1. Background
Gelatin is a soluble protein obtained by partial hydrolysis of collagen, the main insoluble fibrous protein constituent on bones, cartilages and skin (1). Unique collagen and gelatin structures influence their physical properties, such as solubility, swelling, water uptake, moisture absorption, water evaporation, transparency, color, odor, gel strength, and thermal stability. Physical properties of gelatin itself influence gelatin quality and potential applications in food and pharmaceutical industries (2, 3). The food and pharmaceutical applications of gelatin are mainly based on gel-forming, film-forming and viscoelastic properties. Recently, an increasing number of new applications have been found for gelatin in products such as emulsifiers, foaming agents, colloid stabilizer, hydrogels, fining agents, packaging materials, wound dressing and micro-encapsulating agents (4, 5). The highly hydrophilic nature of gelatin is its disadvantage when considering the use of gelatin films as protective barriers because they tend to swell or dissolve when contacting with the surface of foodstuffs with high moisture content. Consequently, the current trends in designing gelatin-based biodegradable materials for food packaging and medical applications are focused on developing films with improved mechanical and water resistance properties by combining gelatin with other biopolymer, synthetic polymer, plasticizer as well as cross-linker agents (6-10).
Gelatin is reported as one of the first carriers of bioactive components. Enrichment of gelatin films with natural antioxidant and/or antimicrobial substances will extend the functional properties of these biodegradable films and provide active packaging biomaterials. There is growing interest in using plant extracts as natural sources of antioxidant/antibacterial compounds in formulating gelatin films (11-14). In this context, plant essential oils and their main components are gaining a wide interest in health industry for their potentials as antioxidant and antimicrobial agents, since they are generally recognized as safe (GRAS) (15, 16).
To our knowledge there is no report on the on the antioxidant and antimicrobial activity of gelatin films incorporated with Trachyspermum ammi (TAM) is a useful plant in the umbelliferae family. In traditional medicine the plant is used for the treatment of different diseases, such as asthma, epilepsy, stomachache, flatulence, intestinal parasites, weak digestion and influenza.
In this study the gelatin films with antioxidant and antimicrobial activities were utilized from gelatin solutions containing different Trachyspermum ammi essential oil TAM concentrations. The mechanical, water solubility, water swelling, water vapor permeability and color properties of gelatin/TAM films were measured according to the American Society for Testing and Materials (ASTM). Antioxidant activities of the gelatin films incorporated with TAM were examined using 2′-azino-di (3-ethylbenzthiazoline-6-sulfonate) (ABTS) decolorization. The gelatin films containing TAM were individually tested against two Gram-negative bacteria (Pseudomonas aeroginosa and Escherichia coli) and two Gram-positive bacteria (Staphylococcus aureus and Bacillus subtilis) commonly found in human pathogenesis.
2. Objectives
The current study proved that gelatin films grafted with TAM have excellent physical, mechanical and water resistance, as well as good antioxidant and antibacterial activities. Flexibility, strength, water resistance, strong antibacterial, and excellent mechanical properties make gelatin films grafted with TAM a suitable antimicrobial active food packaging.
3. Materials and Methods
3.1. Preparation of Gelatin Film Forming Solutions
To prepare gelatin film forming solutions, 10g of bovine gelatin powder (Sigma-Aldreich, Germany) was dissolved into 80mL of distilled water at ambient temperature and the temperature was increased to 60°C using a Hotplate-stirrer and the mixture was stirred for 30 min at this temperature. After cooling to 37°C, TAM (obtained by hydro-distillation) with different concentrations (2, 4, 6, and 8% w/w based on the weight of the gelatin powder = 1, 2, 4, and 8mg/mL based on the gelatin solutions) as antimicrobial agent, glycerol (25% w/w based on the weight of the gelatin powder = 25 mg/mL based on the gelatin solution) (Merck, Germany) as plasticizer and glutaraldehyde (2% w/w based on the weight of the gelatin powder = 2 mg/mL based on the gelatin solution) (Merck, Germany) as cross-linker were added to gelatin solutions and then distilled water was added to the final 100mL and homogenized (7, 17).
3.2. Preparation of Gelatin Films
To cast the films, 10mL of gelatin film forming solutions containing different TA concentrations were transferred into the polyester Petri dish (Farazbin Kimia Co., Teheran, Iran, the radius of 74mm) and placed at room temperature until the films dried. The obtained dry films were peeled off and stored until analysis. Thicknesses of films were measured to the nearest 0.01mm with a digital micrometer (The L.S. Starrett Co. LTD. Great Britain, Uk) and the average was taken 120 ± 5µm.
