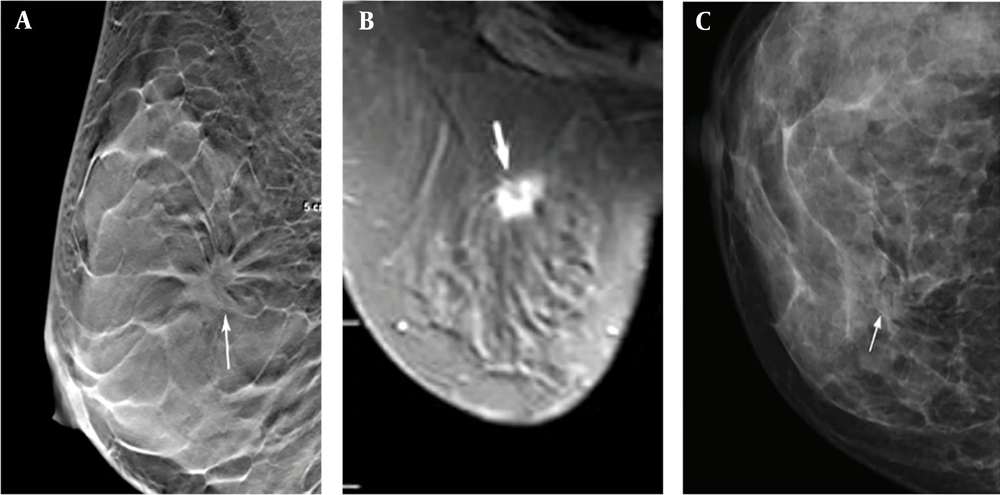
FFDM is currently recognized as an optimal imaging technology to detect breast tumors. However, the visibility of lesions decreases due to overlap with glandular tissues, leading to the reduction of diagnostic sensitivity and specificity (
9). Traditional mammography provides 2D tomographic images that may not detect signs of high diagnostic value, such as burrs or lobes, and may yield false images of malignant lesions, inevitably increasing the false negative and false positive rates (
10).
DBT, as a novel technology that combines traditional tomography with digital image processing, can represent the mammary glands from different angles. The low-dose projection data are reconstructed in a computer post-processing software to acquire mammary gland images at any depth parallel to the detector level (
11). Compared to traditional FFDM, DBT avoids the overlap of normal glandular tissues with the lesion in imaging, especially for dense mammary glands, improves the visibility of lesion edges (lobes or burrs), and clearly displays lesions with different shapes, heights, and positions hidden in the fibrous gland; therefore, it increases the detection rate of cancer lesions, as well as the sensitivity and accuracy of diagnosis, while reducing the false positive rate (
12).
DBT can clearly display the lesion edges (burrs), suggesting its higher diagnostic accuracy and detection rate for early breast cancer, especially dense breast tissues (
13), as confirmed in the present study. DBT combined with FFDM seems to have a higher diagnostic accuracy for breast cancer compared to FFDM alone (
14). In the present study, the sensitivity, specificity, and accuracy of FFDM combined with DBT for the diagnosis of breast cancer were 92.38, 90.48, and 90.95%, respectively, which were higher than those reported for FFDM or DBT alone. Although DBT performs better in the detection of lesions and has a higher diagnostic accuracy for breast cancer compared to FFDM, its application is still limited due to increased radiation dose and image readings.
MRI can provide high-resolution images of soft tissues, as well as multi-parameter, multi-sequence, and multi-orientation images. Dynamic contrast-enhanced MRI is recognized as the most adequate imaging technology for the examination of breast tissue (
15). However, the results of the present study showed 100% sensitivity and only 80.95% specificity for MRI, probably because of the overlap in the MRI signs of benign and malignant breast lesions or insensitivity of MRI to calcification. Traditional mammography combined with MRI shows a higher diagnostic accuracy for breast cancer, which benefits early diagnosis (
16). In the present study, the sensitivity, specificity, and accuracy of FFDM combined with MRI for breast cancer diagnosis were 100.0, 86.67, and 95.24%, respectively, which were higher than those of FFDM or MRI alone. Although MRI seems to have a higher diagnostic efficacy for breast cancer, its application is still limited due to contraindications, long duration, and high cost.
