1. Introduction
The usual sites of metastases in pancreatic cancer are the liver and peritoneal cavity. Other less common sites are the lung, bone, and brain (1). Unusual sites such as muscle, skin, heart, pleura, stomach, umbilicus, kidney, appendix, spermatic cord, and prostate have also been reported (2-4). Skeletal muscle metastases from pancreatic cancer have by far been considered as an infrequent occurrence. Although the true incidence of skeletal muscle metastases in patients with pancreatic cancer is not known, skeletal muscle metastases from pancreatic carcinoma are exceedingly rare with only a few cases reported in the literature. We present a 34-year-old man with left calf pain, tumescence, and transient epigastric pain for 3 months. In CT and MRI, this appeared to be a primary soft tissue tumor. However, calf muscle biopsy demonstrated metastatic pancreatic neuroendocrine carcinoma.
2. Case Presentation
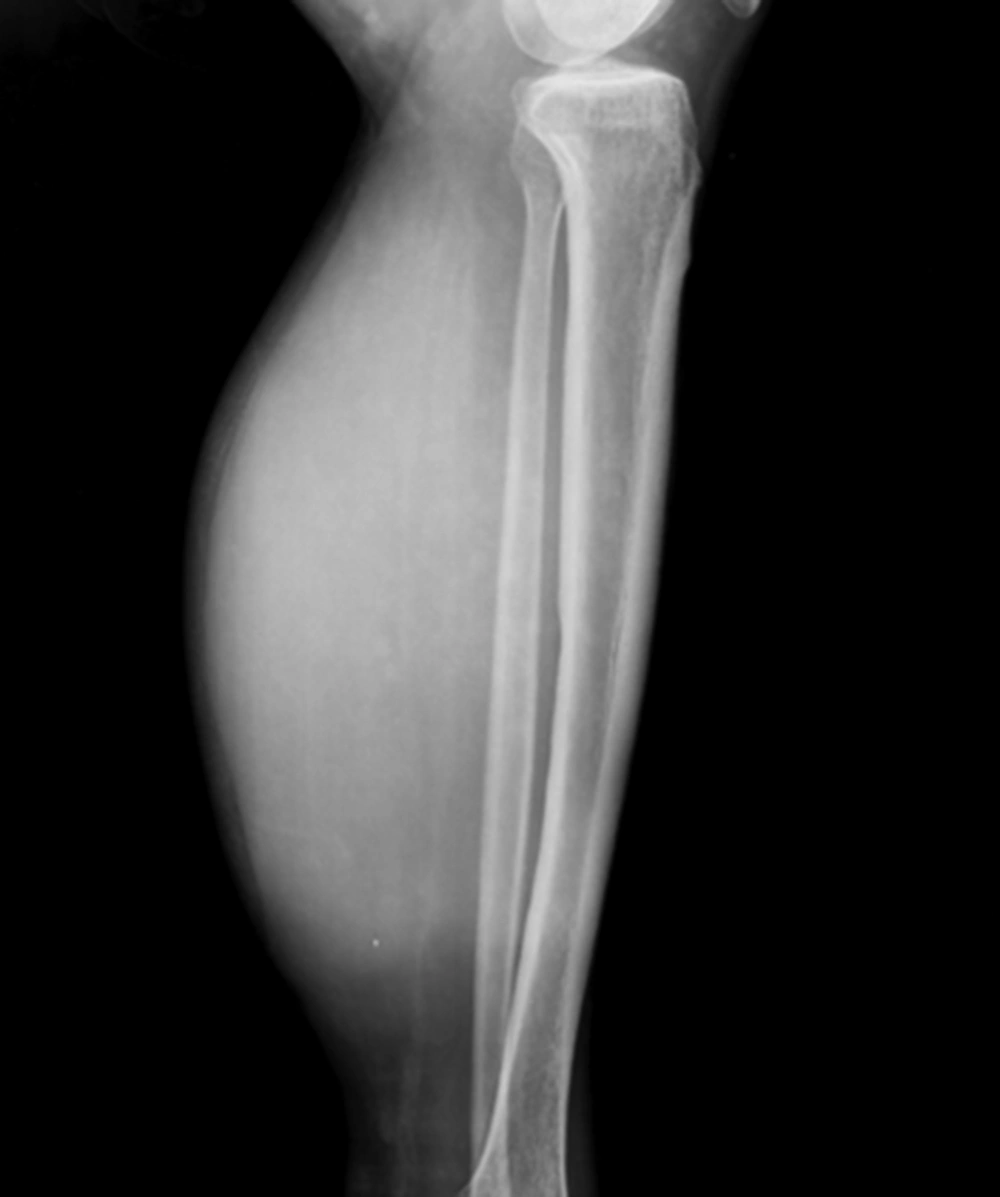
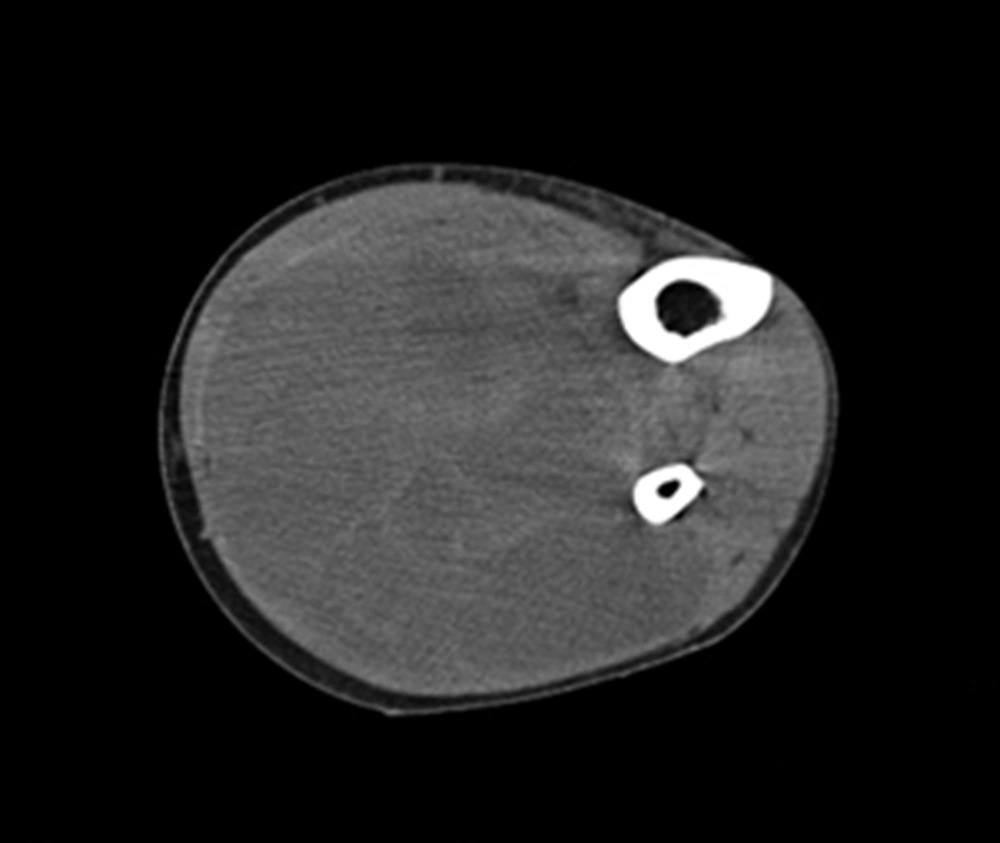
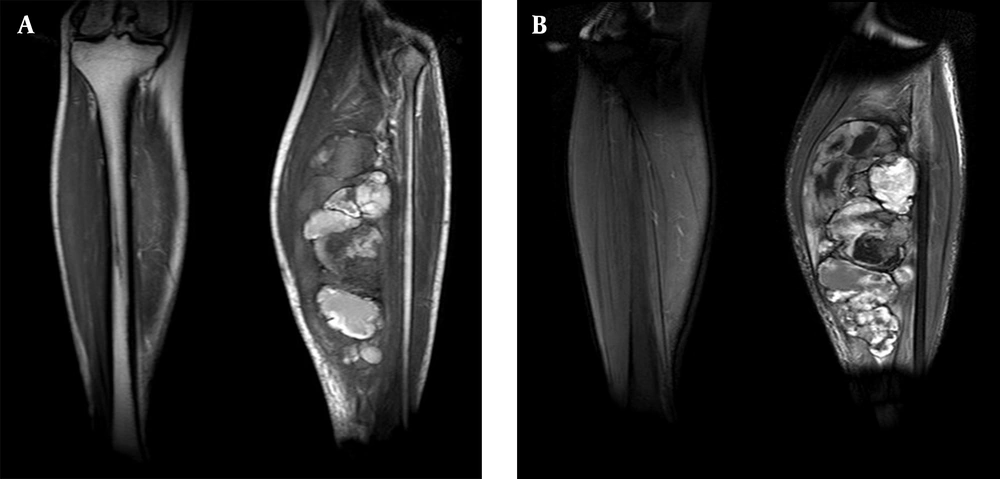
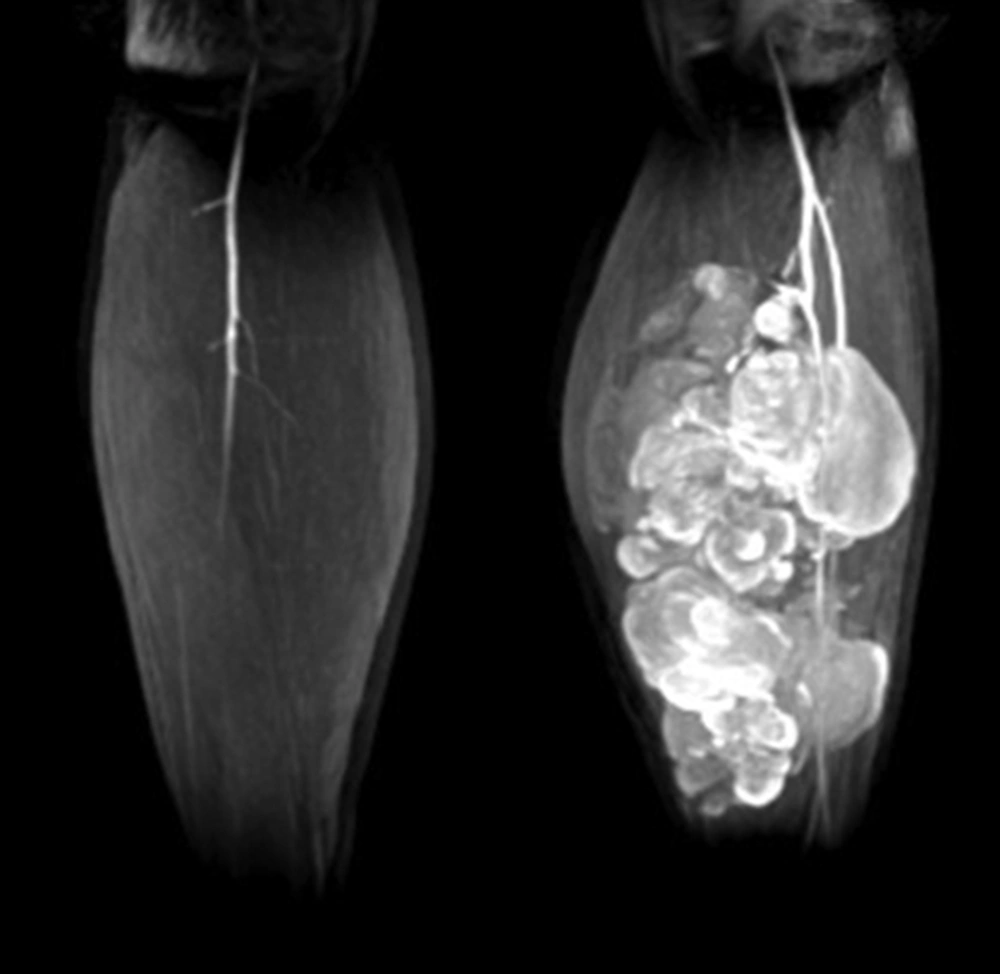
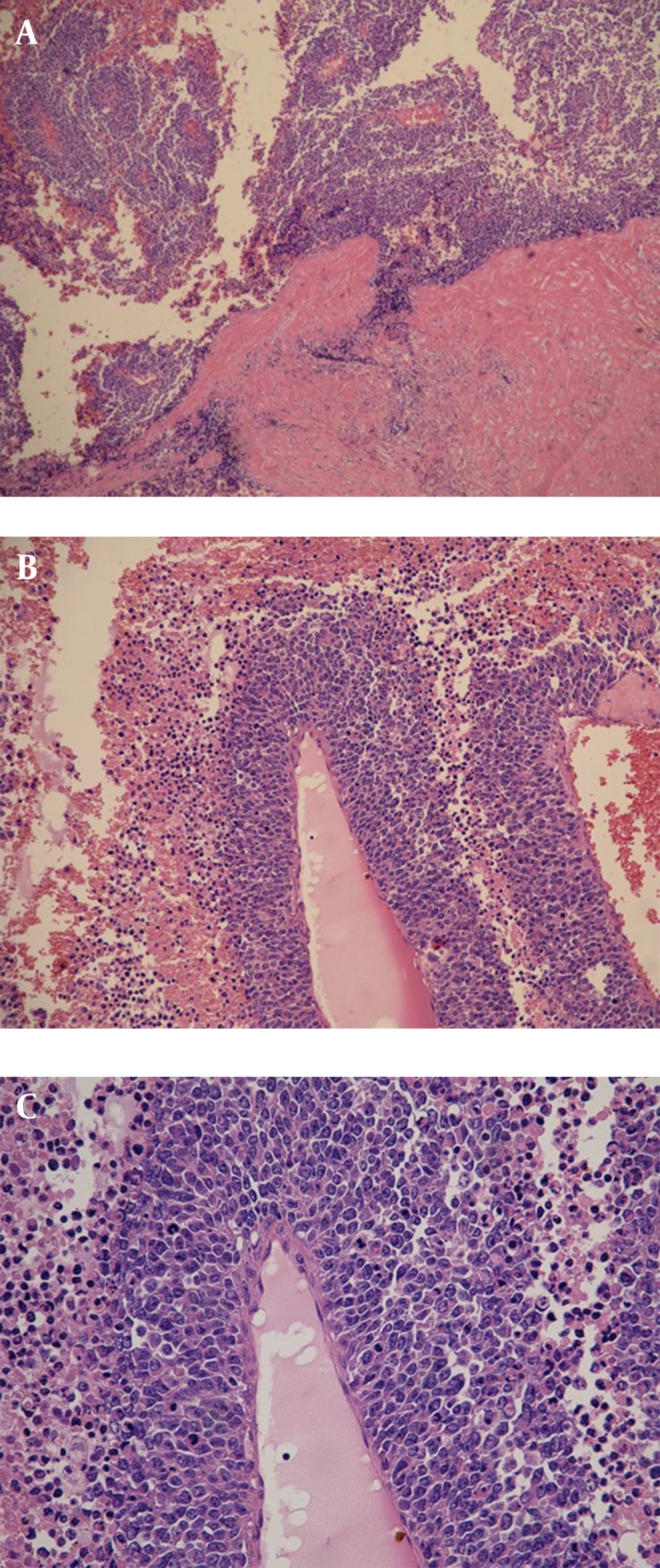
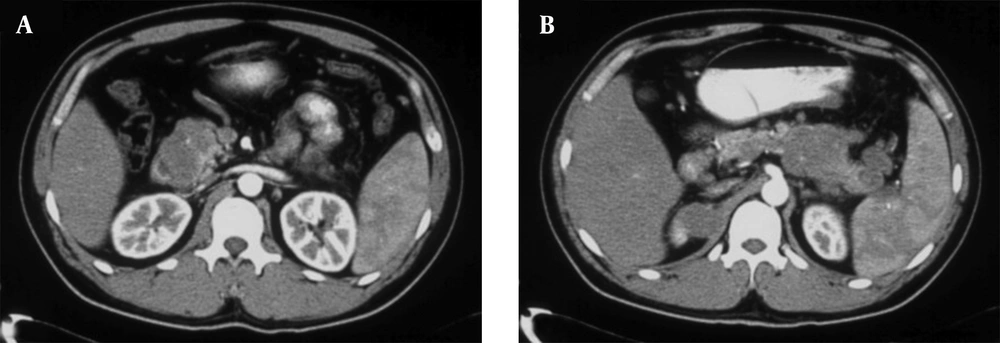
A 34-year-old man of Asian descent presented with a 3 month history of pain and tumescence in the left calf. He also complained of occasional and transient vague epigastric pain, without fever, nausea, or vomiting. There was no family history of gastrointestinal cancer. On physical examination, the left calf was swollen, with a large (8 cm) palpable lobulated and firm mass with an ill-defined margin. Abdominal examination was unremarkable: nontender and without any masses. Incidentally, his CA-125 level was 82.77 U/mL (reference range: 0-35 U/mL). Left calf plain films demonstrated