1. Background
2. Objectives
3. Patients and Methods
3.1. Patients
3.2. Imaging Analyses
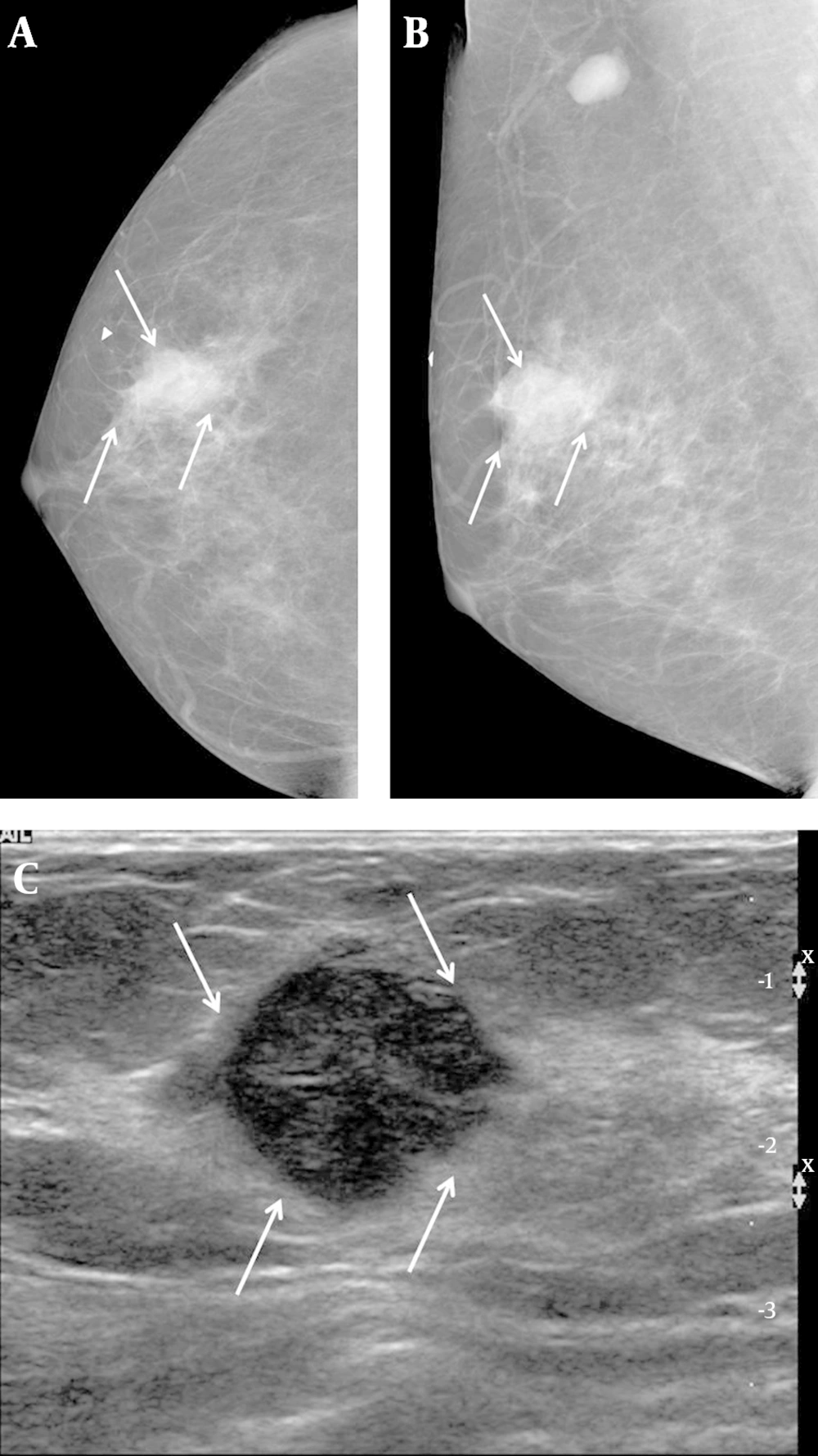
A 57-year-old woman complained of a palpable mass in her right breast. A, Right craniocaudal and B, Mediolateral oblique views on mammography showed an indistinct margin, with a round mass and without calcifications on the BB marker site in the right upper outer area (arrows). An enlarged lymph node is also seen in the right axillary area. C, Transverse ultrasound revealed a hypoechoic mass that was microlobulated with a round shape (arrows), and which we categorized as BI-RADS category 4b. US-CNB was performed, and the case was confirmed to be medullary carcinoma. The patient underwent breast-conservation surgery and was subsequently labeled as a histologic grade 3, TNBC. TNBC: Triple-negative breast cancer; US-CNB: Ultrasound-guided core needle biopsy
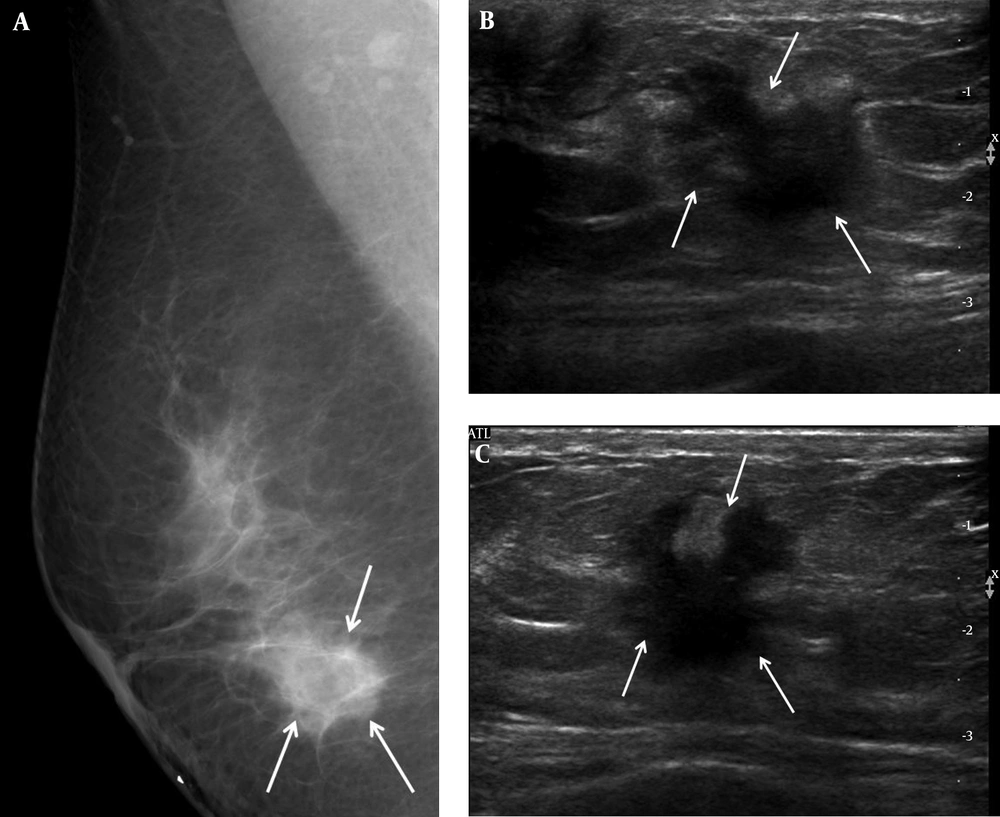
A 46-year-old woman complained of a palpable mass in her right breast. A, Right mediolateral oblique view on mammography showed an ill-defined marginated mass (arrows) on the BB marker site in the right lower area. B, Transverse and C, Longitudinal ultrasound revealed a hypoechoic mass with an irregular shape and a spiculated margin (arrows); we categorized it as BI-RADS category 5. US-CNB was performed, and confirmed invasive ductal carcinoma. The patient underwent modified radical mastectomy, after which the mass was labeled histologic grade 2, luminal A breast cancer. US-CNB: Ultrasound-guided core needle biopsy
