1. Background
Alterations in maternal thyroid hormone secretion are common in pregnancy. Both hyper- and hypothyroidism during pregnancy are associated with several adverse outcomes, in both mother and fetus (1, 2). Optimal treatment of maternal thyroid dysfunction is important, to achieve a successful pregnancy outcome (2, 3). Despite the publication of the latest two guidelines by the American Thyroid Association (ATA) and the Endocrine Society (ES) for the management of thyroid diseases, in pregnancy (3, 4), the proportion of clinicians following these guidelines in their routine clinical practice is unknown; however, it is unclear whether practices of clinicians differ among endocrinologists, internists and general practitioners (GPs). Patients with thyroid disorders are managed by both endocrinologists and general practitioners and may be more optimally managed with the guidance of specialists. Recent surveys have been published on the management of hypo- and hyperthyroidism during pregnancy, involving 321 Asian endocrinologists from 21 Asian countries, which indicated a high accordance of the management to the recent ATA guidelines, despite wide variation in management between individuals (5, 6).
2. Objectives
The present study was undertaken to compare the practices of endocrinologists and generalists in the management of thyroid dysfunction in pregnancy, considering the latest guidelines of ATA and ES.
3. Materials and Methods
During the November 13 - 16, 2013 meeting of the Asean federation of endocrine societies (AFES) in Jakarta, Indonesia, questionnaires were distributed to endocrinologists, internists and general practitioners (GPs) attending the meeting. The survey questionnaire was based on clinical case scenarios, posing questions about the clinical practices related to screening for thyroid dysfunction and the management of hypothyroidism and hyperthyroidism in pregnancy. The survey questionnaire has been validated by previous studies in Europe (7). In total, there were 25 multiple choice questions on the screening of thyroid function and management of hyper- and hypothyroidism, during pregnancy. The responders were allowed to provide their own answer, if it was not included in the questionnaire choices given.
All frequencies were adjusted on a 100% basis, excluding the non-responders and the results have been predominantly presented in the text and tables, as percentages, rounded up to a whole number. To compare the distribution of responders between group 1 (endocrinologists) and group 2 generalists (internists + GPs), the chi-square for goodness - of - fit test was used (Tables 1 to 6). Two sample proportion tests were used for comparison of proportions of the correct answers between endocrinologists and generalists (Figure 1).
| Recommendation | Responders, % | P Value | |
|---|---|---|---|
| Endocrinologists | Generalists | ||
| Seeking pre-pregnancy counseling | 0.009 | ||
| PTU | 43 | 55 | |
| MMI (CMZ) | 49 | 45 | |
| Refer for definitive treatment with surgery before pregnancy | 4 | -- | |
| Refer for definitive treatment whit radioiodine before pregnancy | 4 | -- | |
| At 8 weeks of pregnancy | < 0.001 | ||
| PTU | 58 | 39 | |
| PTU in the first treatment, then MMI (CMZ) | 33 | 51 | |
| MMI (CMZ) | 9 | 9 | |
Treatment Options in a Woman Diagnosed With Graves’ Disease a
| Tests | Responders, % | |
|---|---|---|
| Endocrinologists | Generalists | |
| Tests use to monitor b | ||
| TSH and FT4 | 43 | 51 |
| TSH , FT4 and FT3 | 30 | 34 |
| FT4 alone | 14 | 9 |
| TSH, TT4, TT3 | 4 | 3 |
