1. Background
Endothelial dysfunction (ED) is an early sign of vascular compromise (1) and likely to be present prior to the development of atherosclerosis as it precludes cardiovascular disease (CVD). The major risk factors for CVD are obesity, diabetes, dyslipidemia and elevated blood pressure among others (2). Although CVD is rare in children (3), the risk factors are sometime present since childhood. Moreover, ED was noted in asymptomatic children and young adolescents who had risk factors to develop atherosclerosis without having CVD (4).
Currently, measured glycated hemoglobin (A1C) is the most commonly used and best evaluated marker for glycemic control and vascular complications prediction in diabetic patients (5). The Diabetes Control and Complication Trial (DCCT) Research Group 2000 states that strict glycemic control delays the onset and slows down the progression of diabetic vascular complications (6), which include microvascular (retinopathy, nephropathy, and neuropathy) and macrovascular (cardiac disease, peripheral vascular disease and stroke) diseases (3). Different treatment modalities (4), some aiming for better diabetes control, have shown to reverse the ED. Endothelial function is likely to be reversible in the early process of atherogenesis in children and young adults (4). Through the years, techniques to assess the function of the endothelium have been used clinically and routinely to monitor for CVD in adults, approved by the Food and Drug Administration (FDA). However, as of today, the use of these techniques continues to be part of research studies within the pediatric population. The EndoPAT (peripheral arterial tonometry) is a non-invasive device that measures endothelial function calculated as reactive hyperemic index (RHI). The greater the vasodilation, the higher the RHI (7). A Mayo clinic study (8) which compared the EndoPAT results to coronary injection of acetylcholine via cardiac catheterization, concluded that the normal RHI should be > 1.67. Studies of endothelial function using the EndoPAT device are sparse within the pediatric population and within those, different norms for children have been described. We used an RHI of 1.67 as the cutoff for normal within the pediatric population as other studies have also used (9, 10). There are other devices used to evaluate the endothelial function, for example, flow-mediated vasodilation (FMD) measured by ultrasound of the brachial artery, pulse wave velocity (PWV), radial artery tonometry (RAT) and ultrasonographic measurement of common carotid artery intima-media thickness (CIMT) (11).
2. Objectives
In this study, we aimed to assess the endothelial function in children and adolescents with diabetes type 1 (T1DM) and type 2 (T2DM) and in obese non-diabetics (NDM) using the EndoPAT device. We hypothesized that children and adolescents with poorly-controlled T1DM and T2DM as well as NDM obese children have ED reflected by lower RHI.
3. Methods
An Institutional Review Board (IRB) approved cross sectional study using the EndoPAT device was performed in children/adolescents with T1DM and T2DM who were poorly-controlled along with overweight/obese children. We enrolled subjects in the age range of 7 - 21 years from the pediatric endocrinology clinics at SUNY Downstate Medical Center and Kings County Hospital Center, Brooklyn, NY. Written informed consent and assent were obtained from subjects and parents as appropriate. We enrolled 33 T1DM subjects, 8 T2DM subjects and 17 NDM obese subjects. Poorly-controlled DM was defined as A1C ≥ 7.5% more than 6 months after diagnosis. Subjects with TIDM were treated with insulin and T2DM with metformin alone or combination of metformin and insulin. All NDM overweight/obese subjects were normotensive, (systolic blood pressure [SBP] and diastolic blood pressure [DBP] < 90th % based on age, sex, and height as per National Institutes of Health (NIH) guidelines (12), had normal fasting blood glucose concentrations (< 100 mg/dL), normal A1C (< 5.7%) and normal fasting lipid profile (total cholesterol < 170 mg/dL, low density lipoprotein cholesterol (LDL) < 110 mg/dL, high density lipoprotein cholesterol (HDL) > 45 mg/dL and triglyceride (TG) < 90 mg/dL) (13). A positive family history of diabetes, hypertension, hypercholesterolemia or cardiovascular disease was not an exclusion criteria. Mean A1C represents the average value of A1C within the last year, while latest A1C is the last A1C value obtained. This value was obtained within 3 months before the EndoPAT measurements. The following subjects were excluded: T1DM and T2DM with most recent A1C < 7.5%, abnormal renal function (micro-, or macro-albuminuria), retinopathy or neuropathy, presence of hyperlipidemia, pre-hypertension or hypertension, smoking, coronary artery disease, congenital heart disease, carotid artery disease, peripheral vascular diseases, inflammatory or infectious process within the last 3 months, using medications that would affect endothelial function such as oral contraceptive pills or malignancy. Endothelial function was assessed by the same physician with the EndoPAT 2000 after subjects had an overnight fast and within 2 months of the laboratory testing. The EndoPAT device allows non-invasive measurement of endothelial function without the disadvantages of conventional ultrasound measurement. It detects plethysmographic pressure changes in the fingertips caused by the arterial pulse and translates this into a peripheral arterial tone. It is an integrated software program that calculates the RHI automatically between the flow in the arm with reactive hyperemia and the control arm. The subjects lay on a reclining chair with arms supported and hands adjusted at heart level so fingers can freely hang for a minimum of 20 minutes in a quiet, temperature-controlled (72 - 75 degree F) room with dim light.