a large soft tissue mass behind the tibia and fibula without a clear border but confined to the soft tissue without bony abnormality (Figure 1). Computerized tomography (CT) showed a heterogeneous mass with multiple fine compartments (Figure 2). The mass was confined to the soft tissue and did not involve the adjacent bone. Magnetic resonance imaging (MRI) displayed heterogeneous signal intensity on T1-weighted images and fat-suppression T2-weighted images (Figure 3 A, B) with obvious intensification (Figure 4). All these radiological characteristics suggested a primary soft tissue tumor. The patient underwent fine needle aspiration and biopsy. Pathology demonstrated metastatic neuroendocrine carcinoma (Figure 5 A-C). Subsequently, the patient underwent contrast CT of the abdomen, which showed a large, irregular, low-density mass in the head, body, and tail of the pancreas without obvious enhancement (Figure 6 A, B). Besides, there was another hypodense mass in the medial limb of the right adrenal. The patient underwent a pancreaticoduodenectomy and pathology confirmed the diagnosis. Regional lymph nodes were positive for a poorly-differentiated neuroendocrine carcinoma. The mass located in the right adrenal was also resected and confirmed as adenoma, which was unrelated to the lesion in the pancreas. He also underwent below the knee amputation and pathology confirmed the lesion as a metastatic deposit. He was discharged 6 weeks postoperatively after recovery and was planned for chemotherapy.
5. Discussion
Pancreatic neuroendocrine tumors (PNET), a subgroup of gastroenteropancreatic neuroendocrine tumors, have an incidence of 1/100,000 and represent 1-2% of all pancreatic neoplasms (5, 6). PNETs are classified as functional or non-functional based upon the presence or absence of a specific clinical syndrome relevant to hormone oversecretion. Recently, a higher number of nonfunctional pancreatic neuroendocrine tumors have been detected coinciding with the increased use of cross-sectional imaging. The overall prognosis and long-term survival in PNET are far better than in patients with exocrine pancreatic cancer (7), which has one of the highest mortality rates of any cancer with a 5-year survival of less than 5% (8). Pancreatic cancer is known to metastasize rapidly. Liver and peritoneum are the most common sites of metastases in pancreatic cancer, followed by the lungs, bone, and brain (1). Skeletal muscle metastasis is a rare condition (9) and differentiation between a primary soft tissue sarcoma and metastatic carcinoma is difficult without biopsy (10). Skeletal muscle metastases from pