1. Background
Production and bioavailability of testosterone in males decrease as their age progresses (1). There is a parallel elevation in low-grade inflammation, as reflected by increased pro-inflammatory cytokines, in aging males (2). Previous studies suggested that testosterone exerts immunosuppressive effects on the body (3). Testosterone could downregulate the expression of toll-like receptor 4 (TLR4), a trigger for inflammation, in macrophages in vitro (4). However, the importance of the relationship between inflammation and testosterone deficiency is not limited to the field of immunology. This is because both testosterone deficiency and low-grade inflammation are implicated in various health conditions, such as sarcopenia, arthritis, osteoporosis, and cardiovascular diseases (5-8).
The association between testosterone level and inflammation is explored in human observational studies (9-11), clinical trials (12, 13), and animal studies (14-16). Significant inverse relationship between testosterone levels and certain inflammatory cytokines was observed in young and elderly males (9, 10). At the same time, no significant difference in inflammatory cytokine levels was found between the male under androgen deprivation therapy and the ones not receiving the therapy (12). Both positive and negative findings are reported in trials studying the suppressive effects of testosterone on inflammatory cytokines (12, 13). The heterogeneity of the results could be due to the subjects and the dose of testosterone used. A preclinical model could be useful in studying the various medical conditions with low-grade inflammation induced by testosterone deficiency.
The current study determined the effects of testosterone deficiency and supraphysiological supplementation of testosterone on inflammatory cytokine levels in orchidectomized rats. Orchidectomy is the most common method of inducing testosterone deficiency in animal. Testosterone has a half-life of 2 days in rats (17). Eight weeks after orchidectomy, depletion of testosterone in rats affected non-sexual organs sensitive to sex hormone, such as muscle and bone (18, 19). It was hypothesized that testosterone deficiency would cause a state of low-grade inflammation in orchidectomized rats, and testosterone supplementation would reverse this effect.
2. Methods
The study protocol was reviewed and approved by animal ethics committee of Universiti Kebangsaan Malaysia (approval code: FP/FAR/2012/IMA/18-JULY/445-JULY-2012-APRIL-2014). Eighteen 3-month-old Sprague-Dawley rats were obtained from the Laboratory Animal Resource Unit, Universiti Kebangsaan Malaysia (Kuala Lumpur, Malaysia). They were housed at the animal laboratory of department of pharmacology under standard conditions (27°C, ambient humidity, natural dark/light cycle, standard rat chow as food and tap water ad libitum). They were acclimatized for 1 week before being randomized equally into 3 groups, ie, sham-operated (SH) (n = 6), orchidectomy (ORX) (n = 6) and testosterone enanthate-treated (TE) groups (n = 6). The ORX and the TE groups underwent bilateral orchidectomy under general anesthesia (ketamine/xylazine at 100 mg/kg: 10 mg/kg, i.p). The SH group underwent similar surgical stress, but their testes were retained. The TE group received weekly intramuscular testosterone enanthate injection (7 mg/kg) diluted in peanut oil, while, the other groups received equivolume of peanut oil injection per se. After 8 weeks of treatment, the rats were euthanized by ketamine/xylazine (300 mg/kg: 30 mg/kg; i.p) and their blood was collected via cardiac puncture in plain tubes.
Blood was centrifuged at 3000 rpm for 10 minutes at 4°C to separate serum. The serum was stored at -70°C until analysis. The levels of interleukin-1 alpha (IL-1α), interleukin-1 beta (IL-1β), interleukin-6 (IL-6) and tumor necrosis factor alpha (TNF-α) were determined using Procarta® multiplex immunoassay (Affymetrix, Santa Clara, USA). This assay could quantify all 4 cytokines simultaneously, using florescent-dyed microspheres (beads) conjugated with antibodies specific to each cytokine. The beads would pass through laser beams in a flow cytometer. A Luminex® analyzer (Bio-Rad, Hercules, USA) was used to determine the luminescence proportional to each of the analysts in the assay. Antibodies specific to the rat were used. Testosterone level was measured using a conventional enzyme-linked immunoassay (ELISA) kit coated with antibodies specific to rat testosterone (Cusabio, Wuhan, China). Quantification was based on the competition inhibition reaction between the antibody with unlabeled testosterone and horseradish peroxidase-conjugated testosterone. The intra-assay coefficient of variation was 3.9% for IL-1α, 4.0% for IL-1β, 3.3% for IL-6, 1.2% for TNFα, and < 15% for testosterone. The inter-assay coefficient of variation (CV) was 3.3% for IL-1α, 3.7% for IL-1β, 2.5% for IL-6, 4.0% for TNFα, and < 15% for testosterone. The detection limit for each assay was 0.1 pg/mL for IL-1α, 0.5 pg/mL for IL-1β, 6.5 pg/mL for IL-6, 0.7 pg/mL for TNFα, and 0.13 ng/mL for testosterone. Testosterone depletion was considered successful when a statistically significant difference was observed in testosterone level between SH and ORX rats.
