1. Background
Pregnancy has a profound but reversible effect on the thyroid gland and its functions; it is actually a state of excessive thyroid stimulation leading to an increase in thyroid size by 10% in iodide sufficient areas and 20% - 40% in iodide deficient regions (1). Furthermore the physiological and hormonal changes caused by pregnancy lead to an increase of up to 50% in thyroxin (T4) and triiodothyronine (T3) and daily iodide requirement along with a decrease in thyrotropin (TSH) levels, especially in the first trimester (2). In an iodide sufficient area these thyroid adaptations during pregnancy are well tolerated, since stored inner thyroid iodide is sufficient; however in iodide deficient areas these physiological adaptations lead to significant changes during pregnancy (3).
Thyroid dysfunction is the second most common endocrine disease in females of reproductive age (4, 5); 2% - 3% of pregnant females are diagnosed with hypothyroidism (0.3% - 0.5% with overt hypothyroidism, 2% - 2.5% with subclinical hypothyroidism), and hyperthyroidism is also reported in 0.1% - 0.4% of pregnant females. Moreover, about 10% - 20% of all euthyroid pregnant females are positive for thyroid antibodies (6).
Although the complications of hypothyroidism or overt hyperthyroidism in pregnancy outcomes and neonatal and childhood development are well known (7-11), there is still no consensus on the association between subclinical thyroid disorders or increasing thyroid antibodies and complications in pregnancy and childhood. Some studies show that subclinical hypothyroidism and thyroid autoimmunity are associated with adverse maternal and fetal outcomes (11-15) and even reported that euthyroid females with abnormally increased serum thyroid antibodies levels are at increased risk of adverse outcomes of pregnancy such as spontaneous abortion and premature labor (11, 14, 16), whereas others document contrary findings (17, 18).
Given the lack of consensus on universal screening and the lack of evidence on the effect of treating subclinical hypothyroidism and euthyroid TPOAb positive pregnant females for improving pregnancy outcomes, there is still not lock of evidence or guidelines to treat these females; such recommendations need to be developed according to the results of well-designed randomized trials in areas under different statuses of iodine adequacy (1).
To address this need, the present study was designed to determine the prevalence of thyroid dysfunction during pregnancy and determine the effectiveness of levothyroxine treatment on pregnancy outcomes in females with subclinical thyroid dysfunctions and autoimmunity.
1.1. Type of Study
The current study consisted of two phases. The first phase of the study was a population based cross sectional study in which pregnant females, attending prenatal clinics (Shahid Beheshti University of Medical Sciences) were screened for thyroid dysfunction, by collecting medical history, clinical examination and measurement of serum concentrations of TPOAb, TSH, T4 (TT4) and RT3U; participants were grouped as: normal thyroid status, overt thyroid dysfunction (hypothyroidism and hyperthyroidism), TPOAb positive (euthyroid/subclinical hypothyroidism), and TPOAb negative subclinical hypothyroidism; all groups, except overt thyroid dysfunction, were monitored till delivery and any adverse outcomes of pregnancy were recorded. Those with overt hypo or hyperthyroidism or preexisting thyroid dysfunction were excluded from the present study and referred to endocrinologists.
Pregnant females with thyroid dysfunction were selected for the second phase of study, a randomized clinical trial, and randomly divided into two groups; the intervention group was treated with levothyroxine, while the other group received no treatment. In the second stage, the effect of levothyroxine on pregnancy outcome in females with subclinical thyroid dysfunction and autoimmunity was determined. Stages of study are shown in Table 1.
This study protocol was approved by the medical ethics committee of the research institute for endocrine sciences (approval number: 32ECRIES92/07/23).
| Stage | Proceedings |
|---|---|
| Recruitment and completion of the questionnaire | Obtaining written informed consent form and completing the questionnaire |
| Collection of the laboratory samples | Obtaining fasting blood sample sand collecting three urine samples weekly |
| Classification | Determining groups according to their hormonal assessments |
| Start of intervention | Prescribing levothyroxine for the intervention group |
| Replication of laboratory samples | Obtaining fasting blood samples at 20 and 30 weeks of pregnancy |
| Follow-up | pregnancy outcomes and neonatal thyroid screening |
2. Objectives
1) To determine the prevalence of subclinical hypothyroidism and TPOAb+ in pregnant females.