3.3. Mechanical Properties of Films
The mechanical properties of the gelatin films were measured according to the American Society for Testing and Materials 638-02a (ASTM D 638-02a) in texture analyzer (TA.XT Stable Micro System UK). Gelatin films containing different concentrations of TAM were transferred to a closed container with relative humidity of 65% (saturated sodium nitrite vapor) and left for equilibrium for 48hours before mechanical testing. Gelatin films were cut into the rectangles with length of 60mm, width of 10mm and thickness 0.12mm. The tensile strength test was then performed by stretching the film at pretest, test and posttest speeds of 1, 1, and 10mm/s, respectively. The net length between the jaws for all films was almost constant at about 20mm. The texture analyzer ran at auto force mode with the trigger force of 5gr (0.049 Newton). From stress-strain curves, two parameters were measured: 1) tensile strength (TS) was measured as maximum stress and 2) elongation at break (EAB) where the film was torn (3, 17):
TS (N/m2) = (Breaking force / Cross-sectional area of sample)
EAB (%) = [(Increase in length at breaking point / Initial length)] × 100.
The area of film used for each experiment was 6 × 1cm2. However, 2cm of the films were within the jaws, therefore the initial length of the film was taken as 4cm2. All tests were the means of at least three measurements.
3.4. Water Solubility of Films
To determine solubility , one piece of films (60mm × 10mm × 0.11mm) was placed in an oven at 104°C for 24 hours and initial dry weight (Wi) was calculated. Then, the dried films were immersed into a 100mL Erlenmeyer flask containing 50mL of distilled water and placed inside the shaker for 24 hours at 25°C (Incubator with in ventilator, Pars Azma Co. Tehran, Iran). Thereafter, the films were taken out and transferred to the oven of 104°C for 24 hours and the final dry weight (Wf) was calculated. The weight loss or solubility percentage (S%) was determined by the following formula: S (%) = [(Wi-Wf) / Wi] × 100 (3, 17). All tests were the means of at least three measurements.
3.5. Swelling of Films
The gelatin films were dried in an air-circulating oven at 104°C for 24 hours until reaching the constant weight before swelling test (Wi). Squares were cut with the dimensions of 20mm × 20mm × 0.12mm for the swelling experiment. Each sample was immersed into a 100mL Erlenmeyer flask containing 50mL of distilled water. The samples were kept at room temperature during the swelling experiment (24hours). Each sample was taken out of the flask after 24hours, wiped between filter papers to remove the excess surface water and weighed (Wf). The gained weight or swelling percentage (SW %) was calculated by the following equation (3, 17): SW (%) = [(Wf- Wi)/ Wi] × 100. All tests were the means of at least three measurements.
3.6. Water Vapor Permeability of Gelatin Films
The Water vapor permeability (WVP) of the films was determined according to the ASTM E96-95 method. The films were conditioned for 24hours at 25°C and 75% relative humidity before WVP determination. Film samples were mounted on an aluminum cup (height and diameter were 2.1 and 5.6cm, respectively). The cup was filled with 20g of silica gel and covered with a film specimen. The cup was placed at 25°C and 75% relative humidity in desiccators. The weight of the cup was measured at 3hours intervals during one day. Simple linear regression was used to estimate the slope of mass change vs. time plot. The WVP was calculated by the following formula (3, 17):WVP (g.mm/m2.kPa.h) = [(WVTR × T)]/ ΔP. Where water vapor transmission rate (WVTR) is the slope per film area (g/m2.h), T is the film thickness (mm), and ΔP is the partial water vapor pressure difference (kPa) between the two sides of the film (4.2449 kPa at 30°C).
3.7. Antioxidant Activity of Films
Antioxidant activities of the films were determined by decolorization method with 2, 2′-azino-di (3-ethylbenzthiazoline-6-sulfonate) (ABTS, Sigma, Germany) (18). The method was modified to detect the continuous antioxidant release from films. The release tests were performed in 24 well plates. Briefly, cuts (10mm × 10mm × 0.12mm, 20mg) from different parts of the films containing 1, 2, 5, and 10% of the ZMO were added to 2.0mL of diluted ABTS radical solution (7mM ABTS and 2.54mM potassium per sulfate, A734 = 1 ± 0.1). Films without ZMO were used as blank. The program was adjusted to record the absorbance values after shaking the 24 well plates for 30s by a plate reader (BioTekElx 808, Winooski, VT, 05403, USA). The data were recorded up to the steady state was reached for each sample. A standard curve of ascorbic acid ranging from 0.44 to 15.76mg/mL was prepared. Antioxidant activity was expressed as mg ascorbic acid equivalents per gram of films using the standard curve.
