1. Background
Sickle cell anemia (SCA), an inherited hematologic disorder, is a condition that affects the round shape of red blood cells. As a result of abnormally shaped erythrocytes, which look like crescents (previously called ‘sickles’), they get stuck in the blood vessels and prevent organ perfusion. It is a life-long disease that appears when abnormal genes are expressed in homozygotes. People with sickle cell trait (SCT) get the abnormal gene from only one parent and do not usually have SCA symptoms.
Information about making informed reproductive decisions is limited in literature about people with SCA and SCT (1). Affected women are associated with high rates of prenatal and natal complications, and cesarean birth rates tend to be higher in women with SCA (2-6). Because fetomaternal morbidity and mortality increases with prolonged labor, choosing the right type of delivery is important. It is necessary to test for fetal-pelvic incompatibility before making decisions on labor types. Therefore, it is important to identify women who are at risk for dystocia and to choose the most suitable means of delivery (7, 8). Pelvic roof is only one of the several factors that determine prolonged labor and the type of delivery (9). Pelvimetry and related techniques assist in the management of obstetricians’ patients (10). Radiographic and computerized tomographic pelvimetry involves high doses of radiation. The advent of newer technologies like magnetic resonance (MR) scanning has increased interest in this area. It offers an accurate and objective technique to evaluate pelvimetric measurements without using ionizing radiation (7). Dystocia due to pelvic narrowing is one indication that a cesarean section may be necessary. Therefore, looking at the existing literature, it is clear that pelvic diameters that may lead to cesarean sections have never before been studied in women with SCA or SCT.
2. Objectives
The purpose of our study was to determine whether pelvic diameters are different in women with SCA and SCT when compared with healthy individuals by studying MR pelvimetries.
3. Patients and Methods
3.1. Study Population
This was a prospective study of 66 women at Mustafa Kemal University Hospital who underwent MR pelvimetry between March 2015 and June 2015. The study protocol respected the Helsinki declaration principles, and our institutional ethics review board approved the study. Consent forms were obtained from individuals before MR scans.
Our study included 21 women with SCA, 20 women with SCT, and age and sex matched 25 healthy women as the control group. The patients were obtained from hospital records and they were invited to MR examination. The diagnosis of SCA (Hb SS) was confirmed by a combination of clinical, hematological, family, and if needed molecular studies. The controls were selected from healthy women who were admitted to our clinic for various causes but were free from hematological diseases.
Patients who were pregnant, younger than 18 years of age, or had hip replacements were excluded. Age, parity, and delivery outcome data of the individuals were recorded.
3.2. MR Imaging Examination
MR pelvimetry was performed in the supine position using a 1.5 Tesla MR imaging system (Philips, Achieva, Best, Netherlands) by the same radiologist in the daytime. Surface coils were placed over the pelvis, and patients were appropriately placed within the gantry. T1-weighted spin-echo sequences were used with the following parameters: repetition time (TR)/echo time (TE) 400/10 ms, field of view (FOV) 300 mm, 16 slices with a slice thickness of 8 mm and matrix 480×196. This enabled the midsagittal, oblique axial (angulated along the superior border of the symphysis pubis and sacral promontory), and axial sections of the pelvis to be viewed.
