1. Background
Pediatric patients with congenital heart disease (CHD), especially those undergoing cardiac surgery, are at a higher risk of thromboembolism compared to other pediatric populations. These patients have a history of surgical palliation with a foreign shunt material (e.g., aortopulmonary shunt), cavopulmonary anastomosis, Fontan procedure, or implantation of intracardiac devices and stents (1, 2). Aspirin, as the most commonly used medication around the world, is used to prevent thromboembolism in pediatric patients with CHD, without conclusive evidence supporting its antiplatelet efficacy (3, 4).
Aspirin is an irreversible and non-selective cyclooxygenase (COX) inhibitor, which hinders the production of thromboxane A2 (TXA2) in platelets by acetylating a serine residue at position 529 of the COX-1 isoform (5). Almost 5 to 60% of adult patients on aspirin therapy for secondary prevention do not respond properly to aspirin; this phenomenon is known as aspirin resistance (6). However, the incidence of aspirin resistance in the pediatric population is not precisely defined. According to different laboratory studies, the incidence of aspirin resistance ranges from 2.3 to 80% in children (7, 8). The term “laboratory aspirin resistance” should be used when aspirin fails to meet its pharmacological target, as documented by specific laboratory tests. Clinical aspirin resistance occurs when aspirin cannot prevent thromboembolic events. However, different tests have indicated a poor correlation for measuring laboratory and clinical aspirin resistance.
There is no commercially available gold standard for assessing the incidence of aspirin resistance (9-11).
2. Objectives
In this study, we aimed to determine the frequency of aspirin resistance using the AggreGuide A-100 adenosine diphosphate (ADP) Assay in pediatric patients with CHD and to evaluate its correlation with clinical and laboratory parameters.
3. Methods
3.1. Study Design
This cross-sectional, prospective study was conducted on 103 children with CHD who routinely received aspirin therapy for a minimum of eight days at our hospital. The study population was recruited from consecutive patients presenting to the hospital between April 2019 and September 2019. The routine aspirin dose was 3 - 5 mg/kg, and a maximum amount of 100 mg was prescribed daily for the antiplatelet effects in our department. Also, all patients with cyanotic CHD and single-ventricle physiology had received aspirin therapy. The patients who had undergone surgery to correct intracardiac defects with synthetic patches, transcatheter devices, or stents received aspirin therapy for six months following the procedure. On the other hand, patients who had undergone cardiac surgery, or received a blood transfusion within the past month, or used anti-inflammatory drugs concomitantly over the past ten days before the test were excluded from the study.
This study was approved by the Ethics Committee of Çukurova University, Adana, Turkey. All families provided a full informed consent form to participate in this study. The medical records of the patients were reviewed for cardiac diseases and demographic information. The parents were interviewed regarding the patient’s medication use, history of clotting or bleeding disorders, thromboembolic events, and compliance with aspirin therapy. Considering the poor correlation of laboratory and clinical aspirin resistance, we did not change the antiplatelet regimen of the patients based on only the laboratory tests.
3.2. Laboratory Measures
A laboratory analysis was performed, including complete blood count and albumin measurements in the serum. The prothrombin time, partial thromboplastin time, serum fibrinogen levels, and international normalized ratio (INR) were also analyzed for a coagulation screen. The platelet aggregation was measured by the AggreGuide A-100 ADP Assay (Aggredyne Inc., Houston, Texas, USA). The purpose was to measure the platelet aggregation when the patient’s blood contained adenosine diphosphate (ADP), which is known to activate platelets by binding to ADP receptors on the platelet surface, resulting in platelet aggregation. It is known that P2Y12 inhibitors prevent ADP-induced platelet aggregation by blocking the P2Y12 ADP receptors on the platelet surface.
Generally, the AggreGuide ADP Assay cartridges are individually sealed in Mylar® pouches. Each cartridge contains a lyophilized mixture of adenosine diphosphate (10 μM) and excipients to facilitate freeze-drying. The blood samples were assayed in vacuum collection tubes containing sodium citrate (3.2%). They were gently inverted to achieve complete mixing of the contents and tested within 30 minutes to four hours of drawing blood. The results are reported as the platelet activity index (PAI). The PAI defines the degree of ADP-induced platelet aggregation in the whole blood, where PAI < 4.7 suggests a platelet disorder. In other words, there is aspirin resistance when PAI is > 4.7. A peripheral smear examination of the blood samples was performed for platelet aggregation to compare the results with the platelet aggregation test results.
