were honeycombing and scattered interfollicular pigmentations. However, the difference was not significant between the two groups (P = 0.720). Hypothyroid patients were found to have more scale abnormalities (P = 0.002), especially around hair follicles (perifollicular type) (P < 0.001). Vascular changes (P < 0.001), perifollicular concentric vessels (P = 0.012), and interfollicular red loops (P = 0.005) were significantly higher in patients with hypothyroidism than in those with euthyroidism. The results showed no patients with follicular opening disorders (Table 3).
| Variables | Hypothyroidism | Euthyroidism | P-Valueb |
|---|---|---|---|
| Hair shaft category | < 0.001 | ||
| Yes | 44 (53.7) | 21 (25.6) | |
| No | 38 (46.3) | 61 (74.4) | |
| Decreased Vellus hair | < 0.001 | ||
| Yes | 44 (53.7) | 21 (25.6) | |
| No | 38 (46.3) | 61 (74.4) | |
| Scales category | 0.002 | ||
| Yes | 55 (67.1) | 35 (42.7) | |
| No | 27 (32.9) | 47 (57.3) | |
| Perifollicular | < 0.001 | ||
| Yes | 32 (39.0) | 6 (7.3) | |
| No | 50 (61.0) | 76 (92.7) | |
| Scattered | 0.620 | ||
| Yes | 29 (35.4) | 26 (31.7) | |
| No | 53 (64.4) | 56 (68.3) | |
| Diffuse | 0.576 | ||
| Yes | 8 (9.8) | 6 (7.3) | |
| No | 74 (90.2) | 76 (92.7) | |
| Pigmentation category | 0.720 | ||
| Yes | 3 (3.7) | 5 (6.1) | |
| No | 79 (96.3) | 77 (93.9) | |
| Honeycomb | 0.682 | ||
| Yes | 2 (2.4) | 4 (4.9) | |
| No | 80 (97.6) | 78 (95.1) | |
| Interfollicular scattered brown/black pigments | 0.620 | ||
| Yes | 1 (1.2) | 3 (3.7) | |
| No | 81 (98.8) | 79 (96.3) | |
| Vascular category | < 0.001 | ||
| Yes | 52 (63.4) | 27 (32.9) | |
| No | 30 (36.6) | 55 (67.1) | |
| Perifollicular concentric | 0.012 | ||
| Yes | 34 (41.5) | 19 (23.2) | |
| No | 48 (58.5) | 63 (76.8) | |
| Telangiectatic | 0.210 | ||
| Yes | 1 (1.2) | 5 (6.1) | |
| No | 81 (98.8) | 77 (93.9) | |
| Plexiform | > 0.999 | ||
| Yes | 2 (2.4) | 2 (2.4) | |
| No | 80 (97.6) | 80 (97.6) | |
| Interfollicular red loops | 0.005 | ||
| Yes | 19 (23.2) | 6 (7.3) | |
| No | 63 (76.8) | 76 (92.7) |
Comparing Dermoscopic Findings Between Two Groups a
Also, based on the multiple logistic regression model adjusted for age, sex, and hair loss duration as possible confounding factors, the chances of hair shaft disorders, scales, and vascular abnormalities increased by 2.02, 2.73, 3.53, respectively, in the hypothyroid group compared to the euthyroidism group.
5. Discussion
Alopecias can be categorized based on pathogenesis into inflammatory and non-inflammatory types. Based on our results, we found more dermoscopic signs of inflammation such as hair shaft abnormalities (decreased vellus hair), scale abnormalities (particular perifollicular type), and vascular changes (perifollicular concentric vessels and interfollicular red loops) in patients with hypothyroidism compared to those with euthyroidism. In inflammatory cases (e.g., alopecia areata and lichen planopilaris), the consequent injury was the leading cause of hair loss. Alopecia would be irreversible in case of severe damage. On the other hand, non-inflammatory alopecias (e.g., androgenetic alopecia and telogen effluvium) are usually due to alterations in the hair growth cycle, like prolonging telogen, and shortening of anagen phases (13, 14). It is noteworthy that in non-inflammatory alopecias, vellus hair replaces terminal hair initially. They are seen more significantly than usual, and we do not expect to see signs of inflammation around hair follicles. However, in some inflammatory alopecias like lichen planopilaris or discoid lupus erythematosus, increased vascular markings, pigmentary deposition, scaling around follicular openings, and late stages of milky red areas are attributed to scar tissue replacing normal follicles. Sweat gland openings are also observed (12).
Dermoscopy is commonly used to evaluate hair abnormalities in different clinical settings, which enables clinicians to detect hair loss and contributing etiologies earlier and more accurately (15). Also, it warrants proper time and location for biopsy in patients needing histopathologic confirmation. It, therefore, avoids long-term and irreversible consequences (16).
Signs of inflammation around follicles can be detected by dermoscopy. The presence of pigmentation and dilatation of perifollicular vessels and scaling and loss of vellus hair are usually seen in inflammatory alopecias. On the other hand, these changes are generally not expected in non-inflammatory cases (12).
Thyroid hormones significantly affect hair growth and directly affect human hair follicle functions, including anagen prolongation and stimulation of both hair matrix keratinocyte proliferation and hair pigmentation (9). Hypothyroidism can cause non-inflammatory alterations in the growth cycle (prolongation of the telogen phase) (17). Therefore, hypo/hyperthyroid patients suffer losses and changes in hair color and texture (1). Classically, hypothyroid patients have coarse lusterless sparser hair with loss of the lateral third of eyebrows (18). Since alopecia in hypothyroidism is believed to be non-inflammatory, alopecia should be reversed after thyroid hormone replacement. On the contrary, and not uncommonly, alopecia remains despite normal serum thyroid hormone levels. This observation may point to other possible inflammatory processes (17).
To the best of our knowledge, there is no published article about the dermoscopic evaluation of hair loss in hypothyroidism patients. Therefore, we cannot compare our results with published literature data. This is a limitation of our study and, at the same time, shows the novelty of this study. Our results showed decreased vellus hair, concentric perifollicular, and interfollicular telangiectasias pointed to the possibility of underlying inflammatory processes. The persistence of alopecia, even after treatment initiation and thyroid function normalization, may also be attributed to inflammation. These findings have to be confirmed with histopathology in future studies. Still, it can be suggested that if inflammatory pathogenesis is proven pathologically, topical and systemic corticosteroids may have a role in treating alopecia in hypothyroidism.
5.1. Conclusions
Regarding the promising results of this study, we could detect possible dermoscopic signs of inflammation in hypothyroid patients with hair loss. Further investigations are needed because there is a shortage of evidence on this novel diagnostic method.