3.8. Antibacterial Activity of Films Using Disc Diffusion
All microorganisms obtained from the Persian Type Culture Collection (PTCC), Tehran, Iran. The films were individually tested against two Gram-negative bacteria [P. aeruginosaPTCC 1074 (ATCC 9027 and E. coli PTCC 1330 (ATCC 8739)] and two Gram-positive bacteria [S. aureus PTCC 1112 (ATCC 6538) and B. subtilis PTCC 1023 (ATCC 6633)]. The antibacterial activity was measured according to the standard practice for determining resistance of synthetic materials to bacteria (ASTM G22-76). To investigate the antimicrobial activity of the films by disc diffusion, 30mm diameter discs (thickness of 0.12mm) were cut from different parts of the films and sterilized by autoclaving for 30min at 120°C (19). Bacterial suspensions with a turbidity equivalent to a McFarland 0.5 standard were prepared (108 colony-forming units CFU/mL) and then diluted to 105 CFU/mL with Luria-Bertani (LB). The adjusted bacterial suspensions (0.1mL) were spread onto the nutrient agar plates (Farazbin Kimia Co., Teharan, Iran) containing LB. Subsequently, the discs were placed in direct contact with the agar medium. Plates were inverted and incubated at 37°C for 24hours (Incubator with in ventilator, Pars Azma Co. Tehran, Iran). Films without TAM under the same condition were used as the controls. The diameters of clear inhibition zones, including the diameter of the disc, were measured with a ruler and the results were used to evaluate antibacterial potential of the films.
3.9. Antibacterial Activity of Gelatin Films Using Colony Counting
The bacterial colony counting assays were conducted according to the Clinical and Laboratory Standards Institute (CLSI) and ASTM G22-76. Bacteria strains were suspended in LB media and the densities were adjusted to 0.5 McFarland standards at 640 nm (108CFU/ mL) and then diluted to 105CFU/mL with LB. A sample film with 30 mm diameter was placed in a 10mL liquid culture containing 10µL of the cultures. Then, the sample was incubated at 37ºC for 24hours (Shaking Incubator, Shin Saeng, Fine Tech, Korea). From