3.3. Image Analysis
MR images were evaluated in the digital imaging and communication in medicine viewer software-OsiriX (Pixmeo Labs, Geneva, Switzerland). Electronic calipers were used to obtain the following measurements:
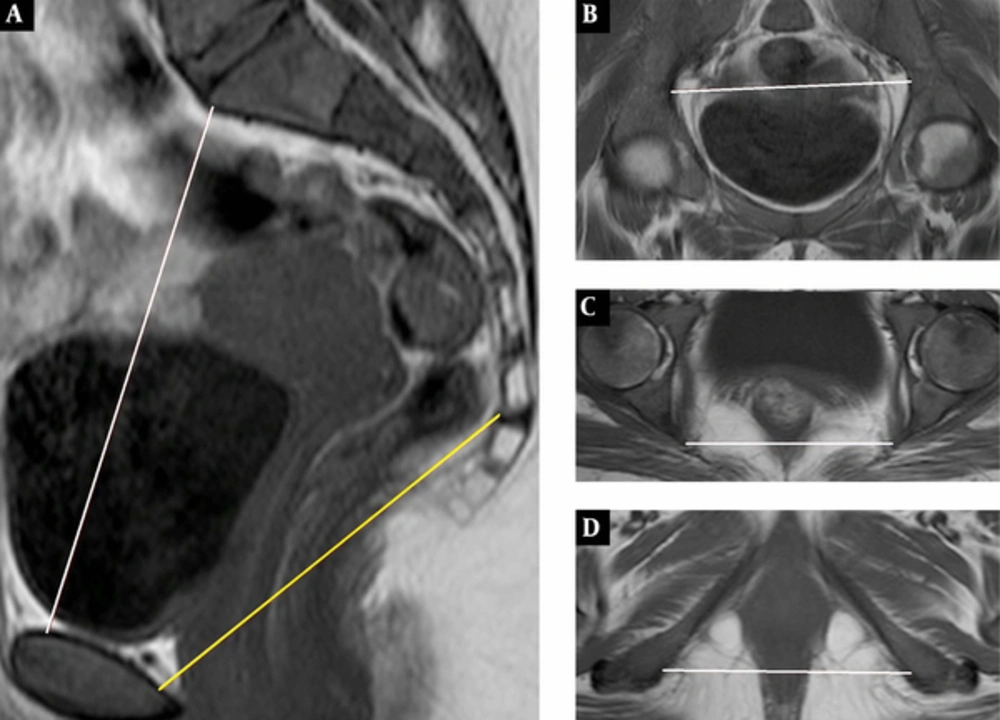
In the midsagittal section, the sagittal inlet diameter (obstetric conjugate) was measured from the sacral promontory to the top of the symphysis pubis and the sagittal mid-pelvis diameter was measured from the sacrococcygeal junction to the bottom of the inner cortex of the symphysis pubis (Figure 1A). In the oblique axial section, the transverse inlet diameter measured the largest transverse diameter of the pelvis (Figure 1B). In the axial section, the transverse mid-pelvis diameter measured the interspinous (narrowest) distance between the ischial spines (Figure 1C) and the transverse outlet diameter measured the intertuberous (widest) distance between the ischial tuberosities (Figure 1D). The mean diameters of women with SCA and SCT were compared with the diameters of women in the control group. A single experienced radiologist did radiologic evaluations.
T1-Weighted spin-echo MR pelvimetric images. A, Sagittal, midline section shows sagittal inlet (obstetric conjugate) and sagittal mid-pelvis distance. B, Oblique axial section shows transverse inlet distance. C and D, Axial sections show transverse mid-pelvis and transverse outlet distance.
3.4. Statistical Analysis
The mean of each of the five diameters ± standard deviation was calculated for all 66 women. Version 21 of the statistical package for the social sciences software (SPSS software Inc., Chicago, IL, USA) software was used for statistical analysis, and a P value of 0.05 or less was considered significant.
4. Results
The total study population consisted of 66 women divided into three groups: 21 women with SCA, 20 women with SCT, and 25 women as controls. The mean patient age was 29.0 years old (SCA: 28.57 years of age, SCT: 30.25 years of age, control: 28.36 years of age). Two women in the SCA group (1 cesarean section, 1 vaginal delivery), 13 women in the SCT group (11 cesarean sections, 2 vaginal deliveries), and 7 women in the control groups (3 cesarean sections, 4 vaginal deliveries) were parous.
Mean values and standard deviations of pelvic diameters for all patients are shown in Table 1. Sagittal inlet and transverse inlet were not significantly different between the three groups (P 0.282 and 0.619, respectively). Also mid-pelvic diameters for the transverse and sagittal mid-pelvis were similar between the groups (P 0.695 and 0.212, respectively). Even though the transverse outlet seems to be lesser in the SCA group (10.34 cm, 11.14 cm and 11.09 cm) compared with other groups, it was not statistically significant (P > 0.512).
| Pelvimetric Data | SCA (n = 21) | SCT (n = 20) | Control (n = 25) | P Value |
|---|---|---|---|---|
| Sagittal inlet (cm) | 11.69 ± 1.11 | 11.60 ± 0.81 | 12.03 ± 0.90 | 0.282 |
| Sagittal mid-pelvis (cm) | 10.83 ± 0.89 | 11.34 ± 1.05 | 11.07 ± 0.79 | 0.212 |
| Transverse inlet (cm) | 12.85 ± 0.69 | 13.06 ± 0.80 | 12.87 ± 0.77 | 0.619 |
| Transverse mid-pelvis (cm) | 10.43 ± 0.96 | 10.66 ± 0.80 | 10.61 ± 0.96 | 0.695 |
| Transverse outlet (cm) | 10.34 ± 1.06 | 11.14 ± 0.91 | 1.09 ± 1.02 | 0.512 |
SCA, sickle cell anemia; SCT, sickle cell trait; SD, standard deviation
aVlues are expressed as mean± SD
5. Discussion
In the literature, there are quite a few publications about MR pelvimetry (11-14). However, this is the first study to evaluate the stability of SCA women’s and SCT women’s pelvises by studying MR pelvimetry. Our results showed that there is no difference in pelvic diameters among SCA and SCT women with non-hemoglobinopathies.
