1. Background
2. Objectives
3. Patients and Methods
3.1. Patients and Data Collection
Study flow chart. According to the percentage of initial drainage volume per CT volume of hepatic abscess, we divided the patients into two groups: group A and B. Moreover, the KHA group and non-KHA group were categorized into the following groups according to the percentage of initial drainage volume per CT volume: group C and D for patients in the KHA group, and group E and F for patients in the non-KHA group. Abbreviations: KHA; Klebsiella pneumonia with monomicrobial hepatic abscess, Non-KHA; non-Klebsiella pneumoniae monomicrobial or polymicrobial pyogenic hepatic abscess.
3.2. CT Technique
3.3. CT Interpretation
3.4. Statistical Analysis
4. Results
4.1. Comparison Between Monomicrobial K. Pneumoniae Hepatic Abscesses and Others
| Characteristics | KHA Group (n = 60) | Non-KHA Group (n = 27) | P Value |
|---|---|---|---|
| Age (y)b | 62.3 ± 13.5 | 64.5 ± 11.5 | 0.462 |
| Sex, male | 38 (63.3) | 17 (63.0) | 0.974 |
| Symptom | |||
| Fever | 42 (70.0) | 19 (70.4) | 0.972 |
| RUQ pain | 19 (31.7) | 7 (25.9) | 0.588 |
| Underlying disease | |||
| Diabetes mellitus | 15 (25.0) | 5 (18.5) | 0.506 |
| Malignancy | 4 (6.7) | 13 (48.2) | 0.001 |
| Biliary disease | 13 (21.7) | 16 (59.3) | 0.006 |
| Interval between symptom onset and final CT prior to the procedure (days)b | 7.7 ± 6.1 | 7.2 ± 7.1 | 0.775 |
| Interval between symptom onset and the procedure day (days)b | 9.0 ± 6.0 | 8.5 ± 7.2 | 0.754 |
| White blood cell count (x109/L)b | 14.5 ± 7.8 | 14.7 ± 7.6 | 0.927 |
| Neutrophil count (x109/L)b | 12.7 ± 7.7 | 12.5 ± 7.4 | 0.926 |
| C-reactive protein level (mg/dL)b | 26.5 ± 34.3 | 19.9 ± 9.5 | 0.173 |
| Size (cm)b | 6.9 ± 2.2 | 6.4 ± 2.3 | 0.371 |
| Number | 0.124 | ||
| Single | 49 (81.7) | 18 (66.7) | |
| Multiple | 11 (18.3) | 9 (33.3) | |
| Location | 0.211 | ||
| Right | 41 (68.3) | 19 (70.4) | |
| Left | 5 (8.3) | 5 (18.5) | |
| Both | 14 (23.3) | 3 (11.1) | |
| Configuration | 0.031 | ||
| Unilocular | 4 (6.7) | 7 (25.9) | |
| Multilocular | 56 (93.3) | 20 (74.1) | |
| Appearance | 0.327 | ||
| Cystic | 50 (83.3) | 25 (92.6) | |
| Solid | 10 (16.7) | 2 (7.4) | |
| Wall thickness | 0.878 | ||
| Thin wall | 21 (35.0) | 9 (33.3) | |
| Thick wall | 39 (65.0) | 18 (66.7) | |
| Margin | 0.456 | ||
| Ill-defined | 13 (21.7) | 4 (14.8) | |
| Well-defined | 47 (78.3) | 23 (85.2) | |
| Rim enhancement | 31 (51.7) | 22 (81.5) | 0.008 |
| Presence of gas density | 5 (8.3) | 7 (25.9) | 0.028 |
| The shortest length to the liver capsule (cm)b | 0.5 ± 1.1 | 0.3 ± 0.5 | 0.206 |
| Mean attenuation (HU)b | 39.2 ± 11.9 | 38.2 ± 17.5 | 0.796 |
| CT volume (mL)b | 103.6 ± 211.3 | 67.2 ± 53.7 | 0.216 |
| Amount of initial drainage (mL)b | 36.5 ± 36.8 | 36.0 ± 34.7 | 0.956 |
| Percentage of initial drainage volume per CT Volume (%)b | 51.2 ± 35.7 | 62.7 ± 46.3 | 0.209 |
Abbreviations: KHA, Klebsiella pneumoniae monomicrobial pyogenic hepatic abscess; Non-KHA, non-Klebsiella pneumoniae monomicrobial or polymicrobial pyogenic hepatic abscess; RUQ; right upper quadrant, HU; hounsfield unit; y, year; PCD, Percutaneous catheter drainage.
