1. Background
Cardiovascular diseases (CVDs) are one of the main causes of mortality in women (1, 2). The risk of mortality and morbidity is significantly higher among women with preeclampsia (3). Evidence suggests that preeclampsia can increase the risk of CVDs among women later in life (4). Besides, preeclampsia can be considered an independent risk factor for future coronary artery disease (4-6).
Preeclampsia and CVDs have common risk factors, including obesity, dyslipidemia, insulin resistance, diabetes mellitus, inflammation, preexisting hypertension, thrombophilia, and a family history of heart disease and stroke (7, 8). Besides, metabolic changes due to preeclampsia, such as insulin resistance, can accelerate the progression of atherosclerosis in these patients (8, 9). It is known that the plasma and serum markers of endothelial activation are elevated in preeclampsia. Therefore, it is necessary to pay attention to endothelial dysfunction as a definite pathogenic mechanism in atherosclerotic changes and preeclampsia (8, 10).
On the other hand, recent studies have suggested increased carotid intima-media thickness (CIMT) based on high-resolution B-mode ultrasonography as an independent risk factor for predicting early atherosclerosis (11). The measurement of intima-media thickness (IMT) of the common carotid artery and common femoral artery is a simple non-invasive marker for evaluating atherosclerosis (8, 12).
2. Objectives
The present study aimed to determine the relationship between preeclampsia and CIMT. For this purpose, the IMTs of the common carotid artery, carotid bulb, internal carotid artery, and common and superficial femoral arteries were measured in pregnant women with preeclampsia and then compared with those of normotensive pregnant and nulligravid women.
3. Patients and Methods
3.1. Study Design
This case-control study was conducted between May 2019 and January 2020. The ethics committee of Golestan University of Medical Sciences evaluated and approved this study (ethical code: IR.GOUMS.REC.1398.287). Pregnant women, hospitalized in Sayyad Shirazi Hospital (Gorgan, Iran) because of early-onset preeclampsia (gestational age < 34 weeks), were enrolled in the study. Besides, two control groups were recruited, including normotensive pregnant women and nulligravid women.
3.2. Inclusion and Exclusion Criteria
The diagnosis of preeclampsia in pregnant women was based on clinical and laboratory criteria by the International Society for the Study of Hypertension in Pregnancy (ISSHP) (13). The controls were matched with the patient group regarding age, sex, and body mass index (BMI). On the other hand, the exclusion criteria were as follows: women with a history of chronic hypertension; gestational hypertension; clinical evidence of heart disease or atherosclerosis; kidney disease; smoking; diabetes; thyroid disorders; collagen vascular disease; history of cardiovascular treatments; multiple pregnancies; multiparity; miscarriage; and unwillingness to continue the study.
3.3. Imaging
The IMTs of carotid and femoral arteries were measured bilaterally, using a Philips ultrasonography machine and a 7-12 MHz linear surface probe; these measurements were performed in the supine position. The arterial segments were defined as follows: A segment 1 cm proximal to the carotid dilation as the common carotid arterial wall; a segment between the carotid flow divider and carotid dilation as the carotid bulb; a 1-cm arterial segment distal to the flow divider as the internal carotid; a 1-cm arterial segment proximal to the femoral dilation as the common femoral artery; and a 1-cm arterial segment distal to the flow divider as the superficial femoral artery. Expert radiologists with more than 10 years of experience performed ultrasonography.
3.4. Statistical Analysis
Data were analyzed in SPSS version 25 (IBM Corp. Released 2017. IBM SPSS Statistics for Windows, Version 25.0. Armonk, NY: IBM Corp.). After assessing the normal distribution of data, one-way analysis of variance (ANOVA) was used to investigate differences in quantitative data between the groups. The least significant difference (LSD) test was used in ANOVA as the post hoc test. P-values less than 0.05 were considered significant.
4. Results
A total of 300 women were enrolled in this study, each group consisting of 100 participants. The basic characteristics of the participants are summarized in Table 1. The means ± standard deviation (SD) of age in preeclamptic, normotensive and nulligravid women were 27.78 ± 3.23, 27.16 ± 3.09, and 27.11 ± 2.95, respectively. The mean ± SD of gestational age in preeclamptic and normotensive pregnant women was 31.65 ± 3.028 and 32.52 ± 3.075 weeks, respectively. The levels of high-density lipoprotein (HDL), low-density lipoprotein (LDL), cholesterol, and triglyceride were not significantly different between the groups (Table 1).
| Variables | Preeclamptic pregnant women | Normotensive pregnant women | Nulligravid women |
|---|---|---|---|
| Age (y) | 27.78 ± 3.23 | 27.16 ± 3.09 | 27.11 ± 2.95 |
| Gestational age (weeks) | 31.65 ± 3.028 | 32.52 ± 3.075 | - |
| Body mass index (kg/m2) | 25.77 ± 3.22 | 25.18 ± 3.22 | 24.92 ± 3.62 |
| Cholesterol (mg/dL) | 188.32 ± 34.08 | 179.54 ± 33.35 | 182.63 ± 32.27 |
| Triglyceride (mg/dL) | 142.27 ± 40.81 | 134.46 ± 35 | 140.32 ± 42.19 |
| High-density lipoproteins (HDL) (mg/dL) | 51.24 ± 7.59 | 52.74 ± 7.30 | 51.25 ± 7.03 |
| Low-density lipoproteins (LDL) (mg/dL) | 108.55 ± 35.61 | 99.90 ± 35.44 | 103.32 ± 34.06 |
a Values are expressed as mean ± SD.
