1. Background
Prevalence of diabetes mellitus type 2 (DM) is increasing globally. The health and economic burden of this disease is very large and with an increasing trend. It is estimated that in the western countries, the prevalence of DM would increase 40 - 45% (1). Diabetes mellitus type 2 is an established risk factor for development of coronary artery disease (CAD) especially in women, and approximately 20% to 30% of the patients undergoing coronary artery bypass graft (CABG) have DM (2-4).
The prevalence of DM has a rapid growth in India, China, central and south America, Africa, and the Middle East (5, 6). Hyperglycemia is caused by impaired insulin secretion or insulin resistance, while both are known causes of DM type 2 (7). It was noted that an increase in intake of plant sources of fats and proteins in the diet would reduce the risk of CAD and DM type 2 (8). Several studies demonstrated that increased intake of mono- and poly-unsaturated fatty acids (MUFAs and PUFAs) and lower intake of saturated and trans-fatty acids are associated with a lower risk of DM type 2 (9-11).
Replacement of carbohydrates with MUFAs and PUFAs in the diet was shown as a known therapeutic strategy for DM type 2 (12). In this regard, daily intake of MUFAs in diabetic patients can restore high density lipoprotein (HDL)-cholesterol levels and improve blood sugar changes (13). Among the foods containing different fatty acid compositions, nuts have received more attention based on epidemiological findings between their regular intake and protective role against CAD (14, 15). Increase in consumption of nuts was reported to reduce the risk of CAD development and mortality caused by CAD (16, 17). The risks for progression of DM type 2 and health threatening conditions were reduced by consumption of nuts such as walnut (18). In this regard, because of the lower mortality caused by CAD and cancers in the Mediterranean population, the Mediterranean diet is still considered a favorable choice (19). A diet rich in walnut would have a positive effect on endothelial function and endothelium-dependent vasodilation in DM type 2. These dietary changes consequently would reduce the overall risk for CAD (20).
According to the reports of the US department of agriculture (USDA), 100 g of walnut contains 15.2 g of protein, 65 g of fat, and 6.7 g of fiber. Moreover, it contains higher amount of PUFA (47%) when compared with other nuts, out of which 38% is n6 PUFAs (linoleic acid) and 9% is n3 PUFAs (α linolenic acid) (21).
2. Objectives
Considering the potential role of PUFAs in prevention of DM type 2 and lipid profiles improvement, and absence of studies on control of blood sugar in DM type 2 patients using a diet containing walnut, the current study was carried out to evaluate the effect of walnut oil on blood sugar in DM type 2 patients.
3. Methods
3.1. Patient Selection and Randomization
This randomized control clinical trial was performed on DM type 2 patients referred to Shiraz healthy heart house. Patient recruitment was started in May 2013 and ended in June 2013. All of the 100 males and females with DM type 2 were enrolled using the block randomization sampling method. They were regularly monitored in the Shiraz healthy heart house for at least two years. Patients were eligible to be included in the study if they had DM type 2 (FBS > 126), were diagnosed at least for two years according to the definition of the American diabetes association (ADA), took less than three oral hypoglycemic agents, and were in the age range of 30 to 60 years.
Patients were excluded if one of the following criteria was met: alcoholism, smoking, any internal organ dysfunction, being on any therapeutic diet or calorie restricted regimen, and oral consumption of glucose lowering herbs during the past three months before beginning of the trial. All the patients received oral hypoglycemic agents and did not have a history of receiving insulin. Also, during the past three months before the study, their medications and diet regimen must have been kept unchanged.
The patients were randomly assigned to the experimental and control groups (each group with 50 patients) using the block randomization method. The research methodology was explained for the participants before the study and all the participants signed an informed consent as well. Moreover, they were assured of the confidentiality of their data and their right to withdraw from the study if they were not willing to continue. All participants were examined by a physician and a nutritionist at the beginning of the study, and afterward on a monthly basis.
Nutritional recommendations, checking for diet and medication regimen and physical activity were recorded for all patients on a weekly follow-up telephone call program. This study followed the guidelines of the declaration of Helsinki. This study was approved by the ethics committee of Shiraz University of Medical Sciences in advance by CT.P.92.5670 number and was registered in the Iranian Registry of Clinical Trials (IRCT2014022216682N1).
3.2. Walnut Oil Preparation and Administration
Before initiation of the experiment, walnut oil used in this trial was prepared according to the first cold press method and was analyzed on a gas chromatography and a flame ionization detector (FID). Fatty acid compositions were identified by comparing their relative and absolute retention times to those of authentic standards. The experimental group received walnut (Juglans regia L.) oil (15 g/day for three months). The patients in the control group did not undergo any intervention. All patients received dietetic consultation about a balanced diet and were advised according to their calorie needed for maintaining their weight unchanged. They were also asked to leave their physical activity habits unchanged during the study and take their hypoglycemic medications appropriately.
3.3. Sample Collection and Clinical Assessments
Before initiation of the experiment, the systolic blood pressure (SBP) and diastolic blood pressure (DBP) levels were measured by a standard sphygmomanometer (ALPK2, Japan) and were recorded. Blood pressure was measured when the patient had at least 10 minutes rest and did not drink coffee or tea, or did not smoke during the past three hours. Anthropometric measurements, such as weight, and height were done using calibrated equipment. Between 07:00 and 08:00 AM, after at least 12 hours of overnight fasting, a 5-mL blood sample for each patient was collected in clot tubes.
Measurement of fasting blood sugar (FBS) and hemoglobin A1c (HbA1c) values were done using enzymatic assay kits (Parsazmoon, Iran). At the end of a three-month period, another blood sample was collected to measure the FBS and HbA1c levels again by considering the aforementioned procedure. Moreover, weight, SBP and DBP were measured and recorded again.
3.4. Statistical Analysis
The sample size for this study was determined to be 100 males and females with DM type 2, considering α = 5%, power of 80% and also effect size of 40% using n = 2δ2(Z1 - α/2 + Z1 - β)2/(µ1-µ2) formula. The obtained data were analyzed using the SPSS software (version 16, Chicago, IL, USA). Normal distribution of the data was tested by the Kolmogorov-Smirnov test. Comparison of pre- and post-test means of the variables was carried out using paired t-test and the mean comparison between the groups was performed by the t-test. We compared any after measurement adjusted for its before measurement using ANCOVA statistical test. P values of < 0.05 were considered statistically significant.
4. Results
4.1. Patients’ Characteristics
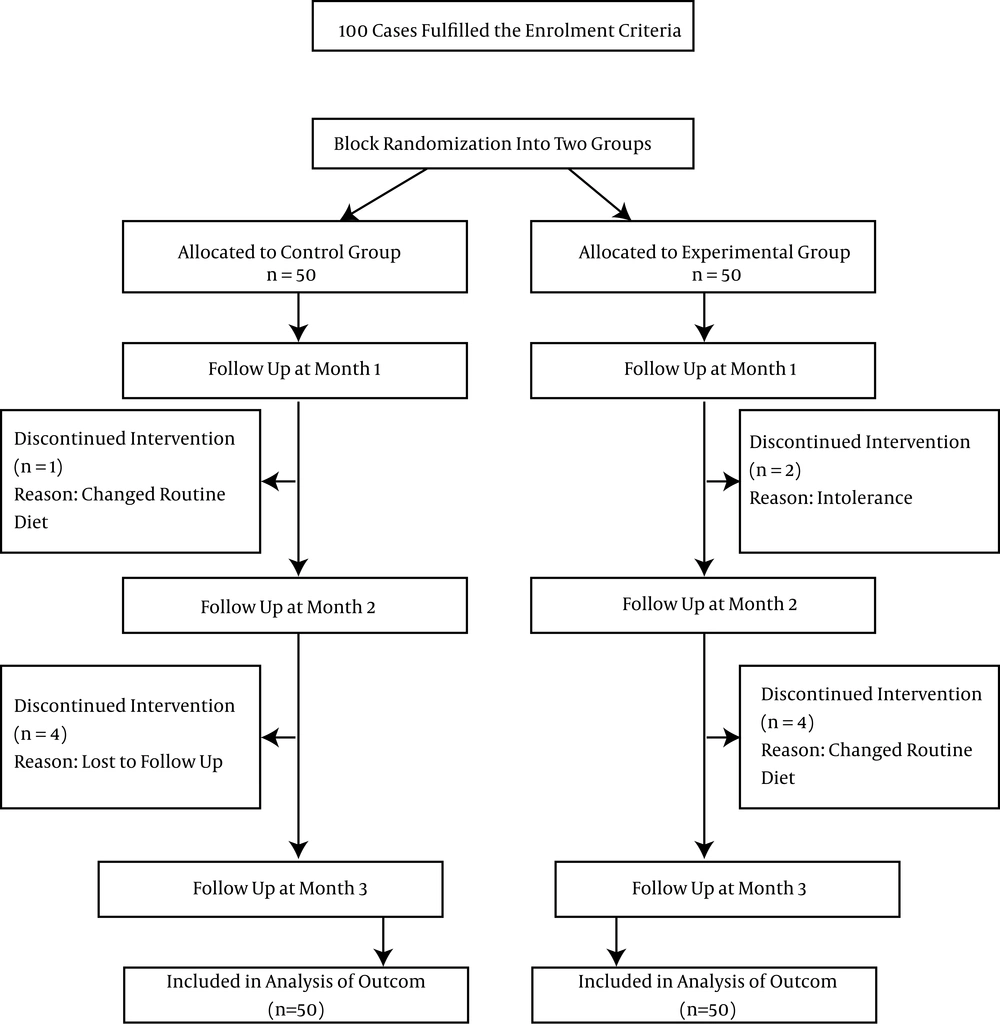
This study was conducted on 100 diabetic patients; 50 in the experiment group and 50 in the control group. Consort flow chart of the study population is presented in Figure 1. Among the participants in the experiment group, two subjects were withdrawn from the study because of gastrointestinal intolerance of the oil and three subjects did not follow their routine diet and were advised for oil consumption and were excluded from the study while the final number of cases in intervention group was 45 (24 females and 21 males). In the control group, one subject was excluded because of changing the advised diet, and four were excluded from the analysis because of loss in the follow-up (23 females and 22 males). The baseline characteristics of the participants in the two groups are presented in Table 1.
| Variable | Experimental, n = 45 | Control, n = 45 | P Value |
|---|---|---|---|
| Age, y | 55.5 ± 10.75 | 54 ± 11.37 | 0.507 |
| Body weight, Kg | 75.9 ± 8.56 | 73.81 ± 7.15 | 0.212 |
| BMI, Kg/m2 | 27.60 ± 2.47 | 27.21 ± 2.27 | 0.435 |
| SBP, mmHg | 137.75 ± 14.5 | 132.57 ± 17.01 | 0.124 |
| DBP, mmHg | 81.62 ± 7.65 | 81.13 ± 7.01 | 0.753 |
| FBS, mg/dl | 158.37 ± 48.1 | 153.88 ± 54.76 | 0.681 |
| HbA1c, % | 7.003 ± 1.08 | 6.97 ± 1.208 | 0.894 |
Abbreviation: BMI, Body Mass Index; DBP, Diastolic Blood Pressure; FBS, fasting blood sugar; HbA1c, hemoglobin A1c; SBP, systolic blood pressure.
aData are presented as mean ± Standard Deviation, independent t- test was used.
4.2. Fasting Blood Sugar and HbA1c Levels
The interventions were well tolerated and adherence to the advised diet regimen and walnut oil consumption (15 g/day for three months) was good. According to the obtained results, the two groups were not significantly different in distribution of age, height, weight, Body Mass Index (BMI), SBP, DBP, FBS level, and HbA1c (Table 1).
Three months after the beginning of the experiment, a statistically significant decrease was observed in the FBS level of the experimental group by 8.24% ± 16.77 (P = 0.001) from 158.37 ± 48.16 before the intervention to 137.91 ± 23.24 after the intervention. Such finding was not observed for the control group and the FBS level remained relatively unchanged. Comparing the two groups after the experiment indicated that the FBS level significantly decreased in the experiment group (P < 0.05).
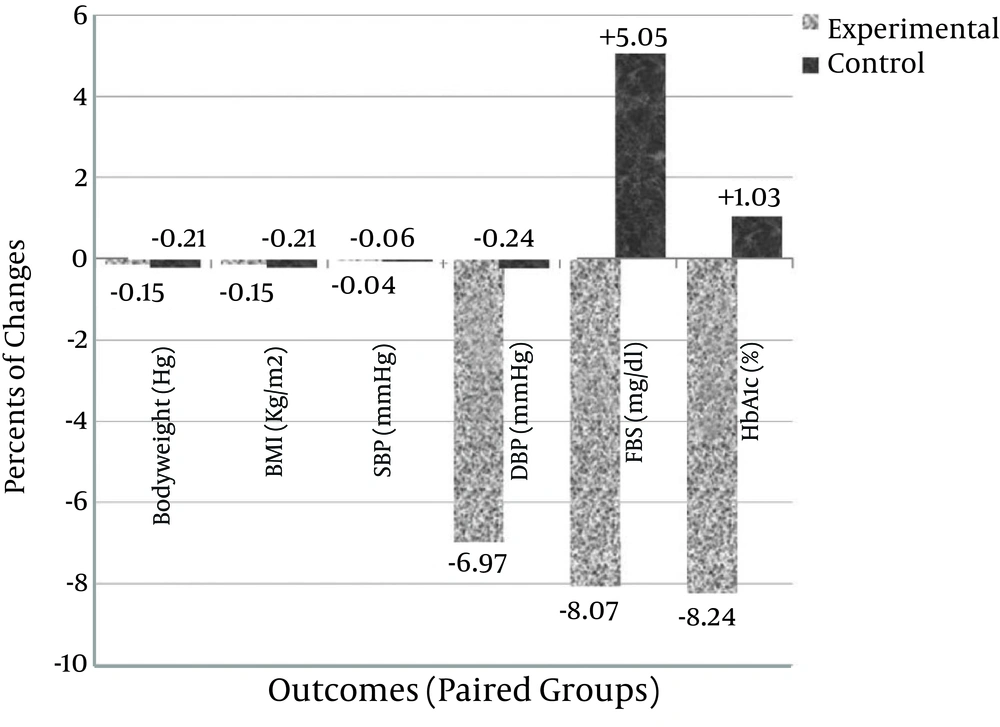
Also, the HbA1c level was significantly decreased in the experimental group by 7.86% ± 21.97 (P = 0.005); from 7.00 ± 1.08 before intervention to 6.37 ± 1.29 after intervention, but this value did not change significantly in the control group. Mean percentage of change for each group is demonstrated in Figure 2. Comparing of the two groups after the intervention indicated that the HbA1c level significantly decreased in the experimental group (P < 0.05). The values before and after the experimental variables and within group changes (Mean ± SD), as well as, after treatment values of the variables compared between the groups using analysis of covariance with adjustment of baseline values are shown in Table 2.