2) To identify whether TPOAb+ euthyroid/subclinical hypothyroid females are at an increased risk of pregnancy complications.
3) To identify whether TPOAb-/subclinical hypothyroid females are at an increased risk of pregnancy complications.
4) To determine the effect of levothyroxine on pregnancy complications in females with subclinical thyroid dysfunction and autoimmunity.
5) To determine the effect of levothyroxine on neonatal TSH levels at birth (three to five days of birth) in females with subclinical thyroid dysfunction and autoimmunity.
3. Patients and Methods
3.1. The First Phase
3.1.1. Subjects
The enrolment criteria in the first stage of the study was being in the first trimester of pregnancy (4 - 13 weeks of gestation) and having no preexisting thyroid dysfunction.
3.1.2. Sampling Framework
Using the cluster sampling method, 2,000 first trimester pregnant females were selected from the ones receiving prenatal care in centers under coverage of Shahid Beheshti University of Medical Sciences, responsible for monitoring the health of over ten million people in Tehran province (over two thirds of Tehran’s population), including northern, western and eastern Tehran and surrounding counties (19); it is documented that the socio-demographic characteristics of this population does not differ from the total population of Tehran, based on reports of the statistical center of Iran (19).
A stratified, multistage probability cluster sampling method, with a probability in proportion to size procedure, was used. Otherwise, the subjects of the present study were recruited using cluster sampling method from 24 randomly selected prenatal clinics. To select these clinics authors initially divided them to four geographic subgroups, following which six clinics from each subgroup were randomly selected. Subsequently the proportion of samples required in each clinic was calculated based on the total number of pregnant females visited during the previous six months.
3.1.3. Study Procedure
A training course was conducted for those midwives providing prenatal care in the selected centers to explain the objectives and protocol of the study; these trained personnel were in charge of conducting interviews and physical examinations. First trimester pregnant females referred to prenatal care centers were invited to participate in this study after being given a detailed explanation of the study procedure. Signed consent forms were obtained from all eligible females enrolling in the study. Pregnancy age was determined based on the date of the first day of last normal menstrual period; if this date could not be specified, it was determined by ultrasonography at the first prenatal visit. A fasting blood test was obtained and three urine bottles were given to each of the study participants to collect urine samples on three different days of the current week. The serum of the samples were separated by centrifugation and transferred to the laboratory of the research institute of endocrine sciences of Shahid Beheshti University of Medical Sciences, according to the standard cold chain; serum concentration of TPOAb, TT4, T-uptake and TSH were then measured immediately and the thyroid status of each females was determined (Table 2). Urinary iodine was measured in three urine samples to obtain the median. Free thyroxin index (FTI) was calculated using Equation 1:

| Tests | Methods | Kits | Equipment |
|---|---|---|---|
| T4 | Radioimmunoassay (RIA) | Izotop kit, Budapest co., Hungary | Gama-counter (Dream Gamma-10, Goyang-si, Gyeonggi-do, South Korea) |
| T-uptake | Enzyme immunoassay (EIA) | Diaplus kit, San Francisco, CA., USA | ELISA reader (Sunrise, Tecan Co. Salzburg, Austria) |
| TPOAb | Immunoenzymometric assay (IEMA) | Monobind kit, Costa Mesa, CA., USA | ELISA reader (Sunrise, Tecan Co. Salzburg, Austia) |
| TSH | Immunoradiometric assay (IRMA) | Izotop kit, Budapest co., Hungary | Gama-counter (Dream Gamma- 10, Goyang-si, Gyeonggi-do, South Korea) |
All pregnant females, regardless of thyroid status received standard prenatal care. All prenatal visits were recorded and each adverse pregnancy outcome was managed according to the standard guidelines. The study checklist was completed at each visit (Table 3).
