1. Background
Metabolic syndrome (MetS) which is characterized by insulin resistance, hypertension, hyperlipidemia and abdominal obesity is a common health problem in older adults (1). The prevalence of MetS increases with age (2, 3). Previous studies demonstrated different impacts of MetS on health outcomes. For example, in some studies, it was shown that MetS could be associated with higher morbidity and mortality of cardiovascular diseases, functional dependence and poorer quality of life in the elderly (4). Some other studies represented the positive impact of Mets on survival benefits (5). The overall risk estimate of all-cause mortality for MetS was reported 1.17 (1.07 - 1.28) (6).
Incidence of MetS increases with age (1); it is estimated that about one-third of US adults (1) and 55% of adult population aged 60 and over have MetS (2). In Asian countries, the prevalence of MetS was ranged from 10% to 20% (7). In Iranian population, however, the overall weighted prevalence of this syndrome was reported 31% (95% CI: 28 - 35) (8). The rate of MetS was ranged from 10% to 60%, depending on sex, age and region (9).
2. Objectives
3. Methods
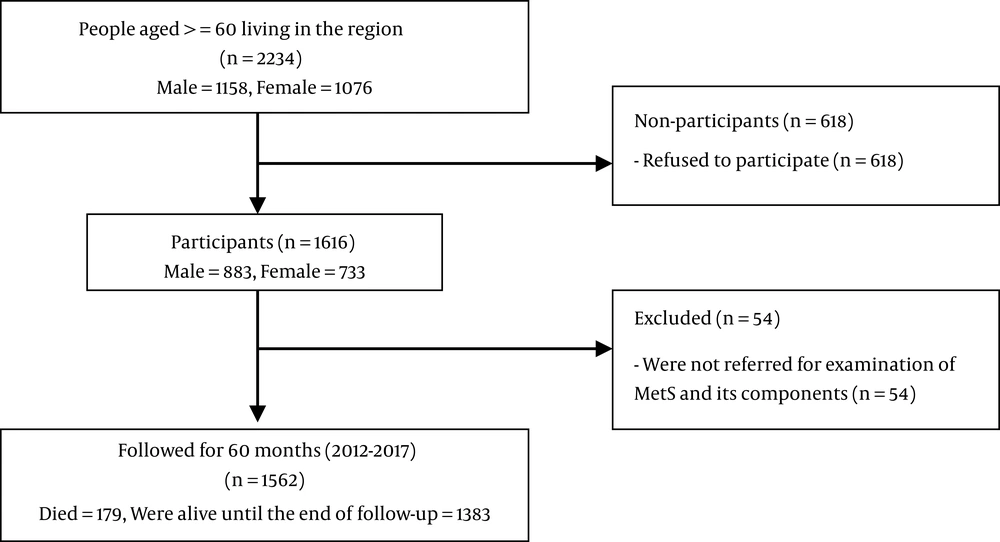
This prospective study was conducted based on data related to the Amirkola Health and Ageing Cohort Project (AHAP) (12, 13). Totally, 2234 people aged 60 years and over live in Amirkola, north of Iran. The entire population was invited to participate in the research, of whom 1616 individuals agreed to participate. For diagnosis of MetS, the subjects were invited for examination, but only 1562 individuals completed the examination. All the individuals were followed for 60 months to determine their vital or death status. The five-year survival rate and some related factors such as age, gender, and any of 25 chronic diseases (including cardiovascular, neurologic, psychological, renal, and hepatic disorders) have been assessed.
Metabolic syndrome was defined according to four sets of definition: Iranian definition (14), International Diabetes Federation (IDF) definition (15), 2001 Adult Treatment Panel (ATP) III (16) and 2005 Adult Treatment Panel (ATP) III (17). These four diagnostic sets are summarized in Table 1.
| Diagnostic Criteria | Measures and Categorical Cut-Off Points |
|---|---|
| Raised serum triglyceride (≥ 150 mg/dL), reduced HDL cholesterol (< 40 mg/dL in male and < 50 mg/dL in female), and raised blood pressure (systolic BP ≥ 130 and/or diastolic BP ≥ 85 mmHg) in addition to central obesity and previously diagnosed type 2 diabetes or raised fasting plasma glucose (FPG), as below: | |
| 2005 Adult Treatment Panel (ATP) III | Waist circumference: male ≥ 102 cm and female ≥ 88 cm with FPG ≥ 100 mg/dL |
| 2001 Adult Treatment Panel (ATP) III | Waist circumference: male ≥ 102 cm and female ≥ 88 cm with FPG ≥ 110 mg/dL |
| International Diabetes Federation (IDF) definition | Waist circumference: male ≥ 94 cm and female ≥ 80 cm with FPG ≥ 100 mg/dL |
| Iranian definition | Waist circumference ≥ 95 cm for male and female with FPG ≥ 100 mg/dL |
To assess mortality rate and exact time of death in the study population, several sources were used: (1) Iranian electronic death registration system (18); (2) health portal related to the Ministry of Health and Medical Education of Iran; (3) databank related to cemetery organization; (4) portal of the Authority Registration Office of Iran; (5) following the participants via their family physicians; and (6) contacting their family with phone numbers available in regional healthcare centers.