3.3. Statistical Analysis
Continuous variables are presented as mean, median, and range and categorical variables are presented as frequency and percentage. Chi-square or Fisher’s exact test was used for comparing the categorical variables. If the inter-group comparison of numerical variables confirmed our hypothesis, a t-test was performed, while if our hypothesis was rejected, the Mann-Whitney U test was used to compare independent groups. Moreover, for a logistic regression analysis, the backward Wald method was used to determine independent risk factors for aspirin resistance. A multivariate regression analysis was also carried out by adding variables that were significant in the univariate analysis to the model. The results were considered significant at P < 0.05. SPSS for Windows version 23.0 was used to perform statistical analyses.
4. Results
The study population consisted of 103 subjects, including 62 (60.2%) male and 41 (38.8%) female patients. The age range of the subjects was between one month and 17 years (mean: 56 ± 51 months). The average duration of aspirin use was 30 ± 478 days (median: 30 days; minimum: 8 days; maximum: 4015 days). The two target populations included: (1) patients with cyanotic CHD, who underwent biventricular repair (n = 24) and single-ventricle palliation, including the Fontan procedure (n = 4), bidirectional cavopulmonary anastomosis (n = 6), and aorticopulmonary arterial shunts (n = 19); and (2) patients with non-cyanotic CHD following the surgical repair of defects with synthetic patches (n = 30), stent placement for coarctation of the aorta (n = 2), or transcatheter device closure of the atrial septal defect (n = 18). The patients’ characteristics, stratified by the type of heart disease, are shown in Table 1.
| Variables | Cyanotic CHD (n = 53) | Non-cyanotic CHD (n = 50) | Total (n = 103) | P-Value |
|---|---|---|---|---|
| Gender | 36 M/17 F | 26 M/24 F | 62 M/41 F | 0.111 |
| Age (mon) a | 50 ± 51 (1 - 204) | 63 ± 50 (1 - 96) | 56 ± 51 (1 - 204) | 0.216 |
| Weight (kg) a | 13.5 ± 10.9 (3 - 60) | 18.4 ± 13.5 (2.7 - 76) | 15.8 ± 12.4 (2.7 - 76) | 0.027 |
| Pulse oximetry saturation (%) a | 87.2 ± 8.2 (67 - 96) | 98.2 ± 4.7 (96 - 100) | 92.5 ± 8.7 (67 - 100) | 0.0001 |
| Aspirin use duration (days) a | 60 ± 357 (8 - 1460) | 28 ± 579 (8 - 4015) | 30 ± 478 (8 - 4015) | 0.022 |
a Values are presented as mean ± SD (range).
There were no significant differences in terms of gender and age between the cyanotic and non-cyanotic groups. The cyanotic CHD patients showed a significantly lower pulse oximetry saturation compared to the non-cyanotic group (P = 0.0001). The average duration of aspirin use was longer in the cyanotic CHD group than the non-cyanotic CHD group (P = 0.022). Based on the results, the prevalence of aspirin resistance (PAI > 4.7) was 36.9% in children with CHD. Although aspirin resistance in cyanotic CHD patients (41.5%) was higher than in non-cyanotic patients (32%), the difference was not statistically significant (P = 0.414).
No significant difference was found between aspirin-responsive and aspirin-resistant patients in terms of sex, age, duration of aspirin use, and concomitant medication use (Table 2). The thromboembolism history was more significant in aspirin-resistant patients as compared to responsive patients, and the difference was statistically significant (P = 0.003). Although the pulse oximetry saturation was lower in aspirin-resistant patients, the difference was not statistically significant; nevertheless, the difference was very close to the significance level (P = 0.055). No significant relationship was found between aspirin resistance and use of captopril (P = 0.540), furosemide (P = 0.835), gastric-protective drugs (P = 0.519), and enteric-coated aspirin (P = 0.060); the association with the use of enteric-coated aspirin was very close to the significance level. A significant difference was found in the serum albumin (P = 0.032) and fibrinogen (P = 0.0001) levels between the aspirin-responsive and aspirin-resistant groups (Table 3).