3.1. Statistics
All statistical analyses were performed using the Statistical Package for the Social Sciences (SPSS) for Windows version 16 (IBM Corp., Armonk, NY) and SAS version 9.4 (Cary, NC, USA). Data are reported as mean and standard deviation (mean ± SD) unless otherwise specify. Statistical procedures used included ANOVA, t-test, Mann-Whitney U and Spearman correlation. To estimate RHI predictors, multiple linear regressions with stepwise (backward) elimination for selection of variables were used. A P value of < 0.05 was considered statistically significant.
4. Results
Sixty-nine participants were assessed for enrollment in the study, 11 patients were excluded from the analysis (3 patients with A1C less than 7.5%, 3 patients with no EndoPAT device reading obtained, 1 patient with TIDM who developed nephropathy, and 4 patients who developed pre-diabetes). A total of 58 patients were included for data analysis. Different parameters are described in Table 1 while the statistical analysis of groups based on different factors is shown in Table 2.
| T1DM | T2DM | NDM | P Value | |
|---|---|---|---|---|
| Age, y | 12.7 ± 3.8 | 15 ± 2.20 | 12.8 ± 2.7 | 0.07 |
| BMI, kg/m2 | 20.8 ± 4.5 | 31.6 ± 8.3 | 34.1 ± 6.7 | 0.02 |
| BMI Z-score | 0.4 ± 0.9 | 1.7 ± 0.9 | 2.3 ± 0.3 | 0.02 |
| SBP, mmHg | 108.6 ± 9.7 | 116.8 ± 9.8 | 112.1 ± 10 | 0.87 |
| DBP, mmHg | 63.9 ± 8.2 | 69.1 ± 6.9 | 65 ± 7.5 | 0.92 |
| Total cholesterol, mg/dL | 157.6 ± 33.3 | 150.1 ± 43.2 | 149.4 ± 18.2 | < 0.01 |
| TG, mg/dL | 91.1 ± 42.4 | 90.6 ± 44.2 | 82.5 ± 36.4 | 0.82 |
| HDL, mg/dL | 55.9 ± 12.6 | 47 ± 13.8 | 43.3 ± 10.5 | 0.32 |
| LDL, mg/dL | 82 ± 26.1 | 85.2 ± 31.1 | 89.5 ± 18.8 | 0.03 |
| Years diagnosed | 5.74 ± 4.40 | 2.53 ± 1.91 | N/A | 0.05 |
| Mean A1C, % | 10.5 ± 1.9 | 9.7 ± 1.8 | 5.3 ± 0.3 | < 0.01 |
| Latest A1C, % | 10.8 ± 2.0 | 9.9 ± 1.7 | 5.2 ± 0.3 | < 0.01 |
| RHI | 1.4 ± 0.5 | 1.5 ± 0.5 | 1.4 ± 0.3 | 0.79 |
Parameters Among the Study Groups
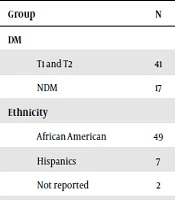
| Group | N | RHI (Mean ± SD) | P Value |
|---|---|---|---|
| DM | 0.86 | ||
| T1 and T2 | 41 | 1.42 ± 0.48 | |
| NDM | 17 | 1.40 ± 0.34 | |
| Ethnicity | 0.08 | ||
| African American | 49 | 1.45 ± 0.46 | |
| Hispanics | 7 | 1.16 ± 0.15 | |
| Not reported | 2 | ||
| Gender | |||
| Among all subjects | 0.04 | ||
| Males | 23 | 1.28 ± 0.36 | |
| Females | 34 | 1.51 ± 0.46 | |
| Among NDM group | < 0.01 | ||
| Males | 8 | 1.17 ± 0.19 | |
| Females | 9 | 1.61 ± 0.30 | |
| Among DM group | 0.36 | ||
| Males | 16 | 1.34 ± 0.42 | |
| Females | 25 | 1.48 ± 0.51 | |
| Puberty status | 0.08 | ||
| Non-pubertal | 12 | 1.25 ± 0.34 | |
| Pubertal | 45 | 1.46 ± 0.46 | |
| Latest A1C | < 0.01 | ||
| A1C < 10 | 18 | 1.70 ± 0.58 | |
| A1C ≥ 10 | 23 | 1.21 ± 0.19 | |
| Mean A1C | 0.09 | ||
| A1C < 10 | 19 | 1.55 ± 0.54 | |
| A1C ≥ 10 | 22 | 1.31 ± 0.38 |
Statistical Analyses of RHI Based on Different Factors