| TSH and TT4 | 5 | 3 |
| Others | 4 | 0 |
| Targets c | ||
| Low TSH and FT4 (or TT4) in the upper end of normal range | 71 | 26 |
| TSH and FT4 (or TT4) in the normal range | 22 | 53 |
| Low TSH and FT4 (or TT4) in the normal range | 5 | 6 |
| Others | 2 | 15 |
Different Tests Responders use to Monitor Antithyroid Drugs Dose in Pregnancy and Targeted Thyroid Hormone Values a
| Recommendation | Responders, % | |
|---|---|---|
| Endocrinologists | Generalists b | |
| Follow up, no treatment | 16 | 15 |
| PTU in the first trimester, then change to MMI/CMZ | 45 | 43 |
| PTU | 32 | 36 |
| MMI/CMZ | 3 | 6 |
| Others | 4 | 0 |
Responders’ Recommendations for Treatment or Follow up of an 8 Weeks Pregnant Woman With Possible Gestational Thyrotoxicosis a
| Recommendation | Responders, % | |
|---|---|---|
| Endocrinologists | Generalists b | |
| Start MMI/CMZ and continue lactation | 61 | 57 |
| Start PTU and continue lactation | 22 | 20 |
| Start MMI/CMZ and stop lactation | 10 | 12 |
| Start PTU and stop lactation | 5 | 11 |
| Others | 2 | 0 |
Responders’ Recommendations for a Postpartum Lactating Woman With Graves’ Hyperthyroidism a
| Starting dose of L-T4 | Responders, % | |
|---|---|---|
| Endocrinologists | Generalists b | |
| Start on a small dose (e.g. 25 - 50 μg daily) | 23 | 53 |
| Start on a full dose (e.g. 100 - 125 μg daily) | 33 | 3 |
| Start on a dose based on pregnancy adopted body weight | 23 | 17 |
| Start for a few days on a double dose (e.g. 200 μg daily), followed by a dose based on pregnancy adapted body weight | 18 | 20 |
| Start on a dose based on pre-treatment TSH level | 3 | 7 |
Starting Dose of Levothyroxine in a Pregnant Woman Diagnosed With Overt Hypothyroidism at 12 Weeks Gestation a
| Tests | Responders, % | P Value | |
|---|---|---|---|
| Endocrinologists | Generalists | ||
| Monitor T4 dose | < 0.001 | ||
| TSH alone | 25 | 3 | |
| TSH, FT4 | 25 | 53 | |
| TSH, FT4, FT3 | 15 | 30 | |
| TSH, FT4, TT3 | 6 | 3 | |
| TSH and TT4 | 12 | 7 | |
| Trimester specific TSH ranges | 17 | 4 | |
| Screen pregnant women | < 0.001 | ||
| TSH alone | 18 | 18 | |
| FT4 | 1 | 9 | |
| TT4 | 0 | 1 | |
| TSH and FT4 or TT4 | 60 | 61 | |
| FT3 or TT3 | 0 | 9 | |
| TSH and thyroid antibodies | 21 | 2 | |
| Urinary iodine excretion | 0 | 0 | |
Different Tests Responders use to Monitor Levothyroxine Dose and to Screen Thyroid Dysfunction in Pregnancy a
4. Results
4.1. Characteristics of Responders
We received 112 responses from eight countries. Five responders were not involved in the management of thyroid diseases in pregnancy and were not included in the current analysis. Therefore, responses from 107 responders were analyzed. There were 74 (69%) endocrinologists and 33 (31%) internists and general practitioners. The countries included: Indonesia, Philippines, Malaysia, Singapore, India, Thailand, Sri Lanka and Myanmar.
4.2. Management of Hyperthyroidism in Pregnancy
Physicians were questioned about the treatment of thyrotoxicosis in a 26-year-old woman, newly diagnosed with Graves’ disease, who wishes to become pregnant. Responders suggested methimazole (MMI)/carbimazole (CMZ), propylthiouracil (PTU), surgery or radioiodine treatments (Table 1). Only 8% of endocrinologist recommended ablative treatment before pregnancy, 4% surgery and 4% radioiodine therapy. The reminder, 92% of endocrinologists and all generalists, would treat this case with antithyroid medications. Compared to endocrinologists, generalists use PTU more than MMI, the difference was statistically significant (P <0.001).