The systolic and diastolic blood pressure, mean arterial pressure (MAP), and heart rate measurements are summarized in Table 2. The mean systolic and diastolic blood pressure and MAP were significantly higher in the preeclamptic group as compared to the other groups (P < 0.001) (Table 2). The mean systolic and diastolic blood pressure and MAP were higher in the controls compared to normotensive pregnant women; however, the differences were not significant. There was no significant difference between the three groups in terms of heart rate.
| Variables | Normotensive pregnant women | Preeclamptic pregnant women | Nulligravid women | P-value |
|---|---|---|---|---|
| Systolic blood pressure (mmHg) | 108.55 ± 12.35 | 135.30 ± 17 | 111.75 ± 11.48 | < 0.001 |
| Diastolic blood pressure (mmHg) | 69.35 ± 9.03 | 87.15 ± 11.06 | 71.95 ± 8 | < 0.001 |
| Mean arterial pressure (mmHg) | 82.42 ± 9.372 | 103.2 ± 12.55 | 85.22 ± 8.625 | < 0.001 |
| Heart rate | 82.50 ± 11.03 | 83.64 ± 15.27 | 81.9 ± 10.45 | < 0.001 |
a Values are expressed as mean ± SD.
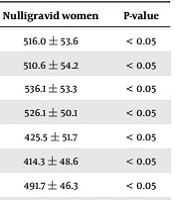
The IMT measurements are presented in Table 3. The thickness of right and left common carotid and internal carotid arteries, right and left carotid bulbs, and right and left common femoral and superficial femoral arteries was significantly higher in the preeclamptic group compared to normotensive pregnant and nulligravid women (Table 3). The thickness of right and left common carotid arteries, right and left carotid bulbs, right and left internal carotid arteries, right and left common femoral arteries, and right superficial femoral artery was higher among normotensive pregnant women compared to healthy controls; however, the differences were not significant (P > 0.05).
| Variables | Normotensive pregnant women | Preeclamptic pregnant women | Nulligravid women | P-value |
|---|---|---|---|---|
| Right common carotid artery (mm) | 525.2 ± 51.1 | 577.1 ± 54.2 | 516.0 ± 53.6 | < 0.05 |
| Left common carotid artery (mm) | 518.4 ± 54 | 573.5 ± 49.9 | 510.6 ± 54.2 | < 0.05 |
| Right carotid bulb (mm) | 538.2 ± 52.2 | 558.2 ± 55.5 | 536.1 ± 53.3 | < 0.05 |
| Left carotid bulb (mm) | 533.9 ± 49.5 | 558.4 ± 50.2 | 526.1 ± 50.1 | < 0.05 |
| Right internal carotid artery (mm) | 429.5 ± 43.4 | 447.7 ± 56.37 | 425.5 ± 51.7 | < 0.05 |
| Left internal carotid artery (mm) | 422.5 ± 47.98 | 446.1 ± 48.4 | 414.3 ± 48.6 | < 0.05 |
| Right common femoral artery (mm) | 501 ± 44.2 | 522.4 ± 47.2 | 491.7 ± 46.3 | < 0.05 |
| Left common femoral artery (mm) | 486.2 ± 44.6 | 516 ± 44.3 | 476.1 ± 44.4 | <0.05 |
| Right superficial femoral artery (mm) | 412.8 ± 57.1 | 430.8 ± 47.6 | 411.4 ± 52.5 | < 0.05 |
| Left superficial femoral artery (mm) | 396.2 ± 45.4 | 428.5 ± 45.4 | 397.5 ± 45.3 | < 0.05 |
a Values are expressed as mean ± SD.
5. Discussion
In clinical settings, evaluation of CIMT helps with the identification of patients with advanced subclinical atherosclerosis, as well as noninvasive quantification of its severity. In the present study, the IMT of all measured sections, including the common and internal carotid arteries, right and left carotid bulbs, and right and left superficial femoral arteries, increased significantly in the group of preeclamptic women compared to the other groups. These findings suggest that subclinical atherosclerosis occurred due to preeclampsia in these women. According to previous studies, an acute rise in blood pressure could lead to acute changes in IMT, even in healthy people (14). According to this finding and other relevant evidence, preeclampsia can cause a chronic defect in systemic and pulmonary circulation in women (15-18).