Statistical analysis was performed using statistical package for social sciences (SPSS) version 20 (IBM, Armonk, USA). Normality of the data was assessed using the Shapiro-Wilk test. Comparison of testosterone and inflammatory cytokine levels between the 3 groups was performed using one-way analysis of variance (ANOVA) with Tukey's (homogeneity assumed) or Dunnett's T-3 (homogeneity not assumed) post-hoc pair-wise comparison. Data were expressed as mean ± standard error. Statistical significance was set at P < 0.05.
3. Results
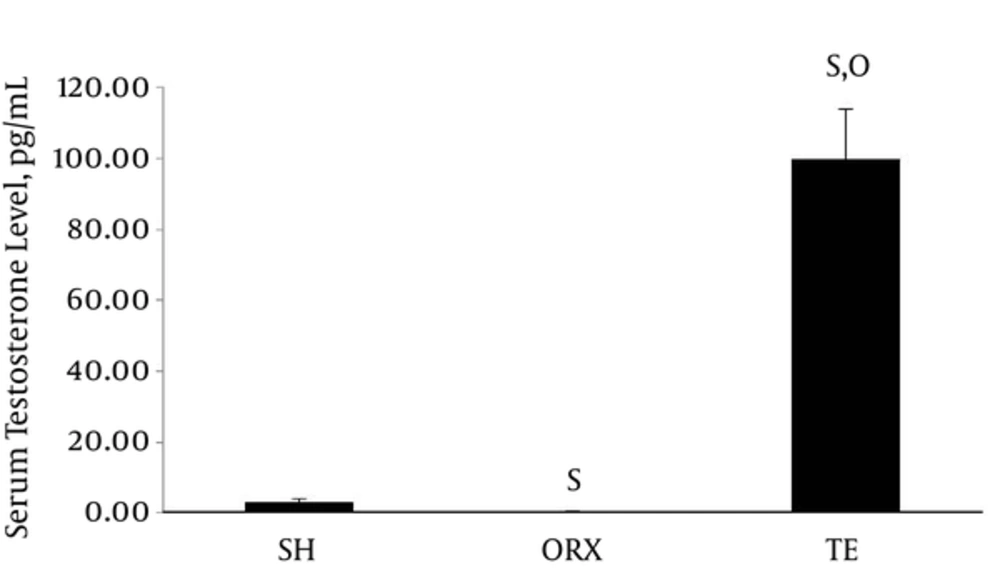
The testosterone level in the TE group (99.86 ± 14.00 ng/mL) was significantly higher compared to those of the SH (3.21 ± 0.98 ng/mL) and ORX groups (0.35 ± 0.02 ng/mL) (P < 0.001). The SH group also had higher serum testosterone level compared to the ORX group (P = 0.023) (Figure 1).
Data are presented as mean ± standard error. The number of rats per group is 6. Letter “s” indicates a significant difference (P < 0.05) compared to sham group, “o” compared to orchidectomized group. Abbreviation: SH = sham group; ORX = orchidectomized group; TE = testosterone enanthate-treated group.
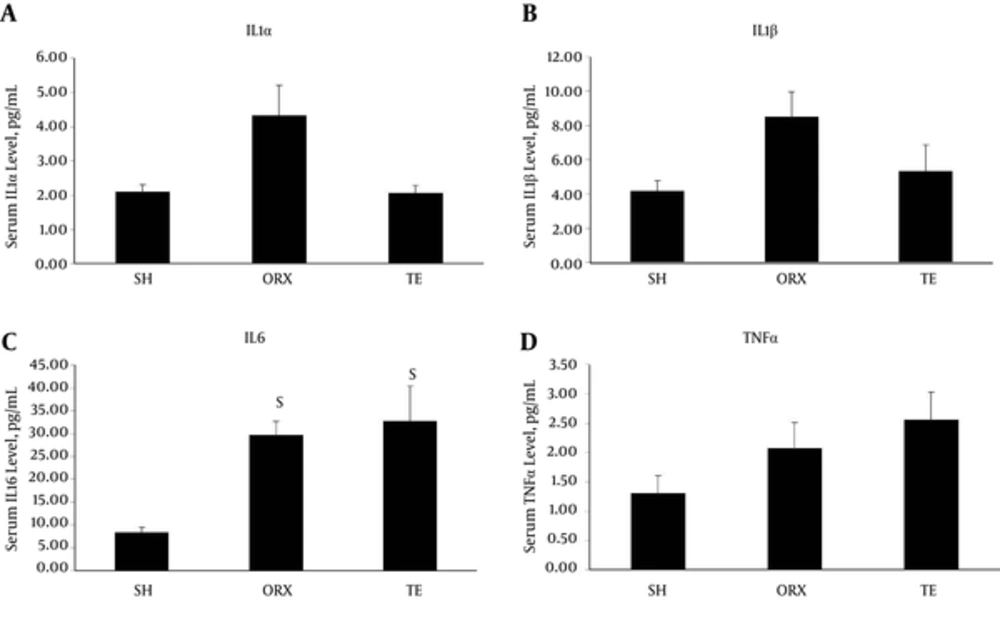
The IL-1α (4.33 ± 0.87 pg/mL for ORX vs. 2.11 ± 0.20 pg/mL for SH; P = 0.131) and β levels (8.50 ± 1.46 pg/mL for ORX vs. 4.18 ± 0.60 pg/mL for SH; P = 0.071) appeared to have elevated in the ORX group, but the increase was not statistically significance. The TE and SH groups shared similar levels of IL-α (2.07 ± 0.23 pg/mL for TE vs. 2.11 ± 0.20 pg/mL for SH; P = 0.998) and β (5.35 ± 1.53 pg/mL for TE vs. 4.18 ± 0.60 pg/mL for SH; P = 0.796) (Figure 2A and B). The level of IL-6 significantly increased in the ORX group (26.69 ± 3.04 pg/mL) compared to the SH group (8.35 ± 1.19 pg/mL) (P = 0.017) (Figure 2C). Administration of supraphysiological testosterone did not suppress the elevation of IL-6 (32.79 ± 2.06 pg/mL; P = 0.891 vs. ORX). TNFα appeared to increase with orchidectomy, albeit not statistically significant (2.08 ± 0.44 pg/mL for ORX vs. 1.30 ± 0.30 pg/mL for SH; P = 0.401), and testosterone replacement did not suppress this increase (2.56 ± 0.47 pg/mL for TE; P = 0.692 vs. ORX) (Figure 2D).
