1. Background
The World Health Organization has focused strongly and seriously on the evaluation and measurement of treatment satisfaction and suggested the conduction of improvement plans across healthcare delivery since the beginning of the 21st century (1). During the last two decades, treatment satisfaction has been highlighted as not only an important parameter in measuring quality but also a reasonable result of individuals’ expectations and experiences of health services (2). One of the questionnaires related to treatment satisfaction with the particular aim of development and evaluation of treatment satisfaction among the patients already undergoing treatment is the Treatment Satisfaction Questionnaire for Medication (TSQM), which previously proved to be an effective scale of patients’ satisfaction with pharmacological treatment (3).
It was shown that patient satisfaction affects their health and treatment-related behaviors and, in turn, affects the success of treatment results (4). Satisfaction with the received services predicts the success of treatment, medical compliance, follow-ups, and proper use of services among patients. In other words, patients’ satisfaction with their prescribed medicine positively affects their continuity of drug treatment, proper use of medication, and medication adherence. Among patients with chronic diseases, the adverse effects of medications, such as the inconvenience of long-term use and ongoing side effects, could cause a patient to cease using their medicine without getting appropriate medical advice from their physician. Although dissatisfaction with the side effects of drugs and discomfort is prominent, poor adherence to drug use might jeopardize its effectiveness, thereby increasing the rate of disease progression (4, 5).
Substance use disorder is currently a public health issue with a high rate of morbidity and mortality. Maintenance therapy with methadone or buprenorphine is an effective treatment for opioid dependence in the community (6). Due to the increasing prevalence of substance abuse disorder in Iran and its destructive effects on patients’ quality of life, attention to the quality of life as an important factor in evaluating treatment outcomes and the effectiveness of treatment has been improved in the last three decades (7). The satisfaction of patients who use methadone maintenance treatment (MMT) services plays an important role in their therapeutic success and harmony; therefore, the realization of dosage adequacy, treatment satisfaction, side effects, and ease of use seem to be necessary (8). Therefore, it seems important to evaluate the satisfaction of opioid agonist usage as medicine using psychometric tools.
Numerous studies were conducted worldwide on the validity and reliability of the TSQM questionnaire (6, 9-12). A study conducted in the United States on eight different groups of patients showed that the reliability coefficient was acceptable with Cronbach’s alpha coefficient of the questionnaire, and therefore this questionnaire could be a good predictor of drug treatment in different patients (6). In 2016, the validity and reliability of the treatment satisfaction questionnaire in cardiovascular patients were assessed in Brazil and showed evidence of the acceptability and practicality of the TSQM (9). A 2018 study evaluated satisfaction with buprenorphine-naloxone treatment in heroin addicts. The aforementioned study was conducted with the help of the TSQM and evaluated general satisfaction, drug treatment, mental status, physical condition, personal performance, acceptance, and anti-addictive effects of secondary substances, and the results showed acceptable internal consistency and correlation coefficients among all studied factors. The results supported the validity and reliability of the questionnaire (10).
Considering both the role of MMT and buprenorphine as effective treatment methods in opioid dependence and the importance of treatment satisfaction in the success and therapeutic alliance, there is a serious requirement to measure treatment satisfaction for periodic care using a valid and multidimensional tool. To the best of our knowledge, there has not been a previous study conducted in Iran using the TSQM.
2. Objectives
The present study was conducted to evaluate the validity and reliability of the TSQM Persian version among patients referred to the methadone clinic of Shahid Beheshti hospital in Kerman, Iran, in 2020.
3. Patients and Methods
The current cross‑sectional study was carried out from February to March 2020 on 288 opioid-dependent patients undergoing maintenance treatment with opioid agonists referred to Shahid Beheshti MMT clinic of Kerman University of Medical Sciences in Iran. Shahid Beheshti Educational and Medical Center is a mental health center that offers focused mental health and substance abuse treatment. It provides specialized inpatient programs and outpatient services, one of which is providing psychotherapy and substitution therapy services for patients with substance use disorders. This center is sited in the Kerman city of the Kerman province since 1981. The participants were selected using the convenience sampling method. Given that the main purpose of the study was to assess the psychometric properties of an instrument, the sample size was considered to be 20 times per item of the tool (13).