| Variables | Aspirin-Responsive Group (n = 65) | Aspirin-Resistant Group (n = 38) | P-Value |
|---|---|---|---|
| Gender | 41 M/24 F | 21 M/17 F | 0.532 |
| Age (mon) a | 57 ± 55 (2 - 204) | 29 ± 40.9 (1 - 187) | 0.114 |
| Pulse oximetry saturation (%) a | 93.8 ± 7.4 (75 - 100) | 90.4 ± 10.2 (67 - 100) | 0.055 |
| Aspirin usage time (days) a | 30 ± 561 (8 - 4015) | 30 ± 278 (8 - 1095) | 0.511 |
| Thromboembolism history | 6 (+)/59 (-) | 13 (+)/25 (-) | 0.003 |
a Values are presented as mean ± SD (range).
| Variables | Aspirin-Responsive Group (n = 65) | Aspirin-Resistant Group (n = 38) | P-Value |
|---|---|---|---|
| Hemoglobin (g/dL) a | 11.7 ± 1.6 (8.1 - 15.4) | 11.8 ± 2.0 (7.7 - 17.1) | 0.802 |
| Hematocrit (%) a | 34.9 ± 5.2 (25 - 52) | 35.5 ± 6.9 (21 - 54) | 0.606 |
| Platelets (x1000/mm3) a | 276 ± 141.7 (114 - 855) | 282 ± 161.2 (97 - 711) | 0.689 |
| Albumin (g/L) a | 34.0 ± 3.9 (26 - 43) | 32.3 ± 3.9 (22 - 41) | 0.032 |
| Prothrombin time (s) a | 13.9 ± 3.1 (11 - 34) | 14.6 ± 3.4 (11 - 31) | 0.338 |
| Activated partial thromboplastin time (s) | 30.2 (15.4 - 70.0) | 32.3 (20.5 - 53.0) | 0.288 |
| INR a | 1.13 ± 0.3 (0.87 - 3.1) | 1.22 ± 0.4 (0.87 - 2.99) | 0.183 |
| Fibrinogen (mg/dL) a | 239 ± 109 (91 - 603) | 400 ± 113 (132 - 639) | 0.0001 |
Abbreviations: BUN, blood urea nitrogen; INR, international normalized ratio.
a Values are presented as mean ± SD (range).
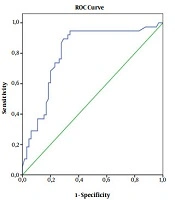
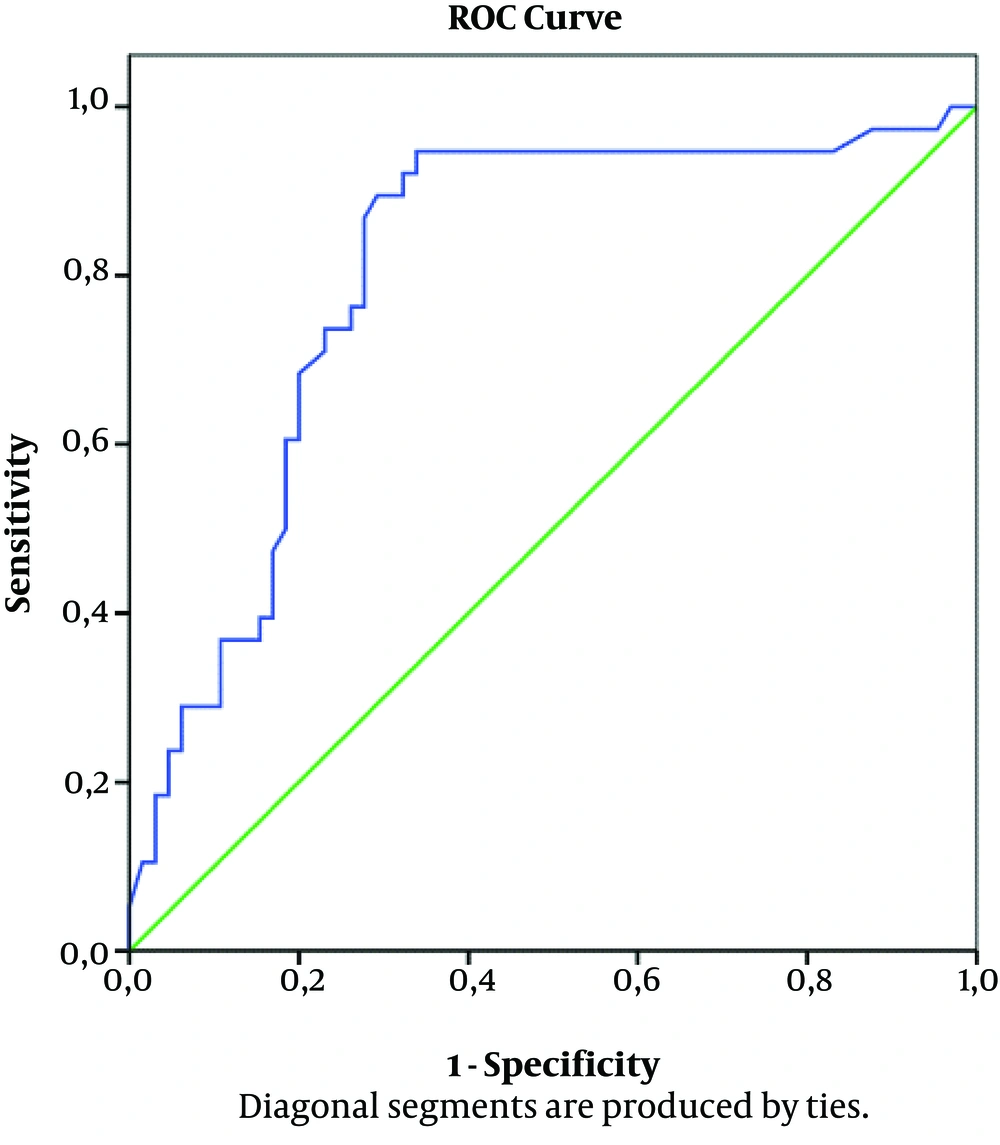
The area under the receiver operating characteristic (ROC) curve was 0.81 (95% CI: 0.72 - 0.89; P = 0.0001) in the ROC analysis. A cut-off value of 287.5 mg/dL for the fibrinogen level predicted aspirin resistance with 86.8% sensitivity and 72.3% specificity (Figure 1). The mean albumin level in the group with aspirin resistance was found to be significantly lower than the aspirin-responsive group. Although a cut-off value was desired for albumin, the ROC analysis did not yield a statistically significant result (P = 0.058).