Percentage of within group changes (after intervention values – baseline values), for each value in this study, is shown. The Y column shows the negative or positive change percentage (%) at the end of the intervention and the X row presents different outcomes paired as control and experimental groups.
| Variables | Phase of Intervention | Experimental Group | Control Group | P Value, ANCOVA for Groups |
|---|---|---|---|---|
| Bodyweight, Kg | 0.787 | |||
| Before | 75.90 ± 8.56 | 73.81 ± 7.15 | ||
| After | 75.75 ± 8.25 | 73.65 ± 7.22 | ||
| Change, % | -0.15 ± 1.12 | -0.21 ± 2.22 | ||
| P-value | 0.225b | 0.504 | ||
| BMI, Kg/m2 | 0.787 | |||
| Before | 27.60 ± 2.47 | 27.21 ± 2.27 | ||
| After | 27.55 ± 2.34 | 27.15 ± 2.30 | ||
| Change, % | -0.15 ± 1.12 | -0.21 ± 2.22 | ||
| P-value | 0.233 | 0.500 | ||
| SBP, mmHg | 0.556 | |||
| Before | 137.75 ± 14.5 | 132.57 ± 17.01 | ||
| After | 137.51 ± 12.27 | 132.42 ± 16.70 | ||
| Change, % | -0.04 ± 3.22 | -0.06 ± 2.06 | ||
| P-value | 0.688 | 0.707 | ||
| DBP, mmHg | 0.788 | |||
| Before | 81.62 ± 7.65 | 81.73 ± 7.01 | ||
| After | 81.42 ± 7.21 | 80.84 ± 6.83 | ||
| Change, % | -0.08 ± 3.97 | -0.24 ± 4.53 | ||
| P-value | 0.692 | 0.605 | ||
| FBS, mg/dL | 0.007 | |||
| Before | 158.37 ± 48.16 | 153.88 ± 54.76 | ||
| After | 137.91 ± 23.24 | 153.93 ± 42.06 | ||
| Change, % | -7.86 ± 21.97 | 5.05 ± 29.14 | ||
| P-value | 0.005 | 0.995 | ||
| HbA1c, % | 0.005 | |||
| Before | 7.00 ± 1.08 | 6.97 ± 1.21 | ||
| After | 6.37 ± 1.29 | 6.98 ± 1.33 | ||
| Change, % | -8.24 ± 16.77 | 1.03 ± 15.76 | ||
| P-value | 0.001 | 0.941 |
Abbreviation: BMI, Body Mass Index; DBP, Diastolic Blood Pressure; FBS, fasting blood sugar; HbA1c, hemoglobin A1c; SBP, systolic blood pressure.
aData are presented as mean ±Standard Deviation, paired sample t test and ANCOVA were used.
bP-value compared before and after intervention.
Considering the data shown in Table 2 and comparison of the values obtained for each parameter before and after the experiment in each group, it can be suggested that none of the secondary outcomes, including weight, BMI, SBP, and DBP variables changed significantly in both groups. No remarkable adverse events were reported, except for two reports of gastro-intestinal irritation sense.