the incubated samples, a 100µL solution was taken and diluted with the appropriate dilution factor and the final diluted microbe solution, and then it was plated and distributed onto the nutrient agar plates (Farazbin Kimia Co., Tehran, Iran). The plates cultured with the films without TAM under the same condition were used as the controls. All plates were incubated at 37ºC for 24 hours and the number of formed colonies was counted. The antibacterial efficacy of the films was calculated by the following equation (20): Colony reduction (%) = [(Number of colonies in the test samples - Number of colonies in the control)/ Number of colonies in the test samples] × 100.
3.10. Statistical Analysis
Data wereexpressed as the means ± standard deviations of at least three independent experiments. The significant differences between treatments were analyzed by a one-way analysis of variance (ANOVA) and Duncan tests at P < 0.05by SPSS version 18 (SPSS18, Abaus Concepts, Berkeley, CA) and Prism 5 (Graph Pad, San Diego, USA) software.
4. Results
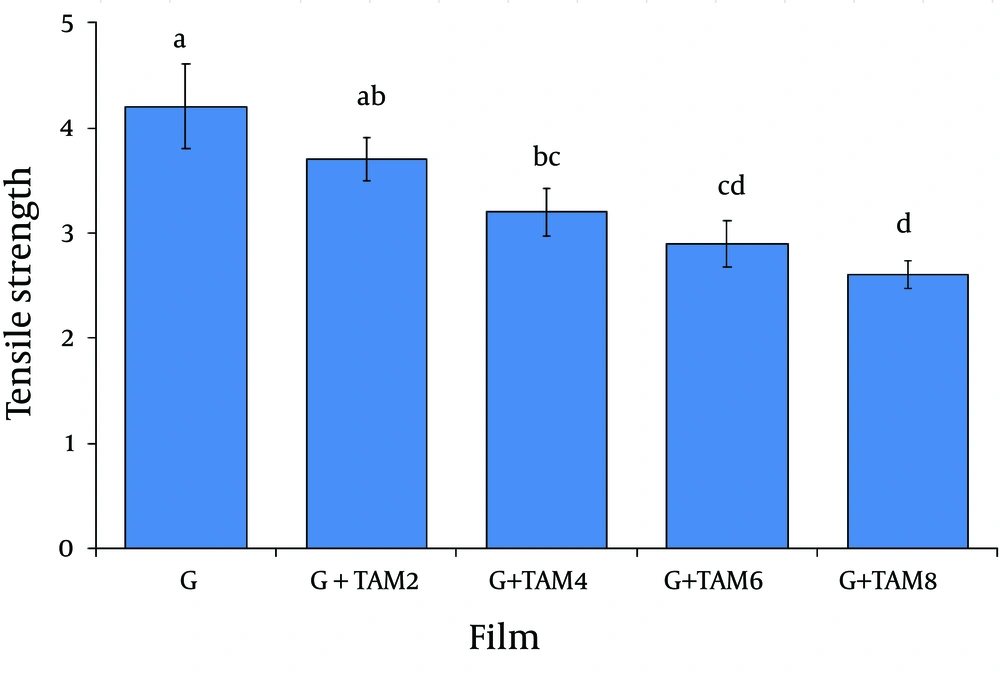
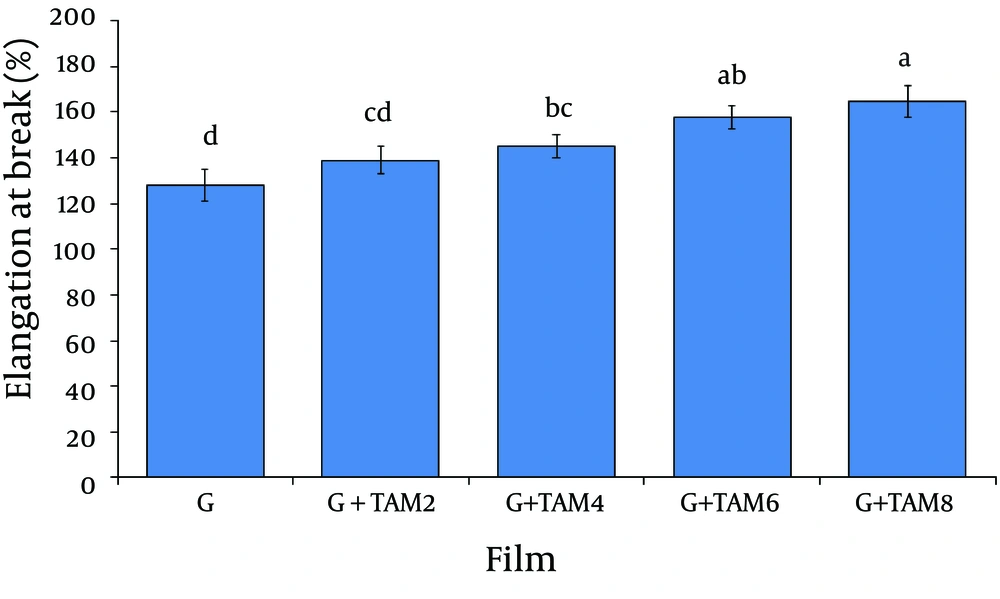
4.1. Mechanical Properties of Films
The mechanical properties of gelatin films are shown in Figure 1 and Figure 2. Tensile strength and elongation at break are parameters that relate mechanical properties of films to their chemical structures. Tensile strength and elongation at break of gelatin films cross-linked with glutaraldehyde were 4.2 ± 0.4MPa and 128 ± 7%, respectively. Incorporation of ZMO into gelatin films caused a significant decrease in tensile strength (from 3.7 ± 0.21 to 2.6 ± 0.13MPa) and increase in elongation at break (from 139 ± 6% to 165 ± 7%) (P < 0.05).
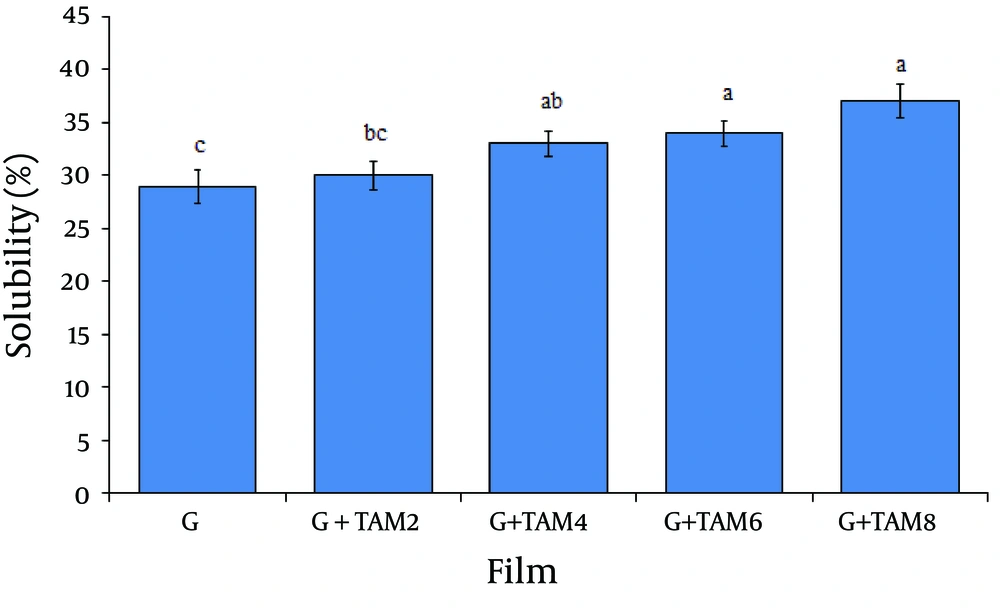
4.2. Solubility Determination of Films
The solubility percentages (weight loss) of gelatin films are summarized in Figure 3.The solubility percentage of gelatin films was 29 ± 1.6%. Incorporation of TAM into the films caused a significant increase in the solubility (From 30 ± 1.3%to 37 ± 1.6%) of gelatin films, dose-dependently (P < 0.05).