Among the 58 subjects (33 T1DM, 8 T2DM and 17 NDM obese children), 85% were African American, 60% female (F) and 79% pubertal. Mean (± SD) age of all participants was 13.1 years (± 3.42). Age, SBP, DBP, TG, HDL, pubertal status (PS) and RHI were not significantly different among them. There was no statistically significant difference in RHI among subjects in T1DM, T2DM, and NDM obese groups (P = 0.79). Within the T1DM group, the RHI was not statistically different between those who were obese and those with normal weight (P = 0.40). The RHI between T1DM and T2DM groups were 1.4 ± 0.5 and 1.5 ± 0.5 (P = 0.50), respectively. The RHI between T2DM and NDM obese group were 1.5 ± 0.5 and 1.4 ± 0.3 (P = 0.47), respectively. There was no difference between latest A1C among T1DM and T2DM groups, P = 0.29, and between the mean A1C amongst T1DM and T2DM, P = 0.27.
The mean RHI of the DM group (n: 41, 1.42 ± 0.48) versus the NDM obese group (n: 17, 1.40 ± 0.34) was similar (P: 0.86) regardless of type of diabetes or body mass index (BMI). No statistical difference in RHI was noted between African American and Hispanic subjects (1.45 ± 0.46 vs. 1.16 ± 0.15, P = 0.08). Males (M) had statistically significant lower RHI (1.28 ± 0.36) compared to F (1.51 ± 0.46), P: 0.04 but after adjusting for PS, the difference was not seen (P: 0.11). Subgroup analysis after adjusting for PS showed the difference persisted in the NDM group (P < 0.01) and no difference in RHI was seen between M and F within the DM group (P: 0.36). There was no statistically significant difference in RHI between pre-pubertal and pubertal children overall (P = 0.08). There was a statistically significant difference in RHI of the DM (T1 and T2) group with latest A1C < 10 (n: 18, 1.70 ± 0.58) compared to those with A1C ≥ 10 (n: 23, 1.21 ± 0.19), P: < 0.01. For every 1% increase in latest A1C, the RHI decreased by 0.097 (P: 0.01) adjusted for age, sex, and type of DM. No statistically significant difference was noted in mean A1C for those with A1C < 10 vs. those with A1C ≥ 10, P: 0.09. There was negative correlation between RHI and latest A1C in the diabetic group, r = -0.932, P: 0.01, while no correlation was noted with the mean A1C and RHI, r = -0.1, P: 0.34. Amongst patients with diabetes, there was no significant correlation between years of diabetes since diagnosis and RHI (r = - 0.74, P = 0.64)). After controlling for pubertal status, no statistically significant association was noted between RHI and age, P = 0.30).
5. Discussion
Our study used the Endopat device to evaluate the endothelial function in T1DM, T2DM and NDM obese children and adolescents. Previous studies done in children and adolescents with diabetes are mainly in T1DM patients (7, 10, 14-17). Prospective studies with the EndoPAT on T1DM adolescents revealed the presence of endothelial dysfunction in 76.7% (7), and a negative correlation between endothelial function and A1C (10). In our study, latest A1C (within 3 months prior to the use of the EndoPat) was an important predictor of endothelial function. Those who had higher A1C had worse endothelial function. Furthermore, diabetic patients with A1C values of 10% or above had statistically significant lower RHI in comparison to those with A1C values below 10%. Well-established norms for RHI in children and adolescents as well as long-term studies identifying the cut off A1C values where ED would develop or would be reversed are definitely needed.