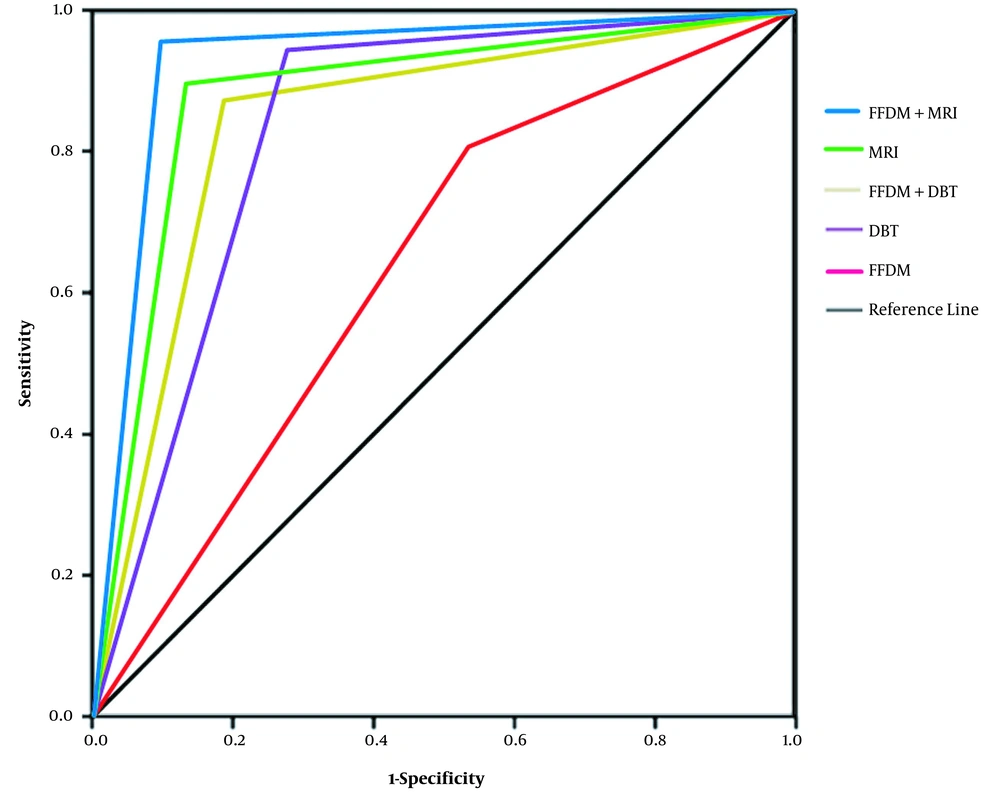
Since BI-RADS category 4 is subdivided into 4A, 4B, and 4C subcategories in the 2013 BI-RADS edition, and only 2 - 10% of 4A lesions are likely to be malignant, in the current study, most lesions were considered to be benign. In this study, BI-RADS-MRI ≥ 4B was defined as malignant lesions to increase pathological relevance. Among five imaging methods, FFDM + MRI had the highest diagnostic efficiency. FFDM + DBT and FFDM + MRI had a markedly higher diagnostic accuracy for breast cancer compared to FFDM.
This study had some limitations. First, the sample size was small (n = 210); therefore, the results need to be confirmed in a larger population. Second, DBT is a novel technology in China, which has not been widely applied; therefore, its advantages remain to be fully validated.
In conclusion, DBT and FFDM + DBT could significantly improve breast cancer diagnosis compared to FFDM; the former methods showed comparable diagnostic efficiencies to MRI and FFDM + MRI. The sensitivity of DBT was lower than that of MRI and higher than that of FFDM, while its specificity and positive predictive value were higher than those of MRI. All of the examination methods had certain diagnostic values, with the highest diagnostic efficiency found in FFDM + MRI. Besides, FFDM + DBT and FFDM + MRI could improve the diagnostic accuracy of breast cancer, which is conducive to early diagnosis.