The inclusion criteria were having morphine test approval, an indication of maintenance therapy, the minimum physical and cognitive ability to participate in psychological interventions, and consent to participate in the study. Patients who answered less than 90% of items were excluded (none in this study).
The questionnaire collects the demographic characteristics of the participants (e.g., age, gender, economic status, occupation, personal and family history of mental disorders, consumed substance, and type of treatment). The TSQM version II examines different aspects of treatment satisfaction and has 11 items in four subscales, including effectiveness (1 - 2), side effects (4 - 6), the convenience of use (7 - 9), and overall satisfaction (10 - 11). Each item is scored 1 (strongly disagree) to 7 (strongly agree), except for the fourth which checks the experience of side effects in the patient as yes/no (3).
The sum of the scores of each subscale is displayed as a number from 0 to 100. In this way, the sum of the scores of each subscale minus the number of the items in that subscale is divided by the maximum score minus the minimum score of that subscale multiplied by 100.
The questionnaire was translated from English to Persian by two professors fluent in English. Then, while comparing the text of the two translated versions, a Persian version was drafted. In the next step, the Persian-to-English translation was done by someone who did not know anything about the original version. In the last step, by comparing the two versions prepared in Persian and English, the required corrections were made, and the instrument was adapted culturally.
The questionnaire was validated by a panel of experts (five psychiatrists who are faculty members of Kerman University of Medical Sciences). The construct validity was evaluated by the analyzing methods of exploratory factor analysis (EFA) and confirmatory factor analysis (CFA). Principal component analysis was conducted on the items using the Kaiser criterion and a scree plot. The CFA was also employed using main model fit indices, including the goodness-of-fit index (GFI), adjusted goodness-of-fit index (AGFI), comparative fit index (CFI), root mean squared error of approximation (RMSEA), non-normed fit index (NNFI), and standardized root mean square residual (SRMR).
The internal consistency and repeatability of the TSQM version II were determined using Cronbach’s alpha and test-retest methods, respectively. To do so, 20 participants completed the questionnaire at an interval of 2 weeks. SPSS software (version 20.0) and LISREL software (version 8.80) were employed to analyze the data.
It took about 5 minutes for the participants to voluntarily complete the anonymous questionnaire. Moreover, they were given clear assurance that their data remain confidential and are used only for research purposes.
The study was approved by the Ethics Committee of Kerman University of Medical Sciences with the ethics approval code of IR.KMU.AH.REC.1396.141.
4. Results
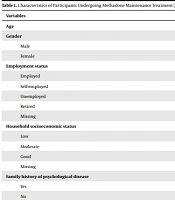
In total, 288 addicts with a mean age of 46.24 ± 11.5 years and a male majority of 85.4% with moderate economic status (58.0%) were assessed. Table 1 shows the characteristics of the participants.
| Variables | Values |
|---|---|
| Age | 46.24 ± 11.5 |
| Gender | |
| Male | 246 (85.4) |
| Female | 42 (14.6) |
| Employment status | |
| Employed | 75 (12.5) |
| Self-employed | 154 (53.5) |
| Unemployed | 27 (9.4) |
| Retired | 73 (25.03) |
| Missing | 4 (1.4) |
| Household socioeconomic status | |
| Low | 114 (39. 6) |
| Moderate | 167 (58. 0) |
| Good | 6 (2.1) |
| Missing | 1 (0.3) |
| Family history of psychological disease | |
| Yes | 114 (39.6) |
| No | 174 (60.84) |
| Concurrent psychological disorder | |
| Yes | 166 (57.6) |
| No | 122 (42.4) |
| Type of maintenance treatment | |
| Methadone | 207 (71.9) |
| Buprenorphine | 81 (28.1) |
Table 2 shows the mean and standard deviations for each of the four TSQM subscales and measures of variability. Mean values on the different TSQM factors stood within the variety of intermediate to high values of each subscale (Table 2).
| Variables | Global Satisfaction | Convenience | Side Effects | Effectiveness |
|---|---|---|---|---|
| Mean | 80.67 | 79.79 | 79.09 | 79.97 |
| Standard deviation | 12.15 | 11.99 | 12.14 | 11.7 |
| Median | 83.33 | 83.33 | 83.33 | 83.3 |
| Minimum | 33.33 | 33.33 | 44.44 | 41.6 |
| Maximum | 100 | 100 | 100 | 100 |
Cronbach’s alpha of 0.80 confirmed the internal consistency of the TSQM Persian version II, and its repeatability using the test-retest method was calculated at 0.9.