Studies have shown that children with T1DM had significant ED when compared with non-diabetics (16, 17). Another study using the EndoPat demonstrated significantly lower RHI scores in obese adolescents when compared to lean controls (18). Krantz et al. demonstrated that the mean CIMT was greater in adolescents with T1DM when compared with controls (19). Bruzzi et al. demonstrated impaired FMD in T1DM children when compared with healthy controls (14). Ciftel et al. compared children with T1DM without complications and healthy children (15). Their study demonstrated ED (using FMD and CIMT methods) and increased arterial stiffness (using PWV) in the T1DM group (15). The SEARCH study compared the endothelial function in young patients with T1DM and T2DM using FMD, PWV and RAT, and concluded that youth with T2DM have worse arterial stiffness when compared with T1DM (P < 0.01) (20). In our study, we included T1DM and T2DM patients to assess if the type of diabetes influences endothelial dysfunction. Surprisingly, the arterial stiffness assessed by EndoPAT among patients with T1DM and T2DM were not significantly different. While the T1DM group had slightly higher A1C levels, there were not statistically different. Subjects with T1DM had more years of disease compared to those with T2DM although not able to reach statistical significance. Despite this, the endothelial dysfunction among these groups was similar. These findings are likely to the fact that in patients with T2DM, ED precedes overt hyperglycemia (21) by several years (2, 22), rather than caused by it. Hyperinsulinemia, insulin resistance and obesity are all factors affecting the endothelium in T2DM patients even before the disease is diagnosed. Conversely, ED in patients with T1DM develops after period of hyperglycemia.
In the Framingham Heart Study in adults, a significant inverse relation was demonstrated between endothelial function (determined by EndoPAT) and multiple cardiovascular risk factors such as male sex, BMI, total/HDL cholesterol, DM, smoking, and lipid-lowering treatment (23). Bruzzi et al. (14) did not find significant correlation between diabetes duration and FMD. Similarly, we did not find significant correlation of diabetes duration with RHI using the EndoPAT device. Of note, the EndoPAT device evaluates endothelial function in the microvasculature while the FMD evaluates such function in larger vessels. Selamet Tierney et al. (24) tested prospectively the reproducibility and feasibility of the EndoPAT and concluded that in healthy adolescents, EndoPAT is feasible, has excellent reproducibility, and provides an easy and reliable means of assessing endothelial function in the pediatric population.
In our study, the endothelial function as reflected by RHI was not statistically different when comparing diabetic patients (T1 and T2) with the NDM obese patients, even when taking type of diabetes and BMI into account. Both groups had a lower RHI score below 1.67 suggesting that each risk factor (diabetes or obesity) have a negative effect on the endothelium. A previous study done by Wadwa et al. (20) suggested that the difference in arterial stiffness between patients with T1DM and T2DM could have been due to elevated blood pressure and central adiposity. In our study, all subjects involved were normotensive at the time of the test though central adiposity was not measured.
As reported by Bhangoo et al., RHI increases during puberty in males as well as females. Endothelial function appears to improve during puberty as a result of hormonal changes, estrogen and DHEAS (25). Bruzzi et al. (14) reported that females had better endothelial function when compared to males in children and adolescents with T1DM. In our study, females had higher RHI score implicating better endothelial function when compared to males in the NDM obese group despite controlling for pubertal status. However, the difference was not sustained within the diabetes groups, T1DM and T2DM.
There have been conflicting data in regards to endothelial function in obese children. Landgraf et al. (26) and Mahmud et al. (27), reported lower RHI in obese children when compared with lean children. However, Tryggestad et al. (28) did not find such difference. In our study, no significant changes in the RHI were noted when the obese and T2DM patients were compared while controlling for BMI or glycemic control.
Our study has limitations. Despite being representative of our population, our sample size was small and unevenly distributed. Our study did not include non-diabetic lean subjects due to difficulty recruiting. Therefore, the findings should be interpreted with caution and further larger studies are needed. Nevertheless, our study provided relevant preliminary information for further studies regarding endothelial function in children.
5.1. Conclusions
Our data showed that patients with poorly-controlled DM as reflected by latest A1C of 10% or more had worse endothelial function as reflected by lower RHI score. Longitudinal studies are needed to elucidate the significance and implications, if any, of these findings. Assessment of endothelial function using EndoPAT device may be useful as part of routine care for DM and NDM obese children and adolescents.