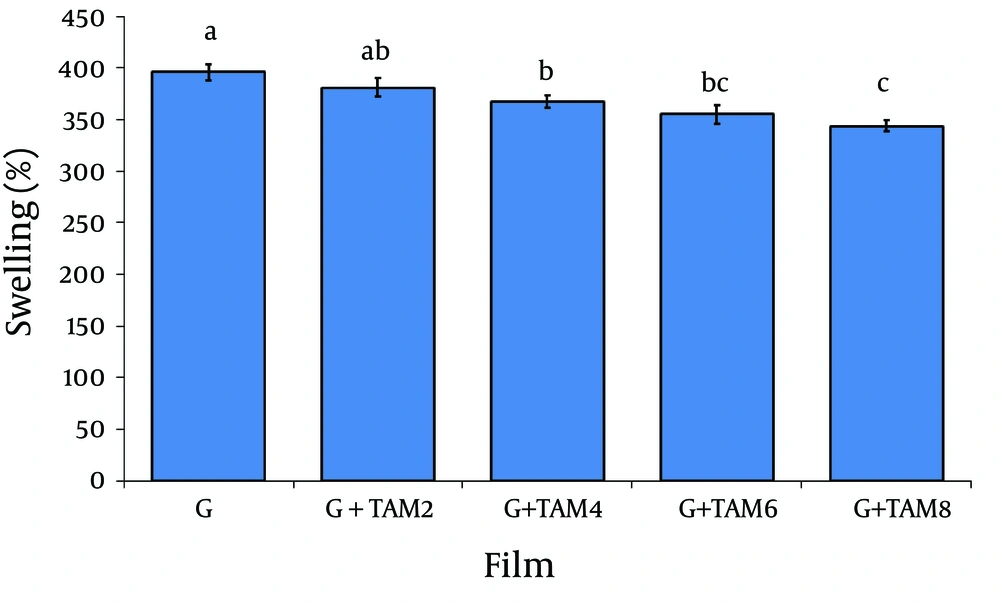
4.3. Swelling Capacity of Films
The results of swelling capacity of gelatin films are summarized in Figure 4. The swelling percentage for gelatin films was 396 ± 8%.Incorporation of TAM into the films caused a significant decrease in the swelling (from 381 ± 9%to 344 ± 5%) of the films (P< 0.05).
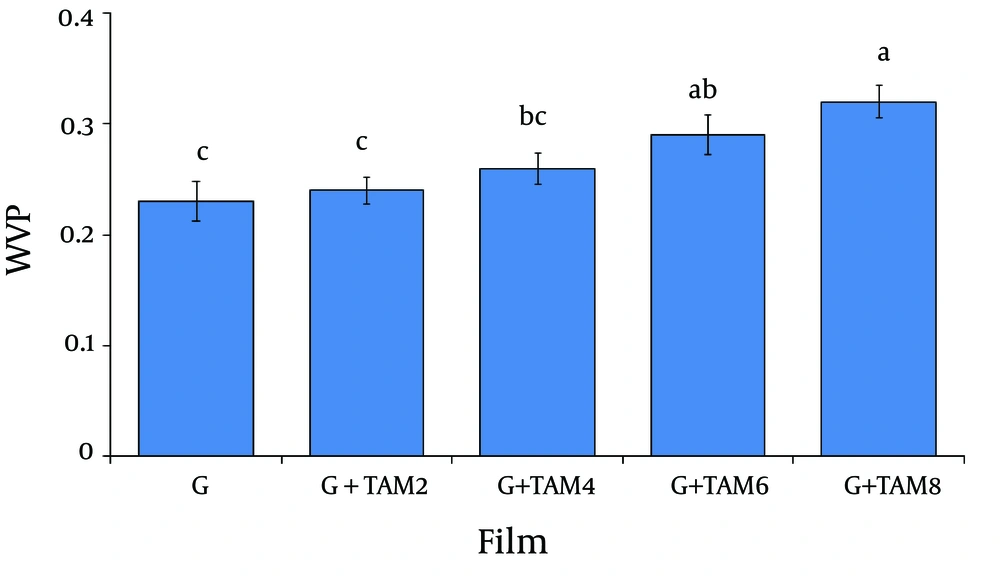
4.4. Water Vapor Permeability of Films
The results of water vapor permeability (WVP) of gelatin films are summarized in Figure 5.The WVP for gelatin films was 0.23 ± 0.018g.mm/kPa.m2h.Incorporation of TAM into the films caused a significant increase in WVP (from 0.24 ± 0.012 to 0.32 ± 0.015 g.mm/kPa.m2.h) of the films, dose-dependently.