The research was approved by the Ethics Committee of Babol University of Medical Sciences with the approval code MUBABOL.REC.1394.54.
Statistical analysis was performed using the SPSS 17 software package. We used the Kaplan-Meier estimate and the log-rank test (to compare survival rate in different groups) for survival analysis. The proportional hazards assumption was visually checked using the log minus log plot. The cox proportional hazards regression model was used for multivariate survival analysis. Moreover, crude and adjusted hazard ratios with 95% confidence interval were calculated. Since a specific value was not assumed as an effect measure, two-sided significance was considered, by default. P value less than 0.05 was considered significant.
4. Results
In this study, 1562 older adults [863 (55.2%) males and 699 (44.8%) females] with the mean age of 69.3 ± 7.4 years were assessed. The mean age was 69.9 ± 7.6 years for men and 68.6 ± 6.9 years for women (P < 0.001). Moreover, of the population, one thousand (64.0%) were illiterate, 457 (29.3%) had primary or secondary school education, and 105 (6.7%) had high school or college education. The mean number of self-reported chronic diseases was 2.72 ± 1.94.
The MetS prevalence based on the four mentioned diagnostic criteria and frequency of MetS components is presented in Table 2. As shown, 71.9% of older adults, including 89.8% of women and 57.4% of men, had MetS based on 2005 ATP III diagnostic criteria. Furthermore, based on Iranian, IDF and 2001 ATP III diagnostic criteria, 74.3%, 68.8% and 66.7% of older adults had MetS, respectively.
| Variable | Male, No. (%) | Female, No. (%) | Total Number (%) | P Value |
|---|---|---|---|---|
| MetS based on ATP III 2005 | 495 (57.4) | 628 (89.8) | 1123 (71.9) | < 0.001 |
| MetS based on ATP III 2001 | 430 (49.8) | 612 (87.6) | 1042 (66.7) | < 0.001 |
| MetS based on IDF | 439 (50.9) | 636 (91.0) | 1075 (68.8) | < 0.001 |
| MetS based on Iranian definition | 561 (65.0) | 599 (85.7) | 1160 (74.3) | < 0.001 |
| Raised blood pressure | 682 (79.0) | 582 (83.3) | 1264 (80.9) | 0.034 |
| Raised serum triglyceride level | 382 (44.3) | 445 (63.7) | 827 (52.9) | < 0.001 |
| FPG ≥ 100 mg/dL | 467 (54.1) | 453 (64.8) | 920 (58.9) | < 0.001 |
| FPG ≥ 110 mg/dL | 287 (33.3) | 326 (46.6) | 613 (39.2) | < 0.001 |
| Reduced HDL cholesterol | 633 (73.3) | 684 (97.9) | 1317 (84.3) | < 0.001 |
| WC ≥ 95 cm (for Iranian definition) | 475 (55.0) | 413 (59.1) | 888 (56.9) | 0.109 |
| WC for male ≥ 102 cm and female ≥ 88 cm (for ATP III definition) | 233 (27.0) | 563 (80.5) | 796 (51.0) | < 0.001 |
| WC for male ≥ 94 cm and female ≥ 80 cm (for IDF definition) | 503 (58.3) | 661 (94.6) | 1164 (74.5) | < 0.001 |
Abbreviations: FPG, fasting plasma glucose; HDL, high-density lipoprotein; WC, waist circumference.
aThe four diagnostic criteria included: Iranian definition; International Diabetes Federation definition; 2001 Adult Treatment Panel III and 2005 Adult Treatment Panel III
The mean and standard deviation of MetS components including systolic and diastolic blood pressure, fasting plasma glucose, serum triglyceride, serum HDL cholesterol and waist circumference were 142.7 ± 22.2 mmHg, 81.5 ± 11.9 mmHg, 118.0 ± 45.7 mg/dL, 160.2 ± 84.0 mg/dL, 38.7 ± 4.4 mg/dL and 95.7 ± 10.5 cm, respectively.