4. Discussion
The results of the current study indicated that orchidectomy induced a significant increase in IL-6 in male rats. The levels of TNFα, IL-1α, and IL-1β also elevated in orchidectomized rats, but the differences with those of sham were not statistically significant. The current study observations suggested that compared to other cytokines, IL-6 was the most responsive to changes in testosterone level in vivo. The current study observations were similar with the findings of Freeman et al. They reported that orchidectomized rats with subphysiological testosterone level showed marginal elevated interleukins and TNFα levels compared to those of age-matched rats, but it was not statistically significant (14). In contrast, a study by Steffens et al. showed no significant differences in serum and tissue IL-6 levels between orchidectomized and sham rats (16, 19). However, orchidectomized rats with ligature-induced inflammation showed significantly higher tissue IL-1β level compared to ligated sham rats in their study (16). It suggested that testosterone deficiency by itself might not induce strong inflammatory response, but it might exacerbate pre-existing inflammatory conditions. This is in accordance with the immunosuppressive actions of testosterone (4).
In the current study, administration of supraphysiological testosterone appeared to prevent the elevation levels of IL-1α, IL-1β, but did not prevent the increase in TNFα and IL-6. Similarly, Freeman et al. also did not observe significant differences in cytokine levels between orchidectomized rats supplemented with physiological (2.5 mg) and supraphysiological (35 mg) testosterone for 4 weeks (14). Steffens et al. also found no significant differences in serum and tissue IL-6 levels between orchidectomized rats and the ones receiving high-dose testosterone (250 mg/kg testosterone esters weekly) (16). These previous animal studies as well as the current one indicated that exogenous testosterone supplementation might not alter the cytokines production in the body. On the other hand, Mendes et al. showed that the combination of ethanol consumption and testosterone treatment (testosterone cypionate 5 mg/kg every other day for 4 weeks) caused a significant increase in TNFα, but not IL-6 in rats (15). However, each compound alone did not significantly change the level of cytokines in rats (15). The results of the study by Mendes et al. pointed out that exogenous testosterone supplementation might aggravate the inflammatory properties of other compounds.
In vitro studies showed that testosterone could modify the quantity of cytokines and the peripheral cells producing it. Corcoran et al. showed that physiological (10 nM) and pharmacological (100 nM) testosterone levels significantly reduced the gene and protein expression of TNFα in macrophages derived from males and females (20). Corrales et al. reported that after testosterone treatment, the number of monocytes and dendritic cells producing IL-1, IL-6, and TNFα in aging type 2 diabetic males with partial androgen deficiency became undetectable by flow cytometry (21). However, these changes might not be significant enough to be detected in vivo as the case of the current study.
The current study had some limitations. Sexually matured young rats were used, although aged rats might be better representatives of elderly males. However, orchidectomy ensured that the rats experienced testosterone deficiency that was similar to hypogonadal human subjects. It was a preliminary study on the effects of testosterone on inflammation in an animal model. Only a limited number of cytokines, all of them pro-inflammatory, were evaluated in the current study. However, they are established markers of inflammation involved in the pathogenesis of various health conditions. The mechanistic studies on the effects of testosterone on the quantity of cytokines and their producing cells should be evaluated in further studies. The apparent elevation in cytokine level, which did not reach statistical significance, might be due to small sample size and low power. This could be improved by increasing the number of animal and duration of treatment. Further studies are needed to validate and strengthen the results.
In conclusion, testosterone deficiency increases the pro-inflammatory cytokine, IL-6, but its effects on other cytokines are marginal. High-dose testosterone does not alter the level of inflammatory cytokines. Testosterone deficiency might not be a good model for low-grade inflammation. Further improved studies are needed to validate the association between testosterone deficiency and inflammation.