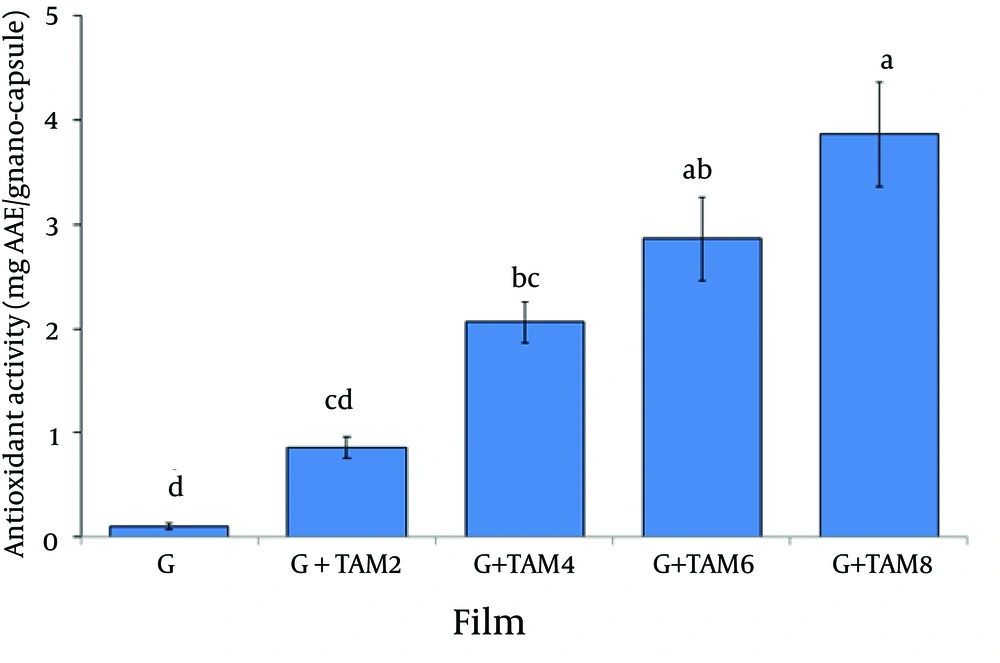
4.5. Antioxidant Activity of Films
Antioxidant activity of the gelatin films incorporated with different carvacrol concentrations was determined by ABTS decolorization method and expressed as mg ascorbic acid equivalent per gram of films (Figure 6). The gelatin films free of TAM showed very low activity against the ABTS decolorization.
Antioxidant Activity of Gelatin Films Incorporated WithTrachyspermum ammiEssential Oil (TAM). TAM2, TAM4, TAM6 and TAM8 are 2%, 4%, 6% and 8% w/w TAM Based on the Gelatin Powder. The antioxidant activity was expressed in milligram ascorbic acid equivalent (AAE) per gram incorporating different concentrations of TAM. Mean values with different letters within a column are significantly different by Duncan’s multiple range tests at (P < 0.05).
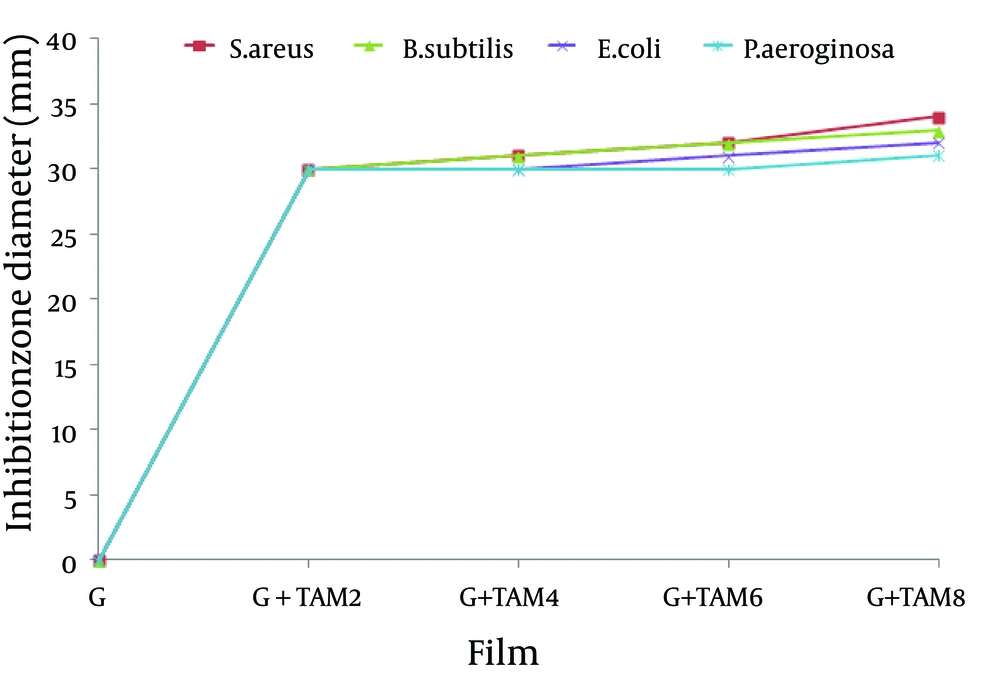
4.6. Antibacterial Activity of Gelatin Films
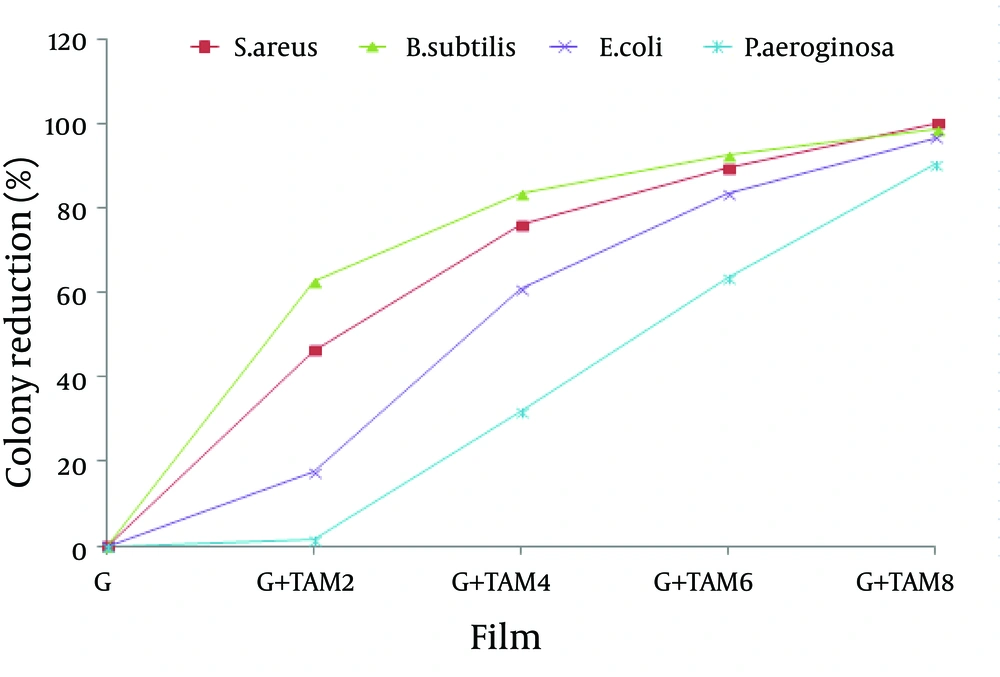
Antibacterial assay of gelatin films incorporated with TAM expressed by disc diffusion method and viable colony counting assay. The results of disc diffusion are summarized in Figure 7. The initial diameter of all films was fixed at 30mm. The diameters of clear inhibition zones, including the diameter of the disk, were used to analyze the antibacterial activity. According to the obtained results, all the gelatin films free of TAM showed no activity against the tested bacteria. The inhibitory zones of the gelatin films containing different concentrations of TAM were obvious. The antibacterial activity of gelatin films containing different TAM concentrations was the highest against B. subtilis and S. aureus followed by E. coli and then P. aeroginosa. According to the Swiss Norm (SN) 195920-ASTM E 2149-01, any compound showing zone inhibition of > 1 mm, is considered as a good antibacterial agent. Thus gelatin films incorporated with TAM are effective against both Gram-positive and gram-negative bacteria while they have more effect on Gram-positive bacteria rather than the Gram-negative bacteria. The results of colony reduction percentage are summarized in Figure 8. According to the