Regarding the treatment of thyrotoxicosis, in a 24 -year-old pregnant woman newly diagnosed with Graves’ disease at 8 weeks of pregnancy, 91% of participants preferred treatment with PTU, of these, 33% of endocrinologists and 51% of generalists switched to MMI/CMZ in the second trimester (Table 1); more endocrinologists (58%) used PTU throughout pregnancy, as compared to generalists (39%).
There was inconsistency in responders’ recommendations on which test to use to monitor the dose of antithyroid drugs; 43% of endocrinologists and 51% of generalists chose thyroid stimulating hormone (TSH) and free thyroxin (FT4) measurement, respectively; 30% and 34% preferred TSH, FT4 and free triiodothyronine (T3), respectively; and 14% and 9% used FT4 alone, respectively. Very few remaining responders used the total forms of thyroid hormones or other combinations. However, the differences between the responses of both groups were not significant (Table 2).
When asked “What are the targeted thyroid test results you aim to achieve with antithyroid drugs in pregnancy?”, 71% of endocrinologists and 26% of generalists aimed to attain low serum TSH and FT4 (or total T4) in the upper end of the normal range (P <0.001); however, 53% of generalists and 22% of endocrinologists aimed to maintain serum TSH and FT4 (or total T4) within the normal range. The remainder used other combinations (Table 2).
Responders were asked about the treatment of a 24-year-old, 8 week pregnant woman with nausea, vomiting, weight loss and palpitation, who had negative thyroid antibodies and thyroid function tests in the hyperthyroid range. Nearly 77% of endocrinologists and 79% of generalists preferred treatment with PTU (Table 3); of these, 45% and 43% would change to MMI/CMZ after first trimester, respectively.
Forty-six percent of endocrinologists and 41% of generalists did not routinely check TSH receptor antibodies (TRAb) in pregnant women with Graves’ disease, treated with antithyroid drugs. The remainder would check TRAb in the first trimester and of these, 32% of endocrinologists and 19% of generalists would repeat TRAb measurement in the third trimester only, if it was positive in the first trimester.
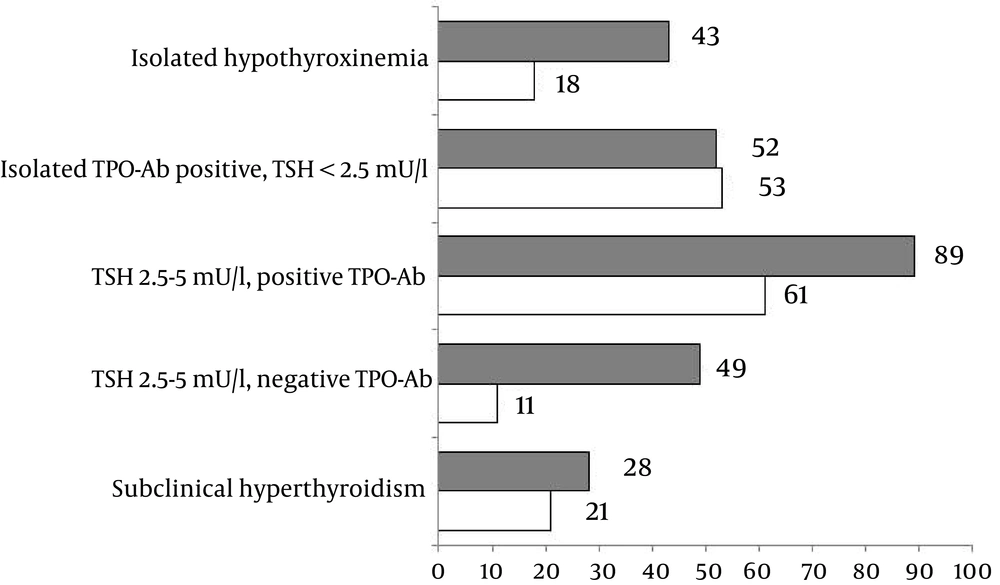
Participants were then asked whether they would recommend treatment or follow up of subclinical hyperthyroidism in pregnancy. About 72% of endocrinologists and 79% of generalists preferred follow up, while the rest chose treatment (Figure 1).