The Kaiser‑Meyer‑Olkin measure was measured at 0.82, and Bartlett’s test of sphericity was statistically significant (P = 0.001, χ2 = 1507.02, df = 28), which indicates the adequacy of sampling and justifiability of the analysis. Three factors were extracted, and the proportion of the variance explained by these factors was 82.41% (58.74%, 15.28%, and 8.39% for the first, second, and third factors, respectively). Table 3 shows exploratory factor loading of the TSQM Persian version II.
In CFA, RMSEA = 0.15, SRMR = 0.05, GFI = 0.9, AGFI = 0.8, CFI = 0.95, incremental fit index = 0.95, and NNFI = 0.92 showed that the model had a reasonable fit to the data (Table 3).
| Item No. | Items | Factor | Factor Loading |
|---|---|---|---|
| 1 | Preventing or treating condition | 1 | 0.86 |
| 2 | Relief of symptoms | 1 | 0.87 |
| 4 | Interference With physical function | 2 | 0.70 |
| 5 | Interference w/mental function | 2 | 0.86 |
| 6 | Interference with mood or emotions | 2 | 0.67 |
| 7 | Ease of medication use | 3 | 0.88 |
| 8 | Planning for medication use | 3 | 0.89 |
| 9 | Frequency of medication use | 3 | 0.78 |
5. Discussion
The current study aimed to determine the psychometric properties of the TSQM Persian version II. In the analyses, the Persian version of the instrument had acceptable psychometric properties. In terms of reliability, the instrument had appropriate internal consistency and excellent repeatability. Atkinson et al. found that TSQM version I and version II had good internal consistency in all its subscales (3). Nevertheless, the current values were slightly lower than those stated by Atkinson et al. In the present study, the highest variability subscale was global satisfaction followed by effectiveness, convenience, and side effects. Furthermore, Bharmal et al. demonstrated that the 9-Item Treatment Satisfaction Questionnaire for Medication (TSQM-9), in which the items related to side effects were not included, had acceptable reliability (14). Before this study, Trujols et al. evaluated the TSQM Spanish version in a sample of methadone-maintained patients and presented an acceptable level of internal consistency (0.70) that remains slightly lower than the results of this study (15).
In terms of validity, EFA showed justifiability of the analysis and the goodness of fit measures in CFA and similar to Atkinson et al.’s study (3) indicated that the model had a reasonable fit to the data. Furthermore, most of the goodness of fit indices in the TSQM-9 had acceptable levels.
This study showed that the average satisfaction score of effectiveness, side effects convenience, and the global satisfaction score is about 80%, which is a high and acceptable value. These results are consistent with the results of Fiellin et al.’s study conducted on 53 opioid-dependent patients that assessed the long-term treatment with buprenorphine (16).
The effective management of diseases requires further attention to the treatment satisfaction assessment. It is extremely important due to the growing number of patients on long-term medication therapy in addition to the prevalence of chronic diseases among senior citizens.
In general, there is not significant evidence on how patients’ satisfaction could affect their medication adherence behavior, and that could potentially amplify the healthcare costs. Therefore, it is vital to identify the reasons for non-adherence to reduce the cost of treatment and the use of medical resources. It is realized that patients’ perspective on treatment is equally important, and physicians should be able to have a profound view on the regular assessment of treatment satisfaction to provide alternative medication for patients with poor medication adherence (3-5).
The accuracy of data remains a limitation of the current study as the questionnaires might not have been completed with sufficient honesty. Furthermore, this study was only performed among the patients of the methadone clinic of Shahid Beheshti Mental Hospital in Kerman and cannot be generalized to other populations. Moreover, this is a cross-sectional study, and further longitudinal studies in this field are necessary.
5.1. Conclusions
It seems that different versions of the TSQM (including the Persian version) in different studies and populations had appropriate psychometric properties and could be embedded as simple, valid, and reliable instruments to evaluate treatment satisfaction in clinical settings. With precise use, this tool provides a method to evaluate and compare patients’ satisfaction with different types of medication and might be a useful reference for physicians not only in better understanding patients’ experiences of specific medicine side effects or discomforts but also in optimizing drug dosage or medication guide. Therefore, clinical care turns out to be further effective.