3.3. Clinicopathological Features
3.4. Statistical Analyses
4. Results
| Variable | Luminal A (n=77) | Luminal B (n = 54) | HER2+ (n = 35) | TN (n = 34) | P Value |
|---|---|---|---|---|---|
| Patient age (mean ± SD, range) | 57.4 ± 11.3 (29 - 82) | 55.3 ± 10.3 (34 - 78) | 60.9 ± 9.0 (45 - 78) | 54.9 ± 11.3 (33 - 80) | 0.064 |
| Symptoms | 0.263 | ||||
| Yes | 49 (63.6) | 27 (50) | 24 (68.6) | 22 (64.7) | |
| No | 28 (36.4) | 27 (50) | 11 (31.4) | 12 (35.3) | |
| MMG finding | 0.037 | ||||
| Asymmetry | 11 (14.3) | 4 (7.4) | 4 (7.4) | 2 (5.9) | |
| Mass | 48 (62.3) | 34 (62.9) | 20 (57.1) | 25 (73.5) | |
| Microcalcification ± mass | 11 (14.3) | 16 (29.6) | 11 (31.4) | 4 (11.8) | |
| Negative | 7 (9.1) | 0 (0) | 0 (0) | 3 (8.8) | |
| US BI-RADS | 1.0 | 0.109 | |||
| 4a | 18 (23.4) | 11 (20.4) | 2 (5.7) | 6 (17.7) | |
| 4b | 8 (10.4) | 2 (3.7) | 7 (20.0) | 5 (14.7) | |
| > 4c | 51 (66.2) | 41 (75.9) | 26 (74.3) | 23 (67.6) | |
| Cancer type | 0.284 | ||||
| IDC | 70 (90.9) | 50 (92.6) | 34 (97.1) | 30 (88.2) | |
| Mucinous | 5 (6.5) | 3 (5.6) | 1 (2.9) | 0 (0) | |
| Medullary | 1 (1.3) | 1 (1.9) | 0 (0) | 3 (8.9) | |
| Papillary | 1 (1.3) | 0 (0) | 0 (0) | 1 (2.9) | |
| Histologic grade | < 0.001 | ||||
| 1 | 28 (37.3) | 12 (23.1) | 3 (8.6) | 1 (3.0) | |
| 2 | 38 (50.7) | 30 (57.7) | 19 (54.3) | 6 (18.2) | |
| 3 | 9 (12.0) | 10 (19.2) | 13 (37.1) | 26 (78.8) | |
| T stage | < 0.001 | ||||
| T1 | 47 (64.0) | 32 (59.3) | 10 (28.6) | 12 (35.3) | |
| T2 | 27 (35.1) | 21 (38.9) | 20 (57.1) | 22 (64.7) | |