The main hypothesis of the present study is that preeclampsia leads to a higher risk of CVD by increasing the CIMT. This hypothesis represents three causal relationships. First, the risk of CVD is higher among women with preeclampsia; second, CIMT can be considered a marker of CVD risk; and third, preeclampsia increases CIMT among pregnant women. There is some evidence supporting the first proposition, including a systematic review and meta-analysis by Brown et al. (19), which reported that women diagnosed with preeclampsia are at an increased risk of future cardiovascular or cerebrovascular events. Moreover, Lorenz et al. (20) performed a systematic review and meta-analysis and verified that CIMT is a strong predictor of future vascular events; this study approved the second proposition. Another systematic review and meta-analysis by Pikir et al. (3) reported that CIMT was higher among women with a history of preeclampsia compared to normal pregnant women; these results approve the third proposition. The present results are also consistent with the third proposition.
Although the study by Pikir et al. (3) supported the relationship between CIMT and preeclampsia, there are ongoing controversies. Some studies suggest CIMT increase in women with preeclampsia, similar to the findings of the present study. Blaauw et al. (8) reported a significant increase in the IMT of common femoral arteries in preeclamptic pregnant women compared to nulligravid and normotensive pregnant women. Additionally, Yuan et al. (21) reported that the IMT and internal diameter of the carotid artery were significantly higher in the group of women with preeclampsia compared to healthy pregnant women. Besides, Andersgaard et al. (22) showed that carotid IMT was significantly higher in women with a history of preeclampsia compared to pregnant women without hypertension and proteinuria in previous pregnancies.
In a study by Gaugler-Senden et al. (23), women with a history of preeclampsia before 24 weeks of gestation showed a significant increase in the carotid artery IMT compared to women with healthy pregnancies (P = 0.03). Moreover, Stergiotou et al. (24) reported that CIMT was significantly higher in women with late and premature preeclampsia compared to the control group. In another study by Brueckmann et al. (25), the carotid IMT was higher in women with preeclampsia compared to normotensive pregnant women, as well as non-pregnant control women. Besides, Verissimo (26) reported a significant increase in the carotid IMT in women with hypertension (chronic hypertension or preeclampsia) compared to healthy pregnant women. Lopes Ramos and Neto (27) also found that CIMT was significantly higher in women with preeclampsia as compared to healthy pregnant women.
On the other hand, several studies revealed that CIMT was not significantly higher among women with preeclampsia. In this regard, Haukkamaa et al. (6) reported that CIMT was higher in women with a history of preeclampsia compared to the other groups, although the increase in thickness was not statistically significant. Akhter et al. (28) found that during pregnancy and also one year after delivery, the IMT difference was not significant between women with preeclampsia and 64 healthy pregnant women. Additionally, Mori et al. (29) showed no significant difference in CIMT between pregnant women with preeclampsia and healthy pregnant women. These controversies can be the results of other factors that can affect CIMT beside preeclampsia. The risk factors might have affected the findings of previous studies, and consequently, there is heterogeneity regarding the relationship between CIMT and preeclampsia in these studies.
Traditional cardiovascular risk factors that are related to CIMT include smoking, alcohol use, age, sex, race, dietary patterns, blood pressure, dyslipidemia, glycemia, habitual endurance exercise, hyperuricemia, obesity-related anthropometric parameters, obesity, and obesity-related diseases. Besides, novel risk factors for CIMT include heredity factors, immunological diseases, certain genotypic indices, rheumatoid arthritis, inflammatory cytokines, lipid peroxidation, infectious diseases, matrix metalloproteinases, and other novel factors and diseases (30). In most previous studies, only some traditional risk factors were investigated, and the majority of them were discarded; this may be a cause of discrepancy between the findings of previous studies. Since the novel risk factors can reduce the effects of confounding factors in future studies, the relationship between CIMT and preeclampsia will become clearer. To determine whether preeclampsia independently causes this pathology or whether these findings are outcomes of other concomitant conditions that appear inevitably (e.g., increased blood pressure), future investigations are needed to compare preeclamptic patients with gestational hypertensive women after matching for blood pressure.
The present study had some limitations. First, there was no group of non-preeclamptic pregnant women with hypertension to compare with preeclamptic women and to investigate the effects of hypertension compared to preeclampsia on CIMT. Second, the intima, media, or intima/media ratios were not measured in this study. Third, carotid plaque was not reported for the participants. Although the limitations did not affect the results considerably, attention to these factors can improve the quality of future research.
In conclusion, preeclampsia was associated with an increased IMT of common and internal carotid arteries, carotid bulbs, and common and superficial femoral arteries. As CMIT can be considered a risk factor for CVD, and the risk of CVD is higher in women with preeclampsia, IMT measurements in preeclamptic women can provide an opportunity to identify those who may benefit from early screening and preventive care to reduce the risk of CVDs in the future.