| Section | Details |
|---|---|
| Demographic information | Age, age of marriage, occupation status, level of education, level of spouse’s education, occupation status of spouse’s |
| Past Medical history | Hypertension, diabetes mellitus, heart disease, respiratory disease, kidney disease |
| Medication | Antihypertensive, anti–diabetes, drugs for heart disease, anticonvulsants |
| Thyroid history | History of thyroid disease, taking thyroid medication, treated with radioactive iodine, history of thyroid surgery |
| Family history of prenatal complications | Miscarriage, premature labor, fetal abnormality, hypertension in pregnancy, gestational diabetes |
| Obstetrical history | History of infertility, gravida, para, abortion, living child, dead child, intra uterine fetal death, mole, type of delivery, fetal sex, twin pregnancy, adverse pregnancy outcomes ( preterm labor, hypertension, gestational diabetes, third trimester hemorrhage, fetal abnormality) |
| Current pregnancy information | Last menstrual period, twin or multiple pregnancies, duration of menstrual cycle, hemorrhage during pregnancy, nausea and vomiting, treatment of nausea and vomiting |
| Physical examination | Weight, height, systolic and diastolic blood pressures, pulse rate, Achilles reflex, thyroid exam, heart sounds, respiratory exam |
| Signs and symptoms of thyroid dysfunction in current pregnancy | Tachycardia, hypertension, irritability and restlessness, sleep difficulty, anxiety, sweating, heat sensitivity, tremor and involuntary tensions, weight loss, changes in skin thickness, more frequent bowel movements, muscle weakness, hair loss, goiter, nail separation, clubbing, Weakness, fatigue, facial edema, anemia, lazy, sleepy, depression, sweating decrease, cold intolerance, joint and muscle pain, weight gain, dry, rough and cold skin, constipation, hoarseness, paresthesia, deafness, slow movement, pre-orbital puffiness |
| Prenatal visit | Weight, systolic and diastolic blood pressure, pulse rate, fundal height, fetal heart rate, high risk symptoms (edema, , headache, vaginal bleeding, uterine contractions, other abnormal signs), drugs (6 - 10 weeks or 11 - 15 weeks, 16 - 20 weeks or 21 - 25 weeks, 26 - 30 weeks, 31 - 34 weeks, 35 - 37 weeks, 38 weeks, 39 weeks, 40 or 41 weeks) |
| Information of discontinued pregnancy | Type of termination of pregnancy (spontaneous abortion, induced abortion, preterm labor), gestational age, alive at birth, gender of the fetus, abnormality |
| Labor and delivery | Time of rupture of membrane, admission blood pressure, type of delivery, the duration of labor, postpartum hemorrhage, inertia (atonic) postpartum |
| Newborn | Gender, 5 - 10 minute Apgar score, birth weight, height, head circumference, anomaly, admission in NICU, neonatal seizures, neonatal icterus |
3.1.4. Sample Size
According to the previous studies the prevalence of thyroid dysfunction was about 15%, considering β = 0.02 and type 1 error of 0.05 the required sample was 1225, by adding 30% loss to follow-up (not providing hormonal profile), a total sample of 1,600 was adequate.