In the multivariate regression analysis, only the fibrinogen level (OR = 1.01, P = 0.0001) and thromboembolism history (OR = 3.78, P = 0.035) were found to be independent indicators of aspirin resistance (Table 4). Based on the comparison of platelet aggregation in the peripheral blood smears and aspirin resistance, while the count of platelet clusters in 36 patients with aspirin resistance was twice as high as aspirin-responsive patients, none of the subjects in the aspirin-responsive group showed platelet aggregation, and the difference was statistically significant (P = 0.0001). Of all patients with a thromboembolism history, 13 were aspirin-resistant, and six were aspirin-responsive; the difference between the groups was statistically significant (P = 0.003).
| Variables | B | SE | Wald | df | P | Odds Ratio | 95% CI for Odds Ratio | |
|---|---|---|---|---|---|---|---|---|
| Lower | Upper | |||||||
| Fibrinogen | 0.009 | 0.002 | 17.5 | 1 | 0.0001 | 1.01 | 1.01 | 1.02 |
| Thromboembolism history | 1.331 | 0.632 | 4.4 | 1 | 0.035 | 3.78 | 1.09 | 13.07 |
| Constant value | -7.030 | 1.843 | 14.5 | 1 | 0.000 | 0.001 | ||
5. Discussion
Children with cyanotic CHD, who have undergone surgical palliation, are at a significant risk of thromboembolic complications, and aspirin has been used to prevent these complications in these patients (12). The majority of studies on aspirin resistance have been conducted on cyanotic CHD patients (3, 8, 12). In this regard, Berganza et al. found the prevalence of aspirin resistance to be 72% in CHD patients. However, only two out of 25 patients had coronary anomalies, and all other patients had cyanotic CHD (3). In a study by Mir et al., aspirin resistance was found in 80% of patients with single-ventricle physiology who required single-ventricle palliation in the immediate postoperative period (8).
Moreover, Heistein found that patients with cyanotic heart disease were more likely to be aspirin-resistant (39.5%) compared to those with non-cyanotic heart disease or no structural heart disease (17.5 vs. 20%; P = 0.04). Although cyanotic CHD patients were significantly younger and had a higher blood cell index than non-cyanotic patients, no significant association was found between these parameters and aspirin resistance (4). In the present study, the prevalence of aspirin resistance in patients with CHD was 36.9%, which is similar to the results of various adult cohorts (9.5 - 51%) (13-15). The prevalence of aspirin resistance in children with cyanotic CHD was found to be higher than non-cyanotic children, similar to the study by Heistein et al.; however, the difference was not significant. In the current study, we could not find a parameter that would affect aspirin resistance in the cyanotic and non-cyanotic groups. However, the relationship between oxygen saturation and aspirin resistance was almost statistically significant, which might be due to the small number of cases in our study. Nevertheless, no study was found to compare saturation and aspirin resistance.
In this study, no significant correlation was observed between aspirin resistance and the patient’s sex, age, duration of aspirin use, and concomitant medication use. In another study by Heinstein et al. on children with CHD, the mean age of patients was 3.5 and 7.5 years in the aspirin-resistant and aspirin-responsive groups, respectively, and there was no significant difference between the two groups (4). In the study by Berganza et al., the mean age of the aspirin-resistant group (10.7 years) was higher than the aspirin-responsive group (8.1 years), although the P-value was insignificant at 0.054 (3). In studies conducted on adults, the prevalence of coronary artery disease increased with increased platelet activation due to advancing age. In this regard, Pamukcu et al. found that the prevalence of aspirin resistance was higher in older patients, although there was no significant difference in terms of gender (16). So far, many studies on adults have found no significant relationship between aspirin resistance and the patient’s age or gender (17, 18).
The inhibition of TXA2 biosynthesis and platelet aggregation seem to occur consistently in healthy individuals receiving short courses of aspirin therapy (19, 20). In adult studies, a progressive increase was found in the platelet aggregation and thromboembolic complications with long-term aspirin therapy in patients who were previously shown to be sensitive to aspirin (18, 21). Although the mechanism of prolonged administration is unknown, it can be explained by the progression of atherosclerosis or the progressive decrease in compliance over time. However, there is no information indicating the relationship between the duration of aspirin use and aspirin resistance in the pediatric population. In our study, no significant relationship was found between these variables, which could be explained by the relatively short aspirin use.
Upper gastrointestinal bleeding as a result of the reduced production of protective gastric prostanoids and impaired platelet aggregation is a major side effect of aspirin therapy. To overcome this adverse side effect, enteric-coated aspirin was developed. It has been suggested that the enteric coating reduces the antithrombotic effects of aspirin (22, 23). No significant difference was found between the groups using enteric-coated and non-coated aspirin in terms of aspirin resistance in our study; nonetheless, the difference was very close to the significance level, which might be related to the small number of cases. Most of our patients were required to use less than 100 mg of aspirin per day, because their body weight was less than 30 kg. Therefore, the aspirin dosage was adjusted by diluting the tablet into a liquid; however, a true comparison is not possible, as the enteric coating breaks down.
Drug interactions are important causes of aspirin resistance. Angiotensin-converting enzyme (ACE) inhibitors, such as captopril and diuretics (e.g., furosemide), are the most common drugs used against pediatric cardiac diseases. Another product of the COX pathway is TXA2, a potent prostaglandin with prothrombotic and vasoconstrictive effects (24). Lin et al. demonstrated that platelet TXA2 mediates some of the vasoconstrictive effects of angiotensin II; therefore, ACE inhibitors may act as vasodilators by attenuating the production of this prostaglandin. Similarly, salicylates inhibit the COX enzyme and TXA2 production (25). Platelet aggregation was diminished during therapy, which was paralleled by a reduction in the platelet TXA2 generation; therefore, captopril could have supportive effects for aspirin through its antiplatelet function.
