1. Introduction
Cytomegalovirus (CMV) belongs to a group of herpes viruses, with a high seroprevalence of 58.9% in the general population (1). In immunocompetent individuals, a primary CMV infection causes no symptoms, except for a self-limited febrile disease. Following a primary infection, CMV establishes a lifelong virus latency in various cells and is reactivated in susceptible individuals (2). On the other hand, CMV infection can cause various symptoms in immunocompromised patients and solid organ transplant recipients (2). The reported symptoms and clinical features of CMV infection in solid organ transplant recipients include fever, malaise, neutropenia, thrombocytopenia, and atypical lymphocytosis.
A CMV infection can even cause a tissue-invasive disease, involving the liver, kidneys, pancreas, gastrointestinal tract, lungs, and the central nervous system (3). However, cutaneous or subcutaneous CMV infections have been rarely reported in the literature, even in immunocompromised patients, including acquired immunodeficiency syndrome (AIDS) patients, leukemia patients, and organ transplant recipients. Only sporadic cases have been reported, while there are no large-scale studies (4). According to previous case reports, most CMV-related skin lesions are ulcers or maculopapular rashes, mainly localized on the perianal, genital, and perigenital areas (4, 5).
Here, we present a case of an unusual subcutaneous CMV infection and examine the correlation of imaging and pathological findings. To the best of our knowledge, the radiological findings of this infection have not been reported yet.
2. Case Presentation
The patient was a 45-year-old male patient with a one-month history of left axillary swelling and pain. He had received kidney transplantation four years ago for end-stage renal disease due to diabetic nephropathy. His medication history included tacrolimus and mycophenolic acid for immunosuppression. Other medications used by the patient included methylprednisolone, pravastatin sodium, triamcinolone, losartan, aspirin, kalimate, carvedilol, insulin glargine, and insulin aspart.
For further analysis, a contrast-enhanced chest computed tomography (CT) scan was carried out. The CT scan showed multiple, variable-sized nodular lesions in the subcutaneous fat layer of the left axilla, right posterior shoulder, and right posterior chest wall (Figure 1A and B). The nodules showed homogenous iso-attenuation to skeletal muscle, ranging from 40 to 50 Hounsfield units (HU). The nodules did not show calcification or discernible enhancement (Figure 1C and D). On CT scan, the nodules were suspected of a post-transplant lymphoproliferative disorder (PTLD), with or without necrotic changes. However, clinically, the lesions were suspected to be subcutaneous abscesses.
A 45-year-old male kidney transplant recipient, with a one-month history of left axillary swelling and pain, presented with nodular lesions on CT scan. Pre-contrast and contrast-enhanced axial CT images of subcutaneous tissue show an invasive cytomegalovirus (CMV) infection in the left axilla, right posterior shoulder, and right posterior chest wall areas. A and B, Pre-contrast axial CT images (C7 and T2 levels) show multiple, variable-sized nodular lesions in the subcutaneous fat layer of the left axilla, right posterior shoulder, and right posterior chest wall areas (arrows). The nodules show homogenous iso-attenuation to skeletal muscle, ranging from 40 to 50 Hounsfield units (HU); C and D, Contrast-enhanced axial CT images reveal that the nodules do not have any discernible enhancement (arrows).
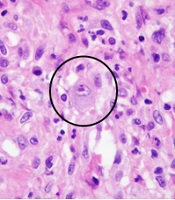
A surgical incisional biopsy was performed for diagnosis. Microscopic examination indicated multiple inflammatory cells, including CMV-infected cells (Figure 2A), shown by CMV immunostaining (Figure 2B). Based on the pathological findings, the nodules were compatible with a tissue-invasive CMV disease. The patient was administered ganciclovir. On the 8th day after treatment, a follow-up CT scan was performed to determine his response to treatment. The CT scan showed partial regression of nodular lesions, probably responding to the treatment (Figure 3A and B).
Histopathological examination of subcutaneous nodules in the chest wall. Severe chronic active inflammation diffusely involves the subcutaneous tissue. A, Inflammatory cells include lymphocytes, plasma cells, and neutrophils (hematoxylin and eosin staining, × 400 magnification). The cytomegalovirus (CMV)-infected cells are shown as circles; B, These cells are visualized by CMV immunostaining (× 200 magnification).
3. Discussion
Various terminologies have been used to describe CMV infections in solid organ transplant recipients. A CMV infection refers to virus isolation or detection in any body fluid or tissue specimen, regardless of the symptoms or signs; it is diagnosed when there is evidence of infection with attributable symptoms or signs (6, 7). A tissue-invasive CMV disease refers to a CMV infection with specific organ involvement (e.g., colitis, pneumonitis, hepatitis, nephritis, myocarditis, and meningoencephalitis) (7). In the present case, a tissue-invasive CMV disease with subcutaneous involvement was confirmed.
Our case developed multiple subcutaneous nodular lesions in the setting of immunosuppression. Before surgical biopsy, PTLD was considered as one of the most suspicious differential diagnoses. PTLD is the second most common type of post-transplant malignancy following non-melanoma skin cancer, with an incidence rate of up to 10% (8). More than half of PTLD cases present with extranodal masses, which commonly involve the skin, gastrointestinal tract, lungs, liver, and the central nervous system, as well as the allograft itself (9).
The radiological findings of our patient could not differentiate between a CMV disease and PTLD according to the latest available knowledge. Therefore, early biopsy is inevitable and essential for an accurate diagnosis. Besides, knowledge of radiological features and pathogenesis of PTLD and CMV disease highlights the important role of radiologists in making an accurate diagnosis and guiding biopsy. Favorably, there are multiple drug regimens available for the treatment of subcutaneous and cutaneous CMV infections, including ganciclovir, foscarnet, cidofovir, and valganciclovir (10).
In conclusion, we reported a very rare case of subcutaneous CMV infection mimicking PTLD in a solid organ transplant recipient. It was successfully diagnosed via CT scan and surgical biopsy and treated with antiviral medications.