3.1.5. Grouping of Samples
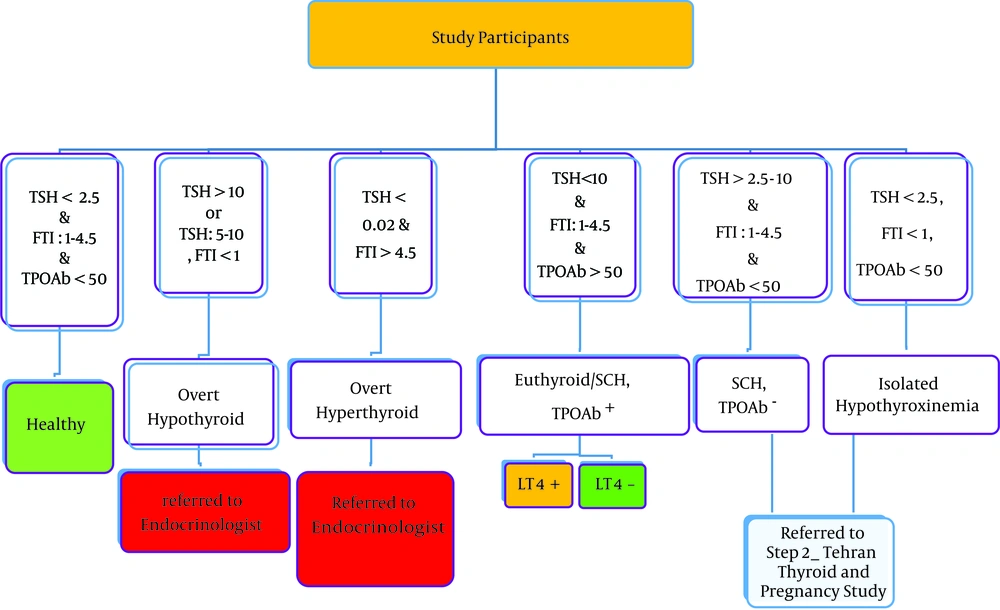
Based on the results of laboratory examinations, patients with TSH level < 2.5 μIU/mL, FTI: 1-4.5 and TPOAb < 50 IU/mL (negative) were considered with normal euthyroid; patients with TSH levels > 10 μIU/mL or 5 - 10 μIU/mL and FTI < 1, were diagnosed as overt hypothyroidism and subjects with TSH levels < 0.02 μIU/mL and FTI > 4.5, were diagnosed as overt hyperthyroidism; subjects with TSH levels between 2.5 - 10 μIU/mL, and FTI between 1 - 4.5, were considered as sub-clinically hypothyroid, regardless of TPOAb status, whether positive or negative. Females with TSH levels < 2.5 μIU/mL, FTI < 1 and TPOAb < 50 IU/mL (negative), were considered as isolated hypothyroxinemia and subjects with TSH levels < 2.5 μIU/mL, FTI between 1 - 4.5 and TPOAb > 50 IU/mL (positive), were considered as positive TPOAb euthyroid.
3.2. The Second Phase (Randomized Clinical Trial)
3.2.1. Clinical Trial Design
The second phase of the study was a randomized clinical trial (the trial was a phase II trial). TPOAb+ euthyroid/sub clinically hypothyroid females were selected for the second phase of the study, and were randomly assigned into two groups: 1) intervention; treatment with standard dose levothyroxine (LT4) and 2) controls; not receiving any thyroid medication.
3.2.2. Participants
Pregnant females positive for thyroid peroxidase antibodies (TPO Ab) without overt thyroid dysfunction (euthyroid TPOAb+ and subclinical hypothyroid TPOAb+) entered the second phase of the study. Pregnant females with subclinical hypothyroid TPOAb- or isolated hypothyroxinemia were referred to phase II of Tehran thyroid and pregnancy study (Figure 1). The enrolment criteria were singleton pregnancy without medical contraindications to take levothyroxine. Exclusion criteria include twin and multiple pregnancies, and possessing overt thyroid disorders (hypothyroidism and overt hyperthyroidism).
3.2.3. Sample Size
Sample size was calculated to assume superiority (in terms of primary outcomes) for two independent proportions in cluster randomization. It was calculated that with a sample of 85 pregnant females in each group, the study may have at least 80% power to show an effect size of 0.3 in the primary outcomes with a type I error of 0.05. Considering 10% loss to follow-up, 92 females in each study group were considered adequate.
3.2.4. Randomization
Study subjects were randomly divided into two groups using permuted block randomization to achieve balance across treatment groups. The number of subjects per block was four. A sealed opaque envelope was assigned to each subject, only the midwife treating the females, who did not participate in any subsequent phases of the study knew which group each patient belonged to.