Responders’ approaches to ultrasonography monitoring of fetus in antithyroid treated pregnant Graves’ disease varied greatly. Nearly 72% of endocrinologists and 56% of generalists routinely monitor the fetus with ultrasound scan, while 18% and 25%, respectively, would do so, only when the mother is positive for TRAb, and the remainders indicate no ultrasound monitoring.
4.3. Management of Postpartum Hyperthyroidism
There was inconsistency in responders’ recommendations on how to manage a postpartum lactating woman with relapse of Graves’ hyperthyroidism. Eighty-one percent of endocrinologists and 77% of generalists would start antithyroid medication (61% and 57% preferring MMI, respectively) with continuing lactation; however, 17% and 23%, respectively, would start antithyroid drugs and suggest stopping lactation, the difference being significant (P = 0.040) (Table 4).
4.4. Treatment of Hypothyroidism in Pregnancy
Physicians asked what dose of levothyroxine (L-T4) would they start, for a 24-year-old, 12-week pregnant woman with overt hypothyroidism, suggested variable doses for T4 replacement. Although most endocrinologists recommended starting on a full replacement dose, empirically or based on pregnancy adapted body weight (Table 5), only 20% of generalists agreed, of whom 53% would start with lower dose of L-T4 (P < 0.001).
Although the majority of responders would not endorse abortion in overt hypothyroid patient, in the late first trimester, 7% of endocrinologists and 13% of generalists would recommend it.
There was inconsistency in responders’ recommendations on how to adjust the dose of L-T4 in a hypothyroid woman planning pregnancy. About 52% of endocrinologists and 80% of generalists would first check thyroid function tests before adjusting the dose. Nearly 33% and 14%, respectively, would recommend increasing the dose of L-T4 by 30%- 50% as soon as pregnancy is confirmed, while 15 and 6% respectively, would increase (L-T4) dose, by two tablets per week, as soon as pregnancy is confirmed, P < 0.001 (Supplement 1).
There was more inconsistency regarding tests chosen to monitor the dose of (L-T4) in pregnancy among endocrinologists, compared to generalists. Eighty-three percent of the latter group used TSH + FT4 or TSH + FT4+ FT3 for this purpose (Table 6), indicating that generalists choose more tests to monitor the dose of L-T4, as compared endocrinologists (P < 0.001).
The results of targeted thyroid function test that responders aimed to achieve with L-T4 in pregnancy were also inconsistent, although the majority of responders (63%) aimed at keeping TSH < 2.5 mU/L in the first trimester and < 3 mU/L in later trimesters (57% endocrinologists and 50% generalists), 32% and 27% targeted TSH and FT4 within trimester-specific reference ranges, and 10% and 23% targeted TSH and FT4 within laboratory reference ranges, for endocrinologists and generalists, respectively, P = 0.001 (Supplement 3).
4.5. Screening Pregnant Women for Thyroid Dysfunction
For no systematic screening, targeted screening of pregnant women at high risk of thyroid dysfunction and universal screening of all pregnant women, endocrinologists voted 11, 73 and 16%, respectively, while the corresponding values for generalists were 21, 52 and 29% (P <0.001). Endocrinologists were more familiar with risk factors used for carrying out targeted screening of pregnant women for thyroid dysfunction, compared to generalists (Supplement 2).
Responders’ timing for screening thyroid function in pregnancy varied: 54% of endocrinologists and 50% of generalists were supported screening during the first antenatal visit, while 28% and 23% during a pre-pregnancy visit, respectively; 16% and 23% did not have specific timing, respectively. Table 5 shows tests used for screening pregnant women for thyroid dysfunction. About 60% of both groups use TSH and FT4 or TT4. More endocrinologists than generalists order thyroid antibodies (P <0.001). Responders were asked whether they would routinely repeat thyroid function tests during the pregnancy, if the initial screening was normal: 50% of endocrinologists and 69% of generalists would routinely repeat the tests in later pregnancy, while 17% and 23% would repeat only in the presence of thyroid antibodies, whereas 17% and 8%, respectively, would not repeat.
