1. Background
Acromegaly is an uncommon condition characterized by the hypersecretion of growth hormone (GH) and consequently, the elevated levels of insulin-like growth factor 1 (IGF-I). Over 90% of patients with acromegaly have a benign GH-secreting pituitary adenoma (1, 2).
Surgical resection is the first-line option for treating the adenomas, with the remission rate of 85% for microadenomas and 40% - 50% for macroadenomas at experienced centers (3). In case of surgical treatment failure, the accepted second-line therapy includes medical therapy with somatostatin analogs (SSA), GH receptor antagonists, and/or dopamine agonists (DAs) such as Cabergoline (4, 5). However, the recent literature indicates that biochemical control is achieved in < 40% of patients treated with SSA therapy (6, 7). Hence, due to inconsistent responses to surgical resection and postoperative medical therapy, investigators are seeking for probable parameters that could be helpful in predicting the clinical outcome.
Studies have investigated some clinical parameters such as age, gender, tumor size, local invasiveness, and presurgical GH levels, obtaining controversial results (3, 8, 9). The nuclear antigen Ki-67, measured by an antibody called MIB-1, recognizes the proliferative activity of the cells (10).
In recent years, some researchers have studied the Ki-67 index level of pituitary tumors and its relationship with demographics, biochemical parameters, clinical behavior, and recurrence, without any discrepancy in the function of the tumor (11-13). However, a dearth of research exists for such a correlation in GH-producing adenomas (14-17).
2. Objectives
The aim of this study was to evaluate the correlation of Ki-67 index level with clinicoradiological and endocrinological parameters in confirmed GH-producing adenomas, as well as with surgical response and medical treatment after surgery.
3. Methods
We retrospectively studied the medical records of 49 patients with GH-secreting pituitary adenoma who underwent surgical resection by three experienced neurosurgeons between 2008 and 2017 in Shariati hospital affiliated to Tehran University of Medical Sciences. This period was chosen because of access to pathology reports. The records were assessed to obtain their clinical, biochemical, radiologic, and pathologic data. The study was performed in accordance with the Declaration of Helsinki.
Acromegaly had been diagnosed based on clinical manifestations. The pituitary adenoma was detected by magnetic resonance imaging (MRI). The biochemical elevation of GH and IGF-1 levels and the lack of GH suppression were determined following the oral glucose suppression test (OGTT), which was confirmed on pathological investigations by an experienced pathologist.
Preoperative GH and IGF-1 concentrations were measured by the enzyme-linked immunosorbent assay (ELISA) (Monobind Inc. USA) at the Hormone Laboratory of Shariati hospital and the results were expressed as ng/mL.
Preoperative MRI studies were performed at the Radiology Department of Shariati hospital on a Siemens 1.5T Avanto 18 channel MRI scanner unit (Erlangen, Germany) with the following sequences: T1/coronal 2.5 mm, T1/sagittal 2.5 mm, T2/ coronal 2.5 mm, and T2/sagittal 2.5 mm.
Postcontrast imaging was also performed. The imaging analyses were done by two experienced radiologists.
The patients' preoperative, three months postoperatively, and the latest available GH and IGF-1 serum levels were registered. The tumors were categorized in MRI studies according to their size as either microadenomas (< 10 mm) or macroadenomas (≥ 10 mm), and their expansion to extrasellar structures was determined. Partial tumor removal was defined as tumor remnant at surgery and/or at MRI imaging three months postoperatively (18). According to the most recent acromegaly guidelines (19, 20), the criteria for remission were defined as the normalization of IGF-I levels, adjusted to age and gender, and GH levels below 1 ng/mL.
Since the GH receptor antagonist (Pegvisomant) was not available in Iran, patients whose diseases were persistent after surgery underwent medical therapy with SSA and/or DAs. Surgically removed specimens were fixed in formalin and then embedded in paraffin. All tissue specimens were examined for anterior pituitary hormones (GH, PRL, ACTH, TSH, FSH, and LH). The Ki-67 antigen was measured using the MIB-1 antibody.