| T3 | 3 (3.9) | 1 (1.9) | 5 (14.3) | 0 (0) |
Abbreviations: BI-RADS, breast imaging reporting and data system; IDC, invasive ductal carcinoma; IHC, immunohistochemical; MMG, mammographic; SD, standard deviation; TN, triple negative; US, ultrasound.
aValues are expressed as No. (%).
| IHC Subtype | Non-TN (n = 166) | TN (n = 34) | P Value |
|---|---|---|---|
| Patient age (mean ± SD, range) | 57.5 ± 10.7 (33 - 80) | 54.9 ± 11.3 (33 - 80) | 0.222 |
| Symptoms | 0.627 | ||
| Yes | 49 (63.6) | 22 (64.7) | |
| No | 28 (36.4) | 12 (35.3) | |
| MMG finding | 0.215 | ||
| Asymmetry | 19 (11.5) | 2 (5.9) | |
| Mass | 102 (61.5) | 25 (73.5) | |
| Microcalcification ± mass | 38 (22.8) | 4 (11.8) | |
| Negative | 7 (4.2) | 3 (8.8) | |
| US BI-RADS | 0.750 | ||
| 4a | 31 (18.7) | 6 (17.7) | |
| 4b | 17 (10.2) | 5 (14.7) | |
| > 4c | 118 (71.1) | 23 (67.6) | |
| Cancer type | 0.021 | ||
| IDC | 154 (92.8) | 30 (88.2) | |
| Mucinous | 9 (5.4) | 0 (0) | |
| Medullary | 2 (1.2) | 3 (8.9) | |
| Papillary | 1 (0.6) | 1 (2.9) | |
| Histologic grade | < 0.001 | ||
| 1 | 43 (26.5) | 1 (3.0) | |
| 2 | 87 (53.7) | 6 (18.2) | |
| 3 | 32 (19.8) | 26 (78.8) | |
| T-stage | 0.027 | ||
| T1 | 89 (53.6) | 12 (35.3) | |
| T2 | 68 (11.0) | 22 (64.7) | |
| T3 | 9 (5.4) | 0 (0) |
Abbreviations: BI-RADS, breast imaging reporting and data system; IDC, invasive ductal carcinoma; IHC, immunohistochemical; MMG, mammographic; SD, standard deviation; TN, triple negative; US, ultrasound.
aValues are expressed as No. (%).