None of the participants was lost to detect their vital or death status. The flowchart of the participants is presented in Figure 1. Moreover, the five-year survival rate of older adults with and without MetS and its components based on the four diagnostic criteria is presented in Table 3. This table shows that neither MetS (with any of the four mentioned diagnostic criteria) nor its components, except raised fasting plasma glucose (P < 0.05), had significant association with the five-year survival rate of older adults (P > 0.05). The mean survival time of older adults with and without MetS was 56 - 57 months. In addition, 88% - 89% of older adults with MetS and 87% - 90% of individuals without MetS were alive in five-year follow-up.
| Variables | Five-Year Survival Rate (Percent ± SE) | Mean Survival Time (Month ± SE) | P Value (Log-Rank) | ||
|---|---|---|---|---|---|
| With This Variable | Without This Variable | With This Variable | Without This Variable | ||
| MetS based on ATP III 2005 | 88.0 ± 1.0 | 90.0 ± 1.4 | 56.7 ± 0.3 | 56.8 ± 0.5 | 0.286 |
| MetS based on ATP III 2001 | 88.2 ± 1.0 | 89.2 ± 1.4 | 56.8 ± 0.3 | 56.7 ± 0.5 | 0.568 |
| MetS based on IDF | 89.3 ± 0.9 | 86.9 ± 1.5 | 57.1 ± 0.3 | 56.0 ± 0.5 | 0.147 |
| MetS based on Iranian definition | 87.9 ± 1.0 | 90.3 ± 1.0 | 57.0 ± 0.5 | 56.8 ± 0.3 | 0.216 |
| Raised blood pressure | 88.0 ± 0.9 | 90.9 ± 1.7 | 56.6 ± 0.3 | 57.3 ± 0.5 | 0.154 |
| Raised serum triglyceride level | 88.5 ± 1.1 | 88.6 ± 1.2 | 56.9 ± 0.4 | 56.6 ± 0.4 | 0.995 |
| FPG ≥ 100 mg/dL | 86.4 ± 1.1 | 91.6 ± 1.1 | 56.2 ± 0.4 | 57.6 ± 0.4 | 0.002 |
| FPG ≥ 110 mg/dL | 84.5 ± 1.5 | 91.1 ± 0.9 | 57.5 ± 0.3 | 55.6 ± 0.5 | < 0.001 |
| Reduced HDL cholesterol | 88.8 ± 0.9 | 86.9 ± 2.2 | 56.8 ± 0.3 | 56.3 ± 0.7 | 0.397 |
| WC ≥ 95 cm (for Iranian definition) | 89.4 ± 1.0 | 87.4 ± 1.3 | 57.2 ± 0.3 | 56.2 ± 0.5 | 0.195 |
| WC for male ≥ 102 cm and female ≥ 88 cm (for ATP III definition) | 89.4 ± 1.1 | 87.6 ± 1.2 | 57.2 ± 0.3 | 56.3 ± 0.4 | 0.239 |
| WC for male ≥ 94 cm and female ≥ 80 cm (for IDF definition) | 89.4 ± 0.9 | 85.9 ± 1.7 | 57.1 ± 0.3 | 55.8 ± 0.6 | 0.054 |
Abbreviations: FPG, fasting plasma glucose; HDL, high-density lipoprotein; WC, waist circumference.
The crude and adjusted hazard ratio of MetS and its components was assessed on the five-year survival rate of older adults using the cox proportional hazards regression model by considering age, gender and number of chronic diseases. The results are presented in Table 4. The table shows that only FBS had a significant crude hazard ratio. However, after adjusting age, gender and number of chronic diseases, MetS significantly decreased the survival rate of older adults (HR = 1.67; 95% CI: 1.16 - 2.41, P = 0.006).
| Variables | Crude Mortality Hazard Ratio (95% CI) | P Value | Adjusted Mortality Hazard Ratio by Age, Gender and Number of Chronic Diseases (95% CI) | P Value |
|---|---|---|---|---|
| MetS based on ATP III (2005) | 1.20 (0.86 - 1.69) | 0.287 | 1.67 (1.16 - 2.41) | 0.006 |
| MetS based on ATP III (2001) | 1.10 (0.80 - 1.50) | 0.568 | 1.48 (1.04 - 2.09) | 0.028 |
| MetS based on IDF | 0.80 (0.59 - 1.08) | 0.148 | 1.09 (0.78 - 1.54) | 0.609 |
| MetS based on Iranian definition | 1.25 (0.88 - 1.78) | 0.216 | 1.63 (1.13 - 2.36) | 0.010 |
| Raised blood pressure | 1.35 (0.89 - 2.02) | 0.156 | 1.14 (0.75 - 1.71) | 0.548 |
| Raised serum triglyceride level | 1.00 (0.75 - 1.34) | 0.995 | 1.29 (0.95 - 1.75) | 0.107 |
| FPG ≥ 100 mg/dL | 1.66 (1.20 - 2.28) | 0.002 | 1.96 (1.42 - 2.71) | < 0.001 |
| FPG ≥ 110 mg/dL | 1.81 (1.35 - 2.43) | < 0.001 | 2.05 (1.51 - 2.78) | < 0.001 |
| Reduced HDL cholesterol | 0.85 (0.58 - 1.24) | 0.398 | 0.97 (0.68 - 1.37) | 0.863 |
| WC ≥ 95 cm (for Iranian definition) | 0.82 (0.61 - 1.10) | 0.195 | 0.92 (0.68 - 1.24) | 0.570 |
| WC for male ≥ 102 cm, and female ≥ 88 cm (for ATP III definition) | 0.84 (0.68 - 1.12) | 0.239 | 1.19 (0.84 - 1.69) | 0.328 |
| WC for male ≥ 94 cm, and female ≥ 80 cm (for IDF definition) | 0.73 (0.54 - 1.01) | 0.055 | 0.97 (0.68 - 1.37) | 0.863 |
Abbreviations: FPG, fasting plasma glucose; HDL, high-density lipoprotein; WC, waist circumference.