3.1. Statistical Analysis
Data were analyzed using SPSS 16.0 software. Normally distributed data are expressed as means ± standard deviation (SD) and skewed variables as median and interquartile range (IQR). Categorical data are presented as numbers and percentages. The Man-Whitney test was performed to determine statistically significant differences. Correlations were assessed using Spearman's correlation coefficient (SCC). Multivariable logistic regression analysis was performed to determine the probable predictors of early and late remission. In this analysis, Ki-67 index, age at the time of surgery, gender, preoperative IGF-1, preoperative GH, cavernous sinus invasion, and suprasellar invasion were entered into the model. A calculated probability of < 0.05 (P value < 0.05) was considered statistically significant.
4. Results
4.1. Patients’ Characteristics
The study sample consisted of 35 (71.4%) females and 14 (28.6%) males, with the mean age of 41.9 ± 12.9 years (range = 22 - 69). The median preoperative random GH level was 33.8 ng/mL (IQR: 22.1 - 77.5) and the mean preoperative IGF-1 level was 794.3 ± 266.7 ng/mL.
Prior to the surgery, one patient was treated with SSA and eight patients received DAs therapy. The majority of the patients (45, 91.8%) had undergone transsphenoidal surgery (TSS) and four (8.2%) of them had received the transcranial approach. The median Ki-67 labeling index detected by the MIB-1 antibody was 1.5% (IQR: 0.9 - 1.75%). The Ki-67 levels were less than 1% in 14 cases, 1% - 3% in 29, and over 3% in six.
Out of the 49 adenomas, 24 (46.1%) were pure GH-secreting adenomas and 25 (53.9%) were mixed GH/prolactin-secreting adenomas.
Based on the MRI reports, the median maximal adenoma diameter was 18.5 mm (IQR: 14.5 - 28.2) and macroadenomas accounted for 94% of the tumors. Unfortunately, we had no access to the follow-up data of eight patients at three months (or later) post-surgery. Among the remaining 41 patients, 31 (75.6%) had both biochemical and MRI follow-up data and 10 (24.4%) had only biochemical follow-up data at three months post-surgery. Sixteen (39%) patients achieved early remission three months after surgery based on the biochemical and/or MRI results. From among 25 patients who did not achieve remission three months after surgery, three subjects (one with SSA therapy post-surgery) met the criteria of remission in their final evaluations. Further follow-ups were available for 33 patients in the period ranging from three months to 10 years (median follow-up: 48.5 months, IQR: 13.25 - 67.25). Among these patients, 29 (87.9%) had both biochemical and MRI follow-up data and four (12.1%) had only biochemical follow-up data. The overall rate of final remission was 45.5% (15 out of 33). There were 13 postoperative complications, including eight cases of diabetes insipidus (DI), four cases with CSF leakages, and one patient with meningitis. Two patients died during the follow-up period because of septic shock and unknown origin.
There were 25 (59.5%) patients with remnant/recurrent disease based on their MRI and/or biochemical data at three-six months post-surgery. Interestingly, none of the adenomas with a Ki-67 index level of > 3% expanded cavernous sinus. Sixteen patients (37.2%) started SSA therapy after surgery, among whom 13 (81.25%) also received DAs.
4.2. Relationship Between Ki-67 and Demographics, Radiologic Data, and Outcome
The Ki-67 index level was slightly higher in men than in women, but the difference was not significant (1.5% vs. 1%, P = 0.323). The younger patients (under 30 years) had significantly higher Ki-67 index levels than those above 30 (2.25% vs. 1%, P = 0.036). On the other hand, there was a negative correlation between age and Ki-67 index level (r = -0.370, P = 0.009).
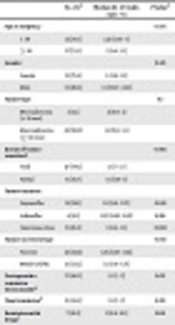
There was not any significant difference in the Ki-67 index level with respect to gender, tumor type, maximal tumor diameter, tumor invasiveness, tumor secretory type, and remission (Table 1).