3.2.5. Study Procedure
Using block randomization, the participants entering the second phase of the study were randomly divided into two groups: 1) intervention; treated with levothyroxine as follows: Dose 0.5 μg/kg/d if they had TSH < 1.0 μIU/mL, 0.75 μg/kg/d for TSH 1.0 - 2.0 2.0 μIU/mL, and 1 μg/kg/d for TSH > 2.0 μIU/mL or a TPOAb titer > 1500 IU/mL; the dosages were maintained throughout the gestation; 2) controls, not receiving any thyroid medication.
A fasting blood sample was obtained at weeks 20 and 30 of gestation from both groups; the serum of samples were separated by centrifugation and transferred to the laboratory of the research institute of endocrine sciences of Shahid Beheshti University of Medical Sciences according to the standard cold chain. The samples were stored at -80°C, and serum concentrations of TT4, T-uptake and TSH were measured at the end of study. Recommended LT4 dosages were maintained throughout the gestation. TSH of their offspring was assessed at three to five days after delivery. All study participants received standard prenatal care and were monitored until delivery. All prenatal visits were recorded and each adverse pregnancy outcome was managed according to the standard guidelines. The study checklist was completed at each visit (Table 3).
3.2.6. Outcomes
In the current study the primary outcome was preterm delivery and secondary outcomes included miscarriage, low birth weight, placenta abruption, and serum concentration of TSH of newborn (third to fifth day of birth).
3.2.7. Statistical Analysis
Continuous variables were checked for normality using the one-sample Kolmogorov-Smirnoff test; categorical variables were expressed as percentages, and compared using Pearson’s χ2-test. Distribution of variables between the two groups was compared using t-test and expressed as mean ± standard deviation. An intention-to-treat (ITT) analysis of the results was used. Logistic regression, liner regression and ANOVA (repeated measure) were used to compare the two groups and to determine the effect of levothyroxine treatment on the outcomes. Statistical analysis was performed by SPSS software version 18.
3.2.8. Limitations of the Study
Limitations of the study included: 1) starting the prenatal visit at pregnancy age > 14 weeks;
2) loss to follow-up; 3) no consumption or lack of adherence to levothyroxine by pregnant females in intervention group.
3.2.9. Timing
The study duration was scheduled three years.
4. Discussion
Despite the high prevalence of thyroid disorders in pregnancy, there is no agreement on optimum approach to pregnant females with subclinical thyroid dysfunction and euthyroid TPOAb+. Various scientific societies, including the America thyroid association (ATA), European thyroid association (ETA), and the thyroid society report lack of sufficient evidence and clinical trials on effectiveness of levothyroxine treatments of such pregnant females in terms of pregnancy or neonatal outcomes; they are mostly referred to a single clinical trial conducted by Negro et al. (11) who found that TPOAb+ pregnant females (euthyroid and subclinical hypothyroidism) are associated with an increased risk of miscarriage (P < 0.05; relative risk (RR) = 1.72; 95% confidence interval (CI): 1.13 - 2.25) and preterm delivery ( P < 0.05; RR = 1.66; 95% CI :1.18 - 2.34). Treatment with levothyroxine had some beneficial effects on these adverse pregnancy outcomes (11, 20). Also, Lata et al. (21) reported that following LT4 treatment, there was no difference in prevalence of abortion between hypothyroid and euthyroid TPOAb+ females, although LT4 treatment was not beneficial in euthyroid TPOAb+ infertile females undergoing assisted reproduction technologies (22). Generally, the data however was insufficient to recommend for or against routine levothyroxine therapy in thyroid antibody positive euthyroid females during pregnancy.
Several studies reported that the prevalence of various thyroid risk factors in different countries, depend on the life style and their Iodine status; as a result, clinical trials need to be conducted in various parts of the world among different races with different prevalence of thyroid disorders. The current study aimed to determine the prevalence of thyroid disorders in a population-based study conducted among pregnant Iranian females and identify the effectiveness of treatment with levothyroxine on pregnancy outcomes in females with subclinical thyroid dysfunctions and/or thyroid autoimmunity. The results of the study can be useful in decision-making and prioritizing the optimum management of thyroid disorders of pregnancy.