obtained results, the antibacterial activity of gelatin films containing different TAM concentrations was the highest against S. aureus followed by B. subtilis, E. colli, and then by P. aeroginosa. Gelatin films from the skin of unicorn leatherjacket incorporated with essential oils (17), chitosan films incorporated with thyme oil (21), and soy edible films incorporated with thyme and oregano essential oils displayed excellent antibacterial activities, similar to the results of the current study.
Antibacterial Activity of Gelatin Films Incorporated With Trachyspermum ammiOil (TAM) by Disc Diffusion Method. Antibacterial activity was expressed as diameter of bacterial growth inhibition zone in the presence of films with different TAM concentrations. Mean values with different letters within a column were significantly different by Duncan’s multiple range tests at (P < 0.05).
Antibacterial Activity of Gelatin Films Incorporated WithTrachyspermum ammiOil (TAM) by Viable Colony Count. Antibacterial activity was expressed as bacterial growth reduction in the presence of films with different Ferula oil concentrations. Mean values with different letters within a column were significantly different by Duncan’s multiple range tests at (P < 0.05).
5. Discussion
5.1. Mechanical Properties of the Films
Gelatin films were mainly stabilized by the weak bond including hydrogen bond and hydrophobic interaction (7). However, TAM incorporation especially at higher concentrations caused a significant decrease in tensile of the films. Addition of TAM possibly resulted in lowering the interaction between gelatin monomers, and hindering the polymer chain-to-chain interactions, which consequently caused a decrease in tensile with the simultaneous increase in elongation at break of the films (22). Gelatin films incorporated with citrus oil showed lower tensile strength but higher elongation at break than the control films free of incorporated essential oil, similar to the results of the current study (18). These results suggested that gelatin films incorporated with TAM could be promising candidates for physically powerful food packaging.