| No. (%)a | Median Ki - 67 Index (Q25 - 75) | P Valueb | |
|---|---|---|---|
| Age at surgery, y | 0.036 | ||
| < 30 | 12 (24.5) | 2.25 (1.05 - 5) | |
| ≥ 30 | 37 (75.5) | 1 (0.9 - 1.5) | |
| Gender | 0.323 | ||
| Female | 35 (71.4) | 1. (0.9 - 1.5) | |
| Male | 14 (28.6) | 1.5 (0.97 - 2.62) | |
| Tumor type | 0.7 | ||
| Microadenoma (< 10 mm) | 3 (6.1) | 2 (0.9 - 2) | |
| Macroadenoma (≥ 10 mm) | 46 (93.9) | 1.5 (0.9 - 1.5) | |
| Extent of tumor resectionc | 0.968 | ||
| Total | 27 (64.3) | 1.5 (1 - 1.5) | |
| Partial | 15 (35.7) | 1.5 (0.9 - 3) | |
| Tumor invasion | |||
| Suprasellar | 16 (38.1) | 1.5 (0.9 - 3.75) | 0.596 |
| Infrasellar | 4 (9.5) | 1.25 (0.92 - 2.62) | 0.918 |
| Cavernous sinus | 11 (26.2) | 1 (0.9 - 1.5) | 0.098 |
| Tumor secretory type | 0.843 | ||
| Pure GH | 22 (46.8) | 1.25 (0.97 - 1.62) | |
| Mixed GH/PRL | 25 (53.2) | 1.5 (0.9 - 1.75) | |
| Postoperative remission three-monthd | 17 (40.5) | 1.5 (1 - 2) | 0.613 |
| Final remissione | 15 (45.5) | 1.5 (1 - 2) | 0.618 |
| Remission with drugsf | 7 (41.2) | 3 (0.9 - 8.5) | 0.193 |
Relationship of the Ki-67 Index with Demographics, Presurgical Data, and Outcomes in the Study Population
Early remission (three-six months after surgery) was detected in 40% of the patients, but no significant difference was detected in the Ki-67 index level between the groups (median: 51.5, IQR: 20.75 - 69.00 in patients without remission vs. median: 41.4, IQR: 8.00 - 69.75 in patients with remission; P = 0.565). Seven were controlled from among 16 patients who needed SSA therapy after surgery. The Ki-67 index was slightly higher in this group, but no significant difference was detected between responders and non-responders (3% vs. 1%, P = 0.193).
4.3. Correlation Between Ki-67 Index and Biochemical Data
There was no correlation between the Ki-67 index levels and the levels of GH (r = 0.098; P = 0.51) and IGF-1 (r = 0.248; P = 0.09) preoperatively, GH (r = -0.091; P = 0.64) and IGF-1 (r = -0.118; P = 0.59) three months post-surgery, and GH (r = -0.256; P = 0.28) at the last follow-up. Surprisingly, the IGF-1 at the last follow-up of 33 patients was negatively correlated with the Ki-67 index level (r = -0.48; P = 0.02).
4.4. Predictors of Outcome
In multivariable logistic regression analysis, the higher preoperative GH serum level was a significant predictor of three-month remission and final remission (odds ratios of 0.88 and 0.93, respectively). The Ki-67 index was not related to short and long-term remission. (Tables 2 and 3)
| Odds Ratio | 95% CI for Odds Ratio | P Valueb | ||
|---|---|---|---|---|
| Lower | Upper | |||
| Ki-67 index | 1.047 | 0.277 | 3.959 | 0.946 |
| Age at surgery | 1.143 | 0.970 | 1.348 | 0.111 |
| Gender | 19.896 | 0.556 | 711.743 | 0.101 |
| Preoperative IGF-1 | 1.010 | 1.000 | 1.021 | 0.059 |
| Preoperative GH | 0.881 | 0.801 | 0.969 | 0.009 |
| Cavernous sinus invasion | 0.166 | 0.002 | 14.645 | 0.432 |
| Suprasellar invasion | 1.271 | 0.067 | 24.064 | 0.873 |
Multivariable Regression Analysis for Predictors of Remission Three Months After Surgerya
| Odds Ratio | 95% CI for Odds Ratio | P Valueb | ||
|---|---|---|---|---|
| Lower | Upper | |||
| Ki-67 index | 1.578 | 0.563 | 4.421 | 0.386 |
| Age at surgery | 1.189 | 0.976 | 1.447 | 0.085 |
| Gender | 8.834 | 0.331 | 235.806 | 0.194 |
| Preoperative IGF-1 | 1.000 | 0.992 | 1.008 | 0.969 |
| Preoperative GH | 0.931 | 0.869 | 0.996 | 0.039 |
| Cavernous sinus invasion | 3.143 | 0.121 | 81.928 | 0.491 |
| Suprasellar invasion | 1.896 | 0.074 | 48.433 | 0.699 |
Multivariable Regression Analysis for Predictors of Final Remission After Surgerya
5. Discussion
In the present study, about 40% of the patients achieved remission three months post-surgery even though more than 90% of the tumors were macroadenomas. Haliloglu et al. (3) in a series of 103 patients and Babu et al. (14) in a series of 58 patients reported 51.5% and 69% of early remission, respectively.