All MetS components (based on 2005 ATP III) together with age, gender and number of chronic diseases have been considered in the cox proportional hazards regression model. Results showed that only FBS ≥ 100 had significant association with the survival rate (HR = 1.90; 95% CI: 1.36 - 2.65, P < 0.001) and other variables [raised blood pressure (HR = 1.10; 95% CI: 0.73 - 1.67; P = 0.642), including raised serum triglyceride level (HR = 1.14; 95% CI: 0.83 - 1.56; P = 0.420), reduced HDL cholesterol (HR = 1.01; 95% CI: 0.68 - 1.52; P = 0.958), and waist circumference (HR = 1.07; 95% CI: 0.75 - 1.51; P = 0.731) did not have significant effect on survival rate.
5. Discussion
The results showed that most of older adults had MetS that could decrease the five-year survival rate of this population.
Some previous studies presented MetS as the most potentially modifiable risk factor for cardiovascular and cerebrovascular diseases (11, 19), which can increase mortality rate in older adults. Moreover, 24% increased risk of cardiovascular mortality and 23% increased risk of all-cause mortality have been reported among older adults with MetS, as compared to those without this syndrome (6). Khosravi et al. showed that patients with MetS who had both raised blood pressure and fasting plasma glucose had a high incidence of myocardial infarction, cerebrovascular accident and mortality (10).
MetS itself was associated with reduction in the five-year survival of older adults. Contrary to our study, Chiang et al. showed that MetS had survival benefits in older men aged 75 and over in Taiwan, even after adjusting for age, diabetes mellitus, serum cholesterol, and triglyceride (5). Rachas et al. represented MetS components, but not MetS itself, for risk prediction of coronary heart disease (20). Considering that MetS is associated with diabetes mellitus (21) and cardiovascular complications (20), the effect of MetS on increased mortality rate can be justified.
In our research, only fasting plasma glucose ≥ 100 mg/dL out of MetS components had significant negative effect on the survival rate of older adults. Similar to our result, Chiang et al. reported that among different components of MetS, raised fasting plasma glucose had the greatest negative effect on survival (5). Rodriguez-Colon et al. revealed that increasing the number of MetS components caused to elevate the risk for incident stroke. People with raised blood pressure or fasting plasma glucose had higher risk for incident cerebrovascular accidents than patients with MetS without these two components (22). Hess et al. in their research reported that MetS was independently associated with a 70% elevation in the risk for sudden cardiac death (19). In another study, Krakauer et al. showed that hyperglycemia, hypertension, and low HDL made the greatest association of MetS with mortality hazard ratio (23). A cohort study with 20 years follow-up showed that patients with MetS and impaired blood glucose had the highest mortality rate in comparison with those who did not have MetS or hyperglycemia (24). Another prospective study represented that during 5-year follow-up, patients with MetS had significantly higher cardiovascular mortality and mortality risk was mostly attributed to hyperglycemia (25). An increase in all-cause mortality can be attributed to hyperglycemia and diabetes mainly because of increased cardiovascular deaths (26).
In our research, MetS components exposed older adults to higher mortality hazard. Therefore, an integrated health program is recommended to screen these components, especially hyperglycemia, and also to explore their cardiovascular and metabolic risk factors. Of course, proper health-promoting intervention including lifestyle modification, pharmacologic or non-pharmacological treatment programs can have beneficial impacts on the quality of life of older people and also on reduced mortality and morbidity among them.
The present study is notable for its large sample size, longitudinal study design, and including four diagnostic criteria for MetS definition. However, we did not follow the cause of death, which is an important limitation of the study. In addition, we followed the participants for five years. Of course, a longer follow-up of the study population can more precisely clarify long-term effects of MetS on survival of the elderly.
5.1. Conclusions
MetS and one of its components, high FPG, have significant associations with survival of older adults.