Expectedly, aspirin resistance was not high in patients using captopril in our study. In another study, the H2 receptor blockers reduced the gastrointestinal damage among adult patients using aspirin for two weeks or longer (26). A higher percentage of aspirin resistance was observed among adult patients using proton-pump inhibitors (27). About a third of our patients (33.9%) used gastric-protective drugs, and all of them were using H2 blockers instead of proton-pump inhibitors. The prevalence of aspirin resistance was not found to be high in these patients.
Moreover, fibrinogen is an acute phase reactant, which increases during inflammatory responses. The plasma fibrinogen plays a role in thrombocyte aggregation, which is the final step in clot formation along with thrombin and contributes to the vascular injury response. During fibrin formation, fibrinogen promotes blood clotting by activating and forming bridges between blood platelets through binding to their glycoprotein IIb-IIIa complex (GpIIb/IIIa) surface membrane fibrinogen receptors (28). The relationship between the increase in fibrinogen levels and aspirin resistance was first shown in a study by Feher et al. (29). They reported that an increase in the plasma fibrinogen levels increased the erythrocyte adhesion and ADP, thereby increasing the platelet aggregation; this mechanism is responsible for aspirin resistance. In recent studies, the relationship between prothrombotic variables and aspirin resistance has been examined. The results of these studies revealed that the fibrinogen level and platelet count were high in patients with aspirin resistance (17, 30). In the present study, when the cut-off value was 287.5 for the fibrinogen level, the sensitivity was 86.8%, and the specificity was 72.3% (Figure 1).
Human serum albumin (HSA) plays an important role in drug transport and metabolism and strongly affects the drug distribution in the plasma. The binding strength of drugs to plasma proteins, especially to HSA, is an important factor in drug development. However, the biological consequences of albumin acetylation are not fully understood in vivo; this phenomenon may be of significance in aspirin resistance (31). While the serum albumin levels were lower in aspirin-resistant patients, we did not find any significant results in the ROC analysis. It seems that the effects of serum albumin level on aspirin resistance need to be determined in large prospective studies.
In the present study, only fibrinogen and a history of thromboembolism in patients were independent risk factors for aspirin resistance. Almost 50% of infants and 30% of older children with a venous thromboembolic disease had an underlying cardiac disease (32). Nevertheless, there are few reports correlating aspirin resistance with thrombotic events in children. In this regard, Emani et al. documented the rate of thrombosis to be 7.4% in the first 30 days after complex cardiac surgical procedures. In their study, thrombosis occurred more commonly in aspirin-resistant patients, compared to those who responded to aspirin (60 vs. 1.2%) (1). The rate of thromboembolism was 25% in aspirin-resistant patients and 10.2% in aspirin-responsive patients. Based on the conflicting evidence regarding the relationship between aspirin resistance and thrombosis, further larger-scale studies are necessary to confirm the results of this study.
There are no findings linking aspirin resistance to the clinical consequences of children. In this study, we established a correlation between aspirin resistance, platelet aggregation based on the peripheral blood smear, and clinical thrombosis. Six patients with a thrombus were classified as aspirin-responsive, and 13 were classified as aspirin-resistant. Platelet aggregation was not detected in 65 aspirin-responsive patients, while it was not observed in only two out of 38 patients with aspirin resistance in the peripheral blood smear. We believe that the assessment of platelet aggregation based on peripheral blood smears can be suitable for distinguishing clinical aspirin resistance from laboratory aspirin resistance.
5.1. Conclusion
Although this study was conducted on a small number of patients with a short duration of aspirin use, our findings suggest that measurement of fibrinogen levels and evaluation of platelet aggregation in the blood smear may be the first step in predicting aspirin resistance. However, further studies with a larger sample size are still needed to establish the definition of aspirin resistance and effective treatment and to evaluate the relationship between laboratory aspirin resistance and clinical outcomes.