When endocrinologists and generalists were asked when they would start L-T4 replacement, following the screening, they indicated the following criteria, respectively: 53% and 36% would start at a TSH above the trimester-specific reference range; 1% and 11% at a TSH above the population reference range; 325 and 39% at a TSH > 2.5 mU/L; 10% and 11% at a TSH > 5 mU/L, and 4% and 3% at a FT4 below the trimester-specific reference range.
Responders’ approaches to various outcomes of screening thyroid function in pregnancy differed (Figure 1): 43% of endocrinologists and 18% of generalists would treat isolated hypothyroxinemia (P = 0.012); 52% and 53% would treat isolated positive thyroid antibodies with normal TSH, respectively; 61% and 49% would treat TSH level >2.5 (and < 5 mU/L) with positive thyroid antibodies (P < 0.001), respectively, and 49% and 11%, would treat TSH levels > 2.5 (and < 5 mU/L), with negative antibodies and no other thyroid disease, respectively (P < 0.001).
5. Discussion
The present study compares, for the first time, current clinical practices in the management of thyroid disease in pregnancy, between endocrinologists and generalists of East Asia. Generally, there was a high degree of consistency between clinical practices of East Asian physicians and the ATA and ES guidelines (3, 4), conforming more closely to the practices of endocrinologists than generalists. In all issues, except one, there were significant differences in clinical approaches between generalists and specialists.
Maternal hyperthyroidism, diagnosed in pregnancy, must be corrected, because it has detrimental effects on both mother and fetal health (8, 9). Concerning probability of PTU hepatotoxicity (10) which limits its use as the first line treatment of hyperthyroidism and the teratogenic effect of MMI (11), ATA and ES recommend PTU during the first trimester, followed by MMI from the second trimester. In the present survey, although most responders follow the guidelines, there is significant discrepancy between subspecialists and generalists, especially regarding shifting therapy from PTU to MMI after the first trimester, which may be due to their own clinical concerns in switching drugs based on the ES suggestions.
For preconception counseling of a Graves’ patient, ATA (3) recommends the use of MMI/CMZ and change to PTU, once the pregnancy is confirmed (3). However, whether this approach would prevent MMI/CMZ associated embryopathy is not clear. Despite the significant differences to this approach, between generalists and subspecialists, most responders suggest drug therapy. Nevertheless, the majority of responders in both groups, especially generalists, would prefer to start with PTU.
Almost half of the responders monitor antithyroid drug treatment with TSH and FT4 and one third with TSH, FT4 and FT3, with no significant difference between the two groups. In addition, most endocrinologists target low TSH and FT4 (or TT4) in the normal range, during such therapy, which is also considered as good practice, since guidelines recommend TSH and FT4, as the main tests for such monitoring and advise to aim for FT4 in the upper end of the normal range, during antithyroid therapy in pregnancy (3, 4). However, it is distressing that 22% of endocrinologists and 53% of generalists target normal range for serum TSH and FT4 concentrations, since this approach may increase the chance of fetal goiter and hypothyroidism (12).
During the first half of pregnancy, differentiation of Graves’ disease and gestational thyrotoxicosis may be difficult. The presence of clinical evidence of autoimmunity, typical goiter and elevated TRAb support the diagnosis of Graves’ disease, while severe nausea, vomiting, weight loss and palpitation, along with negative thyroid antibodies favor gestational thyrotoxicosis (13). In most cases of gestational thyrotoxicosis, no treatment is indicated, and this approach is followed only by 16% of endocrinologists and 15% of generalists, most of whom, unfortunately, preferred treatment with antithyroid medications, with significant discrepancy between the two groups (Table 3). In a recent survey, 40% of East Asian endocrinologists just followed the pregnant women without intervention and 55% chose antithyroid therapies, with PTU preference (6).
Routine measurement of TRAb, in a pregnant hyperthyroid woman under antithyroid drug therapy, is recommended by major professional endocrine organizations (3, 4). Increased serum TRAb, detected in up to 95% of hyperthyroid pregnant women, is a risk for fetal and neonatal hyperthyroidism and lack of treatment will increase fetal and neonatal morbidity and mortality (14). However, 46% of endocrinologists and 41% of generalists indicated that they do not routinely check TRAb, mainly due to lack of availability of this test.
There are also significant differences in the management of hyperthyroidism in a postpartum lactating woman, between generalists and specialists. The ATA and ES both recommend treating a lactating hyperthyroid woman with MMI, due to PTU hepatotoxicity (15). It has been shown that MMI therapy, up to 30 mg daily, does not cause any alterations in thyroid function or the mental or physical development of infants, breast-fed by lactating hyperthyroid mothers (16, 17). It is unfortunate that 15% of endocrinologists and 23% of generalists recommend stopping lactation during treatment with antithyroid drugs. The results of the recent survey, on the management of hyperthyroidism in pregnancy, by East Asian endocrinologists, were similar to opinions of endocrinologists in the current survey, except for the targeted thyroid hormone value during antithyroid treatment, as previously mentioned (6).
Universal screening for thyroid dysfunction in pregnant women has been intensely debated in recent years (18, 19). Although several studies have shown that the targeted case-finding approach misses a significant proportion of pregnant women with thyroid dysfunction (19, 20), both the ATA and ES recommended targeted screening in pregnancy (3, 4). The association of mild maternal thyroid hormone deficiency in pregnancy with impaired neuropsychological development of the offspring (21) and adverse obstetric outcomes has been reported (22). Only one randomized controlled trial suggests that identification of mild thyroid hormone deficiency in low-risk pregnant women, by screening and treatment with L-T4, may reduce obstetric complications (18). In the present survey, most endocrinologists and half of the generalists, or their institutions, perform targeted screening of only the high-risk group, in accordance with ATA and ES recommendations. It is, however, unfortunate that 21% of generalists and 11% of endocrinologists do not carry out any systematic screening. In addition, endocrinologists were more familiar with risk factors for targeted screening than generalists.
Half of the generalists and 54% of endocrinologists reported that they screened thyroid function during the first antenatal visit, and 23% and 28%, respectively, would do so during the first pre-pregnancy visit. One could argue that the identification and treatment of hypothyroidism in the first antenatal visit may be too late to prevent any associated adverse effects. However, implementing systemic screening of thyroid function in all women planning pregnancy would be an enormous challenge, especially for the developing countries of Asia.
Maternal hypothyroidism diagnosed in pregnancy should be treated (3, 4), because maternal thyroid hormones play an important role in early fetal neurological development (21). In this survey, most endocrinologists initiated full replacement dose of L-T4 for pregnant women newly diagnosed with overt hypothyroidism; however, 53% of generalists started on a small dose of L-T4. It is unfortunate that 7% of endocrinologists and 13% of generalists would recommend the option of abortion in overt hypothyroid patients, despite the absence of any published study to support such a practice. It has been reported that IQs of children, whose mothers had been hypothyroid during early pregnancy, were normal and similar to those of their siblings, who had not been exposed to maternal hypothyroidism in utero (23). In another study, treatment of maternal hypothyroidism did not improve IQ or impaired cognitive function in 3-year-old offspring (24).
Most hypothyroid women need an increased dose of L-T4, from very early pregnancy (25, 26). Indeed, about 25% of women on L-T4 replacement, at their first antenatal visit, show biochemical evidence of under replacement (19), which may be prevented by optimizing the L-T4 dose before pregnancy (27). In the present study, 48% of endocrinologists (and only 20% of generalists) would advise women to increase the dose of L-T4, either by 30% - 50% or by two tablets per week, as soon as pregnancy is confirmed, according to previous recommendations (3, 4, 28). The rest of the responders would check thyroid function, as soon as pregnancy is confirmed.