aValues are expressed as No. (%) unless otherwise indicated, percentages are calculated on the basis of each group.
bData are means ± standard deviations
4.2. Percentage of Drainage Volume Per CT Colum of the Abscess (Group A Vs. Group B)
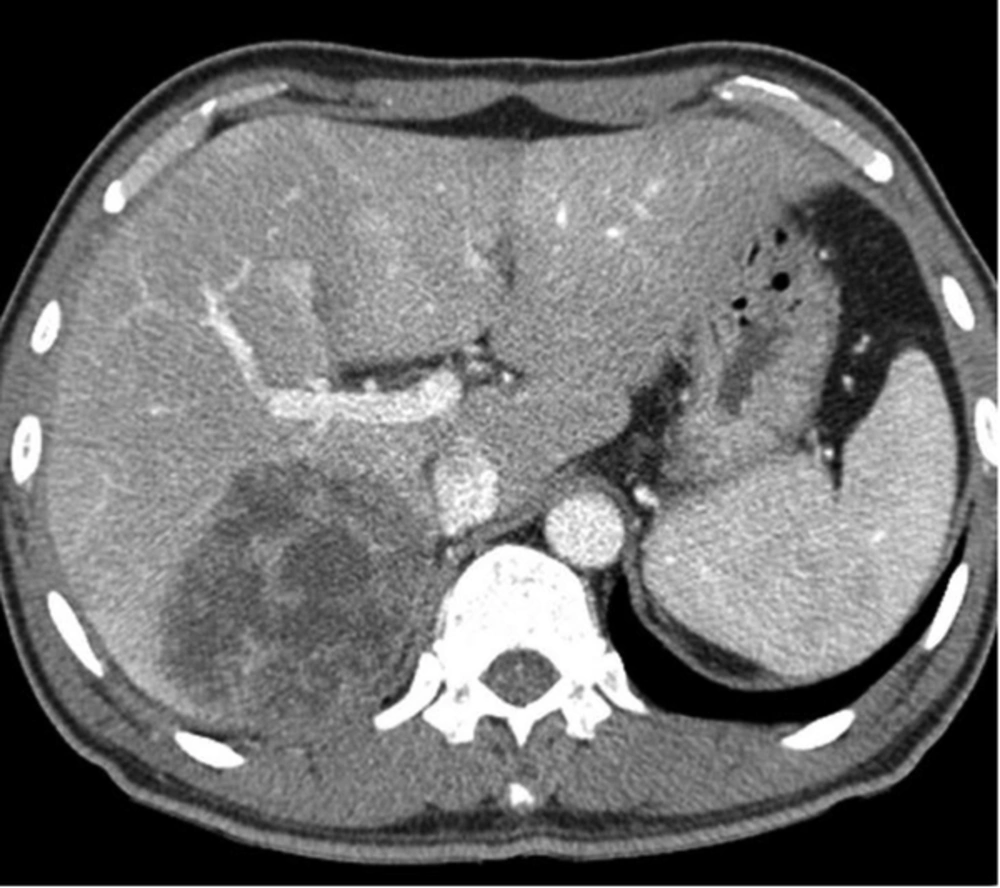
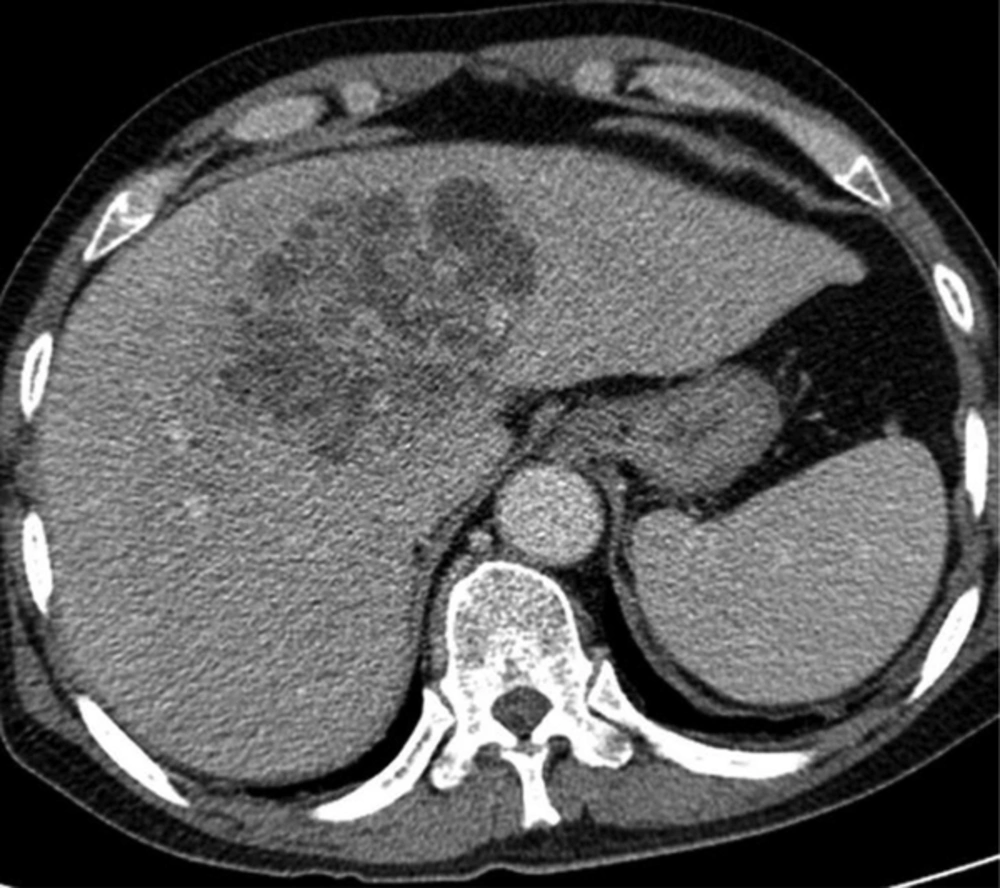
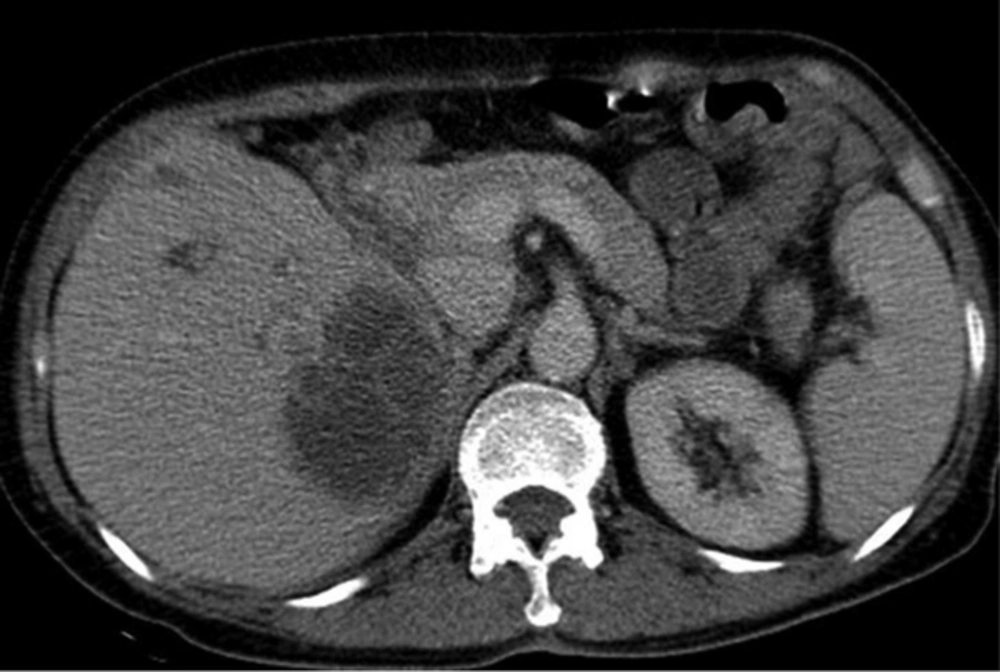
A 50-year-old man with Klebsiella pneumoniae hepatic abscess in group A. Contrast enhanced CT scan shows a single hepatic abscess in the right liver. It is a well-defined, multiloculated, and thin-walled abscess with no rim enhancement. The abscess has a multiple septa and solid appearance with less than 50% liquefied part of the abscess. The mean attenuation was calculated as 47 hounsfield unit (HU).