There are high rates of prenatal and natal complications in women with SCA and SCT (6, 15). Pre-eclampsia and risks of pregnancy-induced hypertension have been demonstrated in both large retrospective studies and small observational studies (16-18). Furthermore, increased fetal growth retardation, premature births, and fetal morbidity were observed in these patients (19, 20). Incidences of abortion and neonatal deaths in previous pregnancies were also significantly increased in women with SCT (6). These maternal and fetal complications are caused by an increase in cesarean section rates (3, 15). When deciding on modes of delivery, it is also important to test for fetal-pelvic disproportion, since fetomaternal morbidity and mortality increase with prolonged labor (9). In our study, cesarean sections and vaginal births were observed in SCA groups despite the small number of patients. A higher number of cesarean sections were detected in the SCT group.
Dystocia due to pelvic narrowing is one indication that a cesarean section may be necessary. In this respect, assessments of exact pelvic diameters in women with SCA and SCT may decrease cesarean rates in these patients. Various radiological techniques are useful for clinical examinations in conformity assessments for vaginal delivery. In this context, various radiological techniques are helpful for clinical examinations in conformity assessments for vaginal delivery. Radiographic pelvimetry has been used for most of this century to predict obstetric results although there is substantial variation for routine utilization in practice. Its use is now being criticized mainly due to the high radiation dose required (21). Newer technologies such as computed tomography and MR scanning are increasingly being favored in this area, especially for MR pelvimetry, which does not involve ionizing radiation (14, 22). Moreover, calculation mistakes for radiographic imaging occur at a rate of approximately 10%; whereas, MR scanning is much more accurate with mistakes occurring at a rate of approximately 1% (7).
MR pelvimetry was introduced in 1985 by Stark et al. It provides pelvic sizes in all planes when imaging soft tissue structures, including the fetus (11). MR pelvimetry has replaced with conventional radiography and computed tomography because it does not contain ionizing radiation and provides accurate and objective data (12).
Keller et al. (12) conducted the most extensive research about MR pelvimetry. Pelvimetric data from 781 women were reviewed and correlated with obstetric backgrounds to reproduce standard values. In this study, pelvimetric measurements were lower in women who had cesarean sections or vacuum extractions when compared with those who delivered vaginally. The pelvimetric parameters associated with the largest measurement mistakes are the intertuberous and sagittal outlet.
Gowri et al. (14) conducted the second most extensive series of studies concerning MR pelvimetry. Pelvimetric data were reviewed from 125 women after previous cesarean sections. All the diameters except the sagittal inlet were significantly larger in women who delivered normally when compared with those who had cesarean sections for any reason. An outlet index and pelvic diameters (transverse inlet, sagittal outlet, interspinous, and intertuberous diameter) were useful cut-off points for vaginal deliveries within their study populations.
Korhonen et al. (23) evaluated 100 MR pelvimetry examinations. Pelvimetric parameters of the pelvic inlet and outlet were measured four times to determine the standard reference for each measurement, and then intra- and inter-observer variations were compared. They found that millimeter differences were insignificant in MR pelvimetry.
Our study has demonstrated that pelvimetric dimensions (sagittal inlet, sagittal mid-pelvis, transverse inlet, transverse mid-pelvis, and transverse outlet) of women with SCA and SCT are indistinguishable from healthy women.
There were some limitations in our study. First, we had a small sample size. Second, MR has a relatively high cost and limited availability. We used age and sex matched control group without body mass index matching due to low weight of SCA patient according to population. This may also cause partial bias and present as another limitation of our study.
Finally, our study demonstrated that the pelvic roofs of women with SCA and SCT are indistinguishable from healthy women. We think that these patients choose cesarean section/ rather than dystocia because of other pregnancy complications. Therefore, the rates of fetomaternal incompatibilities affecting delivery types are the same in healthy individuals and women with SCA and SCT. When making decisions about modes of delivery, these results should be taken into consideration.