Regarding the targeted thyroid function for pregnant hypothyroid women, 89% of endocrinologists and 77% of generalists aimed to achieve TSH and FT4 within the trimester-specific reference range or TSH < 2.5 mU/L in the first trimester and < 3 mU/L in later trimesters, as recommended by current guidelines (3, 4).
More endocrinologists than generalists would treat TPOAb positivity, with TSH between 2.5 - 5.0 mU/L. Roughly, half of the two groups of physicians would treat isolated TPOAb positivity. There is growing evidence on the association between thyroid autoimmunity and adverse obstetric outcomes, such as miscarriage (29), recurrent pregnancy loss (30), preterm delivery and low birth weight (31). Almost half of endocrinologists and 11% of generalists treat TPOAb negative with TSH 2.5 - 5 mU/L, a practice not in line with guidelines, which do not recommend L-T4 for these pregnant women (3, 4), underlining the need for further studies.
There is a lack of consensus on the definition and management of isolated maternal hypothyroxinemia (32). A large observational study has failed to show an association between maternal hypothyroxinemia and adverse obstetric outcomes (33). However, the prospective, nonrandomized Generation R study reported increased risk of lower communication development in children, born to women with isolated hypothyroxinemia (34). Although the majority of the responders practice in accordance with guidelines recommending, against the treatment of maternal hypothyroxinemia in pregnancy, it is remarkable that nearly 43% of endocrinologists reported that they would treat isolated maternal hypothyroxinemia.
In the published surveys on the management of thyroid dysfunction in pregnancy (5, 6), the investigations were performed in endocrinologists, either by internet or during AFES 2013, in Jakarta and in 21 countries from different Asian regions. The current survey presents a comparison of protocols between endocrinologists and generalists and covers physicians from eight eastern and southern Asian countries, revealing wide differences in the practice of specialists and generalists. The practices of Asian endocrinologists, in both surveys, were similar.
The present study, which provides a snapshot of current practices among endocrinologists and generalists in the management of thyroid disease during pregnancy in East-Asia, is unique in demonstrating potentially important differences between the views of sub specialists and generalists on this issue. Similar studies, in other countries, were conducted only among specialists, without any comparison with generalists, as the first line medical care providence. Previous studies have also reported differences in clinical approaches between generalists and specialists, with regard to treating several common conditions (35-38). The results emphasize that many clinicians, in particular generalists, do not adhere to up to date clinical practice guidelines, an important issue that should be considered in continuing education of professional societies.
This study has several limitations. First, the number of participants in this study is small. Second, the responders may not represent all physicians of East-Asian countries and hence, the results are not generalizable, because of the possible selection bias of those who chose to respond, although we could not calculate response rate. Third, variation in the prevailing clinical practices in different countries could have influenced the overall results of the survey. Also, within each group, especially generalists, different backgrounds, age and age of graduation or other confounders have not been considered. It is also not clear whether the results reflect the physicians’ actual practices or are influenced by selective recall, or their desire to give the “correct” answer, as previous studies have highlighted the substantial discrepancies between physicians’ self-reported practices and actual performance (35, 39). Finally, it is necessary to emphasize that this study did not compare patient outcomes, or any measure of cost-effectiveness between generalists and specialists, limiting only to their self-described practices, however this is beyond the scope of this study.
These results indicate that, for optimal management of patients with certain diseases, for which practices are evolving rapidly, especially in high risk groups, such as pregnant women, it would be better to include consultation or continuing involvement of specialists. However, in the current climate of aggressive cost reduction, surveying improvements in outcomes and cost-effectiveness are definitely required to support this conclusion. It is also necessary to develop and implement strategies that will increase physician knowledge on thyroid and pregnancy among generalists and subspecialists, so that their practice patterns would adhere more closely to the published guidelines.