| Characteristics | KHA Group (n = 60) | Non-KHA Group (n = 27) | P Value |
|---|---|---|---|
| Age (y)d | 62.3 ± 13.5 | 64.5 ± 11.5 | 0.462 |
| Sex, male | 38 (63.3) | 17 (63.0) | 0.974 |
| Symptom | |||
| Fever | 42 (70.0) | 19 (70.4) | 0.972 |
| RUQ pain | 19 (31.7) | 7 (25.9) | 0.588 |
| Underlying disease | |||
| Diabetes mellitus | 15 (25.0) | 5 (18.5) | 0.506 |
| Malignancy | 4 (6.7) | 13 (48.2) | 0.001 |
| Biliary disease | 13 (21.7) | 16 (59.3) | 0.006 |
| Interval between symptom onset and final CT prior to the procedure (days)d | 7.7 ± 6.1 | 7.2 ± 7.1 | 0.775 |
| Interval between symptom onset and the procedure day (days)d | 9.0 ± 6.0 | 8.5 ± 7.2 | 0.754 |
| White blood cell count (x109/L)d | 14.5 ± 7.8 | 14.7 ± 7.6 | 0.927 |
| Neutrophil count (x109/L)d | 12.7 ± 7.7 | 12.5 ± 7.4 | 0.926 |
| C-reactive protein level (mg/dL)d | 26.5 ± 34.3 | 19.9 ± 9.5 | 0.173 |
| Size (cm)d | 6.9 ± 2.2 | 6.4 ± 2.3 | 0.371 |
| Number | 0.124 | ||
| Single | 49 (81.7) | 18 (66.7) | |
| Multiple | 11 (18.3) | 9 (33.3) | |
| Location | 0.211 | ||
| Right | 41 (68.3) | 19 (70.4) | |
| Left | 5 (8.3) | 5 (18.5) | |
| Both | 14 (23.3) | 3 (11.1) | |
| Configuration | 0.031 | ||
| Unilocular | 4 (6.7) | 7 (25.9) | |
| Multilocular | 56 (93.3) | 20 (74.1) | |
| Appearance | 0.327 | ||
| Cystic | 50 (83.3) | 25 (92.6) | |
| Solid | 10 (16.7) | 2 (7.4) | |
| Wall thickness | 0.878 | ||
| Thin wall | 21 (35.0) | 9 (33.3) | |
| Thick wall | 39 (65.0) | 18 (66.7) | |
| Margin | 0.456 | ||
| Ill-defined | 13 (21.7) | 4 (14.8) | |
| Well-defined | 47 (78.3) | 23 (85.2) | |
| Rim enhancement | 31 (51.7) | 22 (81.5) | 0.008 |
| Presence of gas density | 5 (8.3) | 7 (25.9) | 0.028 |
| The shortest length to the liver capsule (cm)d | 0.5 ± 1.1 | 0.3 ± 0.5 | 0.206 |
| Mean attenuation (HU)d | 39.2 ± 11.9 | 38.2 ± 17.5 | 0.796 |
| CT volume (mL)d | 103.6 ± 211.3 | 67.2 ± 53.7 | 0.216 |
| Amount of initial drainage (mL)d | 36.5 ± 36.8 | 36.0 ± 34.7 | 0.956 |
| Percentage of initial drainage volume per CT Volume (%)d | 51.2 ± 35.7 | 62.7 ± 46.3 | 0.209 |
Abbreviations: RUQ; right upper quadrant, HU; hounsfield unit; PCD, Percutaneous catheter drainage; y, year.
aValues are expressed as No. (%) unless otherwise indicated, percentages are calculated on the basis of each group.