| Variable | Luminal B (n = 54) | HER2+ (n = 35) | TN (n = 34) | |||
|---|---|---|---|---|---|---|
| OR (95% CI) | P Value | OR (95% CI) | P Value | OR (95% CI) | P Value | |
| Age | 0.981 (0.947 - 1.015) | 0.268 | 1.029 (0.987 - 1.072) | 0.178 | 0.973 (0.929 1.019) | 0.245 |
| Symptoms | ||||||
| No | 1.0 | 1.0 | 1.0 | |||
| Yes | 0.531 (0.255 - 1.106) | 0.091 | 1.099 (0.449 - 2.691) | 0.836 | 0.856 (0.315 - 2.323) | 0.761 |
| MMG finding | ||||||
| Negative | 1.0 | 1.0 | 1.0 | |||
| Asymmetry/mass | > 999.999 (< 0.001 -> 999.999) | 0.951 | > 999.999 (< 0.001 - > 999.999) | 0.962 | 0.364 (0.049 - 2.734) | 0.407 |
| Mass with calcification | > 999.999 (< 0.001 - >999.999) | 0.948 | > 999.999 (< 0.001- > 999.999) | 0.958 | 0.285 (0.027 - 2.983) | 0.295 |
| US BI-RADS | ||||||
| 4a | 1.0 | 1.0 | 1.0 | |||
| 4b | 0.427 (0.075 - 2.417) | 0.336 | 9.005 (1.414 - 57.348) | 0.020 | 2.027 (0.329 - 12.475) | 0.446 |
| > 4c | 1.284 (0.538 - 3.065) | 0.573 | 4.669 (0.970 - 22.468) | 0.055 | 1.398 (0.402 - 4.865) | 0.598 |
| Cancer type | ||||||
| IDC | 1.0 | 1.0 | 1.0 | |||
| Mucinous | 1.200 (0.189 - 7.638) | 0.847 | 3.000 (0.218 - 41.351) | 0.412 | < 0.001 (< 0.001 - > 999.999) | 0.956 |
| Medullary | > 999.999 (< 0.001 - > 999.999) | 0.970 | 0.515 (< 0.001 - > 999.999) | 0.999 | < 0.001 (< 0.001 - > 999.999) | 0.967 |
| Papillary | < 0.001 (< 0.001 - > 999.999) | 0.978 | < 0.001 (< 0.001 - > 999.999) | 0.982 | < 0.001 (< 0.001 - > 999.999) | 0.984 |
| T-stage | ||||||
| T1 | 1.0 | 1.0 | 1.0 | |||
| T2 | 1.226 (0.579 - 2.597) | 0.595 | 3.173 (1.248 - 8.066) | 0.015 | 2.376 (0.876 - 6.446) | 0.089 |
| T3 | 0.469 (0.046 - 4.783) | 0.523 | 6.552 (1.252 - 34.296) | 0.026 | < 0.001 (< 0.001 - > 999.999) | 0.989 |
Abbreviations: BI-RADS, breast imaging reporting and data system; IDC, invasive ductal carcinoma; MMG, mammographic; TN, triple negative; US, ultrasound; OR, odds ratio; HER2, human epidermal growth factor receptor 2; CI, confidence interval; IHC, immunohistochemical.
| Variable | TN (n = 34) | |
|---|---|---|
| OR (95% CI) | P Value | |
| Age | 0.970 (0.931 - 1.011) | 0.149 |
| Symptoms | ||
| No | 1.0 | |
| Yes | 1.026 (0.422 - 2.496) | 0.954 |
| MMG finding | ||
| Negative | 1.0 | |
| Asymmetry/mass | 0.173 (0.025 - 1.192) | 0.075 |
| Mass with microcalcification | 0.246 (0.045 - 1.345) | 0.106 |
| US BI-RADS | ||
| 4a | 1.0 | |
| 4b | 1.520 (0.403 - 5.723) | 0.536 |
| > 4c | 1.007 (0.377 - 2.688) | 0.989 |
| Cancer type | ||
| IDC | 1.0 | |
| Mucinous | 0.267 (0.013 - 5.479) | 0.391 |
| Medullary | 7.092 (1.149 - 43.772) | 0.035 |
| Papillary | 5.061 (0.308 - 83.153) | 0.256 |
| T-stage | ||
| T1 | 1.0 | |
| T2 | 2.155 (0.345 - 13.444) | 0.411 |
| T3 | 23.652 (4.211 - 132.830) | 0.003 |
Abbreviations: BI-RADS, breast imaging reporting and data system; IDC, invasive ductal carcinoma; MMG, mammographic; TN, triple negative; US, ultrasound; OR, odds ratio; CI, confidence interval; IHC, immunohistochemical.