5.2. Solubility Determination of Films
Gelatin is water-soluble, can easily dissolve partially when coming into contact with an aqueous medium, and may lose fibrous structure to high ambient humidity especially for long periods of time. However, cross-linking can stabilize gelatin structure and decrease its solubility in aqueous mediums (7). Generally, the effects of the additives on the solubility of films depend on the type of compounds and their hyrophilicity and hydrophobicity indexs (23). TAM is a hydrophobic material that favorably interacts with hydrophobic domain of gelatin and may hinder gelatin network formation and consequently cause an increase in the solubility of the films (24). Gelatin-chitosan films in the presence of essential oil, showed a significant increase in the film solubility, similar to the results of the current study (25). These results recommended that gelatin films incorporated with TAM could be promising candidates for water resistant food packaging.
5.3. Swelling Capacity of Films
Gelatin is a hydrophilic material expected to absorb molecules of water. The porous gelatin films showed higher swelling capacity because of the porosity in their network structures that allows more water to enter inside the film (26). Incorporation of TAM could reduce swelling capacity of the films which might be related to hydrophobic nature of TAM. Hydrophobic domains of gelatin can essentially interact with TAM through hydrophobic interaction and thereby enhance interfacial interaction between matrix (gelatin) and filler (TAM) (24, 27). This event saturated gelatin network with TAM and water molecules could not diffuse to gelatin network, thereby swelling decreased. These results recommended that gelatin films incorporated with TAM could be promising candidates for water resistant food packaging.
5.4. Water Vapor Permeability of Films
Gelatin is a hydrophilic material that strongly interacts with water molecules and causes a reduction in the water vapor transmission through gelatin films (26, 28, 29). Incorporation of the additive to gelatin films causes a significant change in water vapor transmission through films, while the final WVP capacity is related to hydrophobicity/hyrophilicity index of all compounds in the films. Hydrophobic domains of gelatin can essentially interact with TAM through hydrophobic interaction and thereby enhance interfacial interaction between matrix and TAM. This phenomenon hinders interactions between gelatin chains and water molecules thus water molecules freely pass through the films and consequently cause the increase in WVP (24, 27). The WVP of chitosan films incorporated with thyme oil slightly increased, similar to the results of the current study (21). These results recommended that gelatin films incorporated with TAM could be promising candidates for water barrier food packaging.
5.5. Antioxidant Activity of Films
The gelatin films without TAM showed very low activity against the ABTS decolorization. Various studies have examined antioxidant properties of peptides derived from gelatin in different sources. These studies have shown that peptides derived from enzymatic hydrolysis of gelatin are lipid peroxidation inhibitors, free radical scavengers, and transitional metal ion chelators. The antioxidative properties of peptides are related to their amino acid composition, molecular weight, structure, and hydrophobicity (2, 30). The gelatin films containing different TAM concentrations decolorize ABTS dose-dependently. Fish skin gelatin films incorporated with citrus essential oils (18) and chitosan films incorporated with thyme oil (21)exhibited strong antioxidant activity, similar to the results of the current study. These antioxidant activities may be attributed, at least in part, to the presence of phenols, flavonoids, sesquiterpenes and sulfur-containing compounds in the TAM (31, 32). These results recommended that gelatin films incorporated with TAM could be promising candidates for safe radical scavenger food packaging. Such antioxidant activities could diminish oxidative damage in foodstuff.
5.6. Antibacterial Activity of Gelatin Films
The antibacterial activities recognized in the essential oils from several medicinal plants indicated that the essential oils are related to the attack on the phospholipids present in the cell membranes, which cause increased permeability and leakage of cytoplasm, or in their interaction with enzymes located on the cell wall. Thus, the resistance of Gram-negative bacteria to the essential oils likely lies in the protective role of their cell wall lipopolysaccharide or outer membranes proteins, which restrict diffusion of hydrophobic compounds through the lipopolysaccharide layer (33). Essential oils have the ability to disrupt lipid structure of the bacteria cell walls, leading to destruction of cell membrane, cytoplasmic leakage, cell lysis and ultimately cell death. The decrease in pH that occurs due to cell membrane disruption resulted in a loss of control of cellular processes such as ATP biosynthesis, DNA transcription and protein synthesis (34). Essential oils also penetrate into mitochondrial membrane, leading to the greater permeability of organelle and the potassium ion leakage process. The leakage of ions, especially potassium, out of a cell is a clear indication of membrane damage and cell death (35). TAM gradually released from films to the solution and penetrated into the cell membranes and disrupted the membrane structure and finally caused cell death. These results recommended that gelatin films incorporated with TAM could be promising candidates for safe antimicrobial food packaging. Such antibacterial activity could diminish growth of pathogenic bacteria in the foodstuff.