This study revealed that the Ki-67 index level was significantly higher in younger adults while previous studies failed to demonstrate such a significant association (15, 16).
We observed that the Ki-67 index level was slightly higher in women; however, the gender difference was not significant. This finding is in line with the results obtained by Paek et al. (21) and Alimohamadi et al. (16). In contrast, some studies found significantly higher Ki-67 index levels in men (22) and women (23). Schaller (22), studying 10 male and 8 female patients with GH-secreting pituitary adenomas, demonstrated that the Ki-67 index level was higher in men (2.1 ± 0.4) than in women (1.5 ± 0.3).
In this study, the Ki-67 index level was not correlated with pre- or post-operative GH and IGF-I plasma levels, which is consistent with the studies by Fusco et al. (15) and Sarkar et al. (17).
Tumor secretory type analysis revealed no difference between pure GH and mixed GH/PRL tumor secretion concerning the Ki-67 level index whereas Sarkar et al. (17) found that PRL containing tumors had significantly lower MIB-1 index, and this mixed secretory type was associated with a higher postoperative remission rate.
In this study, the Ki-67 index level was independent of tumor size. Previous studies have often failed to found such a relationship (15-17). Jaffrain-Rea et al. (24), in a large prospective survey of 132 pituitary tumors including 42 GH-secreting adenomas, also did not find any correlation between the adenoma size and the Ki-67 level index, particularly for GH-adenomas.
Over the years, researchers have been seeking pathologic factors to predict the invasiveness of tumors. Cavernous sinus invasion is common in GH-secreting pituitary adenomas (25). Some previous studies have revealed a significant relationship between higher Ki-67 index level and tumor invasiveness. In a study by Fusco et al. (15), 68 acromegalic patients were recruited and followed. It was observed that tumors with cavernous sinus invasion had a higher mean level of Ki-67 index. This finding was confirmed by a number of studies (16, 24, 26); however, other studies (17) including ours did not observe such a correlation. Botelho et al. (27) investigated the presence of tumor markers (p53, Ki-67, and c-erb B2) in 38 patients with GH and/or prolactin-secreting adenomas. According to their results, c-erb B2 was a better predictor of tumor invasiveness.
A number of studies evaluated the probable predictors of surgery outcome in patients with acromegaly, some of which reporting invasion to surrounding structures especially cavernous sinus (28-30), gender (29), older age (16, 31), tumor size (28), and lower preoperative serum GH (28, 31, 32) and IGF-1 (31) as the predictors of surgical remission. In a recent study by Antunes et al. (30), 69 acromegalic patients were recruited. They investigated if some tumor characteristics such as the Ki-67 index level are the predictors of outcome. They found that only was the presence of cavernous sinus invasion a predictor of disease persistence.
In our study, surprisingly, higher preoperative GH levels were associated with better outcome. Some studies have revealed that lower Ki-67 index levels were associated with better outcome (15). However, some other studies, like ours, did not find any significant association (14, 17, 30, 33).
Probable reasons for the discrepancy between these results may be the small sample sizes and retrospective design of the studies. Hence, it is very necessary to establish prospective complementary multidisciplinary approaches in large series of GH-adenomas to achieve more consensus results.
5.1. Limitation
This was a single center retrospective study with relatively small sample size; this might have caused a failure to obtain significant results, as well as to follow up all patients for the long-term. Moreover, no data were available on the agreement of the two radiologists respecting MRI reports. We also had limited access to other biomarkers such as p53, p21, sparsely/densely granulated somatotroph adenoma, etc., which have been mentioned in several studies.
5.2. Conclusions
We found little evidence to ascertain the association of a tumor marker with some tumor behaviors and outcomes. However, the existing data on such an association are not congruent. The only significant correlation appeared to be between younger age and higher Ki-67 level index. Further research is required to find further biomarkers to use for predicting GH- adenomas behavior and treatment outcome.