5. Discussion
A relationship between increased risk of atherosclerosis and CAD was previously shown (22). The study performed on the role of diet in control of blood sugar in DM type 2 patients emphasized on replacement of conventional fats with oils containing PUFAs (23). In our study, the effect of consumption of walnut oil, which contains high levels of PUFAs especially Alpha linolenic acid (ALA) was investigated on blood sugar control in DM type 2 patients. It was shown that consumption of walnut oil for three months (15 g/daily) could significantly reduce FBS and HbA1c levels. However, no obvious changes were observed in the weight, BMI, and blood pressure levels. This is in agreement with the results of some previous studies (20, 21, 24).
It has been demonstrated that oils containing PUFAs could exert their antidiabetic effect by reducing resistance and enhancing sensitivity to insulin via the mechanism of overexpression of glucose transporter GLUT4 and insulin receptors on the adipocyte membrane and also reducing the inflammatory effect on adipose tissue by reducing the inflammatory markers in this tissue (25). In a study on mice receiving a cis 9, trans 11- conjugated linoleic acid (CLA)-enriched diet for six weeks, it was demonstrated that this CLA isomer could reduce insulin resistance, and decrease FBS and serum insulin levels by increasing adipose tissue plasma membrane GLUT4. Moreover, this type of CLA could reduce inflammation in the adipose tissue by a 50% decrease in the tumor necrosis factor- α (TNF-α) level. Therefore, it was suggested that this CLA isomer attenuates insulin resistance by having anti-inflammatory effects on adipose tissue (25).
It was reported that diabetes could potentially be associated with increased oxidative stress (26). Furthermore, oxidative stress could be associated with activation of stress-sensitive signaling pathways or insulin resistance (27). Thus, based on the idea that oxidative stress can act as an activator in initiation and progression of DM type 2, antioxidants have been suggested as a part of DM type 2 treatment (28). Moreover, it has been shown that the high level of antioxidants in nuts could be a protective mechanism against oxidative injury (29).
It has been noted that walnut has a higher antioxidant capacity when compared with other nuts (30). These antioxidants are possibly of phenolic compounds (31), including hydrolyzed tannins, tocopherol (22), and melatonin; all of which have a high antioxidant capacity (32). In the study of Ansar et al., the effect of daily consumption of alpha lipoic acid, as an antioxidant, for two months was compared with the placebo in DM type 2 patients. It was observed that in the group, which received alpha lipoic acid, the FBS and insulin resistance homeostasis model assessment (IR-HOMA) decreased significantly but body weight remained unchanged. In this regard, it was concluded that alpha lipoic acid could be used as an antioxidant in treatment and improvement of glucose homeostasis in patients with diabetes (23).
In our study, FBS level significantly decreased in the group, which received walnut oil (P < 0.05), while no significant change was observed in weight, SBP and DBP. Lack of change in blood pressure was also reported by Jenkins et al. (13). In the study of Rahimi et al. performed on diabetic rats, it was reported that diabetic rats that received walnut oil had a statistically significant decrease in HbA1c level, similar to receiving glibenclamide. Thus, it was suggested that walnut oil could have antidiabetic effects (33).
In a study performed by Tapsell et al., the effects of PUFAs in walnut oil were evaluated on metabolic parameters of DM patients. They reported that dietary fat changed in DM patients (increase in the PUFA/saturated fatty acid ratio) and could effectively decrease FBS, HbA1c, and serum insulin levels after six months of the intervention (24). One of the strong points of our study is using nutritional strategy to stabilize physical activity and dietary intake in patients. These strategies were consulting, planning and monitoring the patient’s diet during the study.
The novelty of our study was to use first cold press oil without any process or extracting. Also our study met some limitations such as lack of testing insulin secretion or insulin receptor respond or sensitivity changes, lack of dose-response analysis, low external validity of the study as it is a single center study and energy metabolism rate or indicators. Also we recommend further studies considering nutritional intake and parameters information as a confounder and retrieving them for analysis, unlike our study in which we only monitored for any possible changes and did not compare statistically between the groups.
In general, the results obtained from our study indicate that changes in dietary oil composition consumed by DM type 2 patients and shifting from fats containing saturated fatty acids to the containing PUFAs and ALA, such as walnut oil, would lead to statistically significant decrease in the blood sugar levels. This eventually improved blood glucose homeostasis and prevented cardiovascular complications of diabetes without changing the weight or blood pressure.
In conclusion, consumption of walnut oil (15 g/day for three months) led to statistically significant decrease in FBS and HbA1c levels and generally improved blood glucose homeostasis. Considering that this nut is a widespread part of the recipe and also use the whole plant instead of the processed product, it is easy and available for everyone. Therefore supplementation with walnut can be effective in attenuation or prevention of the symptoms of DM type 2 as well as its main concomitant disorders, such as metabolic syndrome.