bPercentage of initial drainage volume per CT volume < 50%
cPercentage of initial drainage volume per CT volume ≥ 50%
dData are means ± standard deviations
4.3. Comparison Between the KHA Group with Poor Drainage and Good Drainage (Group C Vs. Group D)
| Characteristics | Group Cb (n = 30) | Group Dc (n = 30) | P Value |
|---|---|---|---|
| Size (cm)d | 7.0 ± 2.4 | 6.8 ± 2.0 | 0.808 |
| Number | 0.739 | ||
| Single | 24 (80.0) | 25 (83.3) | |
| Multiple | 6 (20.0) | 5 (16.7) | |
| Location | 0.894 | ||
| Right | 21 (70.0) | 20 (66.7) | |
| Left | 2 (6.7) | 3 (10.0) | |
| Both | 7 (23.3) | 7 (23.3) | |
| Configuration | 0.612 | ||
| Unilocular | 3 (10.0) | 1 (3.3) | |
| Multilocular | 27 (90.0) | 29 (96.7) | |
| Appearance | 0.006 | ||
| Cystic | 21 (70.0) | 29 (96.7) | |
| Solid | 9 (30.0) | 1 (3.3) | |
| Wall thickness | < 0.001 | ||
| Thin wall | 19 (63.3) | 2 (6.7) | |
| Thick wall | 11 (36.7) | 28 (93.3) | |
| Margin | 1.000 | ||
| Ill-defined | 7 (23.3) | 6 (20.0) | |
| Well-defined | 23 (76.7) | 24 (80.0) | |
| Rim enhancement | 13 (43.3) | 18 (60.0) | 0.197 |
| Presence of gas density | 3 (10.0) | 2 (6.7) | 1.000 |
| The shortest length to the liver capsule (cm)d | 0.7 ± 1.4 | 0.3 ± 0.6 | 0.194 |
| Mean attenuation (HU)d | 44.1 ± 11.8 | 34.2 ± 10.0 | 0.001 |
| CT volume (mL)d | 138.7 ± 291.8 | 68.4 ± 55.5 | 0.204 |
Abbreviations: HU; hounsfield unit; PCD, Percutaneous catheter drainage.
aValues are expressed as No. (%) unless otherwise indicated, percentages are calculated on the basis of each group.
bPercentage of initial drainage volume per CT volume < 50%
cPercentage of initial drainage volume per CT volume ≥ 50%
dData are means ± standard deviations.
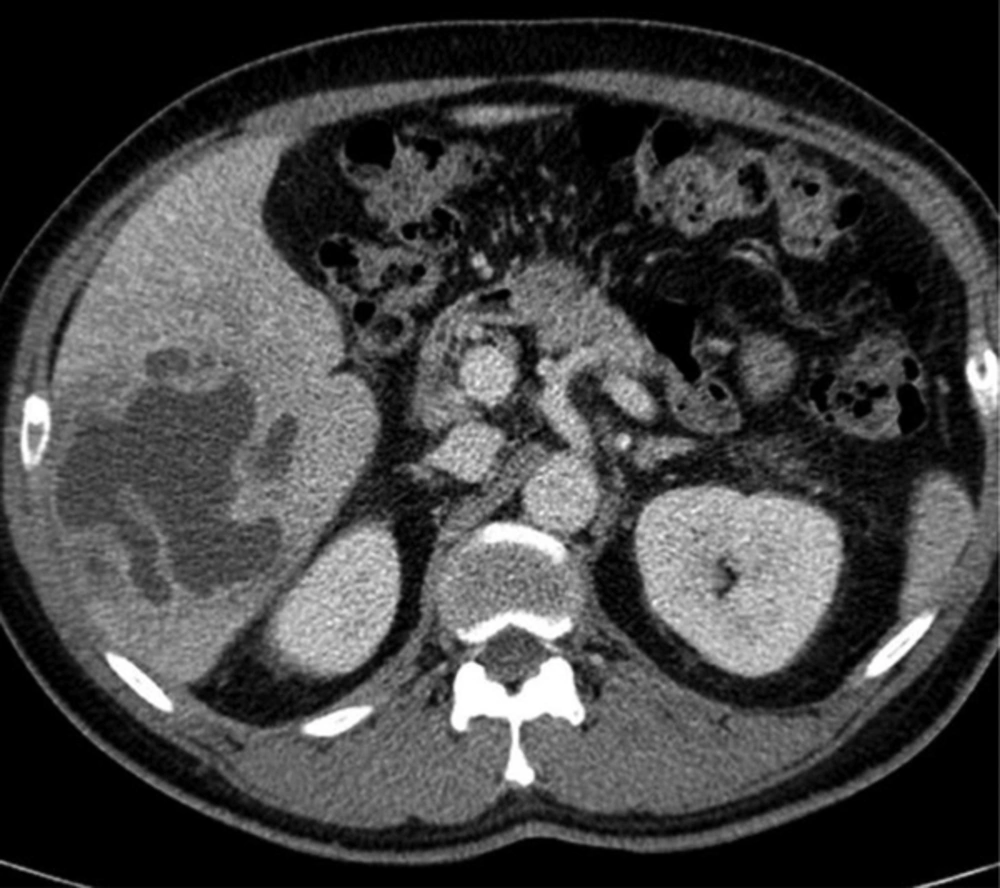
A 61-year-old man with Klebsiella pneumoniae hepatic abscess in group D. Contrast enhanced CT scan shows a single multiloculated abscess with cystic appearance, and thick wall in the right liver. The mean attenuation was calculated as 20 hounsfield unit (HU). The abscess was successfully treated with percutaneous catheter drainage. The patient was discharged from hospital 29 days later.
4.4. Comparison Between the Non-KHA Group with Poor Drainage and Good Drainage (Group E Vs. Group F)
| Characteristics | Group Eb (n = 11) | Group Fc (n = 16) | P Value |
|---|---|---|---|
| Size (cm)d | 6.7 ± 2.2 | 6.3±2.3 | 0.668 |
| Number | 1.000 | ||
| Single | 7 (63.6) | 11 (68.8) | |
| Multiple | 4 (36.4) | 5 (31.3) | |
| Location | 0.300 | ||
| Right | 9 (81.8) | 10 (62.5) | |
| Left | 2 (18.2) | 3 (18.8) | |
| Both | 0 | 3 (18.8) | |
| Configuration | 0.391 | ||
| Unilocular | 4 (36.4) | 3 (18.8) | |
| Multilocular | 7 (63.6) | 13 (81.3) | |
| Appearance | 0.157 | ||
| Cystic | 9 (81.8) | 16 (100.0) | |
| Solid | 2 (18.2) | 0 | |
| Wall thickness | 0.012 | ||
| Thin wall | 7 (63.6) | 2 (12.5) | |
| Thick wall | 4 (36.4) | 14 (87.5) | |
| Margin | 0.273 | ||
| Ill-defined | 3 (27.3) | 1 (6.3) | |
| Well-defined | 8 (72.7) | 15 (93.8) | |
| Rim enhancement | 6 (54.6) | 16 (100.0) | 0.006 |
| Presence of gas density | 2 (18.2) | 5 (31.3) | 0.662 |
| The shortest length to the liver capsule (cm)d | 0.4 ± 0.6 | 0.2 ± 0.5 | 0.518 |
| Mean attenuation (HU)d | 46.3 ± 21.5 | 32.6 ± 12.0 | 0.075 |
| CT volume (mL)d | 70.6 ± 61.8 | 64.8 ± 49.4 | 0.787 |
Abbreviations: HU; hounsfield unit; PCD, Percutaneous catheter drainage.
aValues are expressed as No. (%) unless otherwise indicated, percentages are calculated on the basis of each group.
bPercentage of initial drainage volume per CT volume < 50%
cPercentage of initial drainage volume per CT volume ≥ 50%
dData are means ± standard deviations.
4.5. Clinical Outcome Among the Groups
| Duration of Drainage (Days) | Number of Days for Which the Fever had Subsided (Days) | Number of Days for Which Leukocytosis had Subsided (Days) | Length of Hospitalization (Days) | |
|---|---|---|---|---|
| Group Ab | 7.1 ± 5.7 | 5.8 ± 3.5 | 5.1 ± 6.2 | 25.4 ± 13.6 |
| Group Bc | 8.7 ± 7.2 | 5.3 ± 5.4 | 6.1 ± 6.6 | 24.3 ± 11.1 |
| P Value | 0.248 | 0.640 | 0.454 | 0.706 |
| Group Cb | 7.3 ± 6.4 | 6.1 ± 3.7 | 5.5 ± 6.7 | 26.2 ± 10.8 |
| Group Dc | 8.1 ± 4.8 | 5.5 ± 3.7 | 4.9 ± 4.2 | 20.7 ± 7.7 |
| P Value | 0.619 | 0.511 | 0.663 | 0.026 |
| Group Eb | 6.5 ± 3.0 | 4.9 ± 2.7 | 3.9 ± 4.5 | 23.0 ± 19.9 |
| Group Fc | 10.0 ± 10.5 | 5.1 ± 7.9 | 8.4 ± 9.5 | 31.2 ± 13.4 |
| P Value | 0.229 | 0.943 | 0.111 | 0.251 |
aValues are expressed as mean ± SD.
bPercentage of initial drainage volume per CT volume < 50%.
cPercentage of initial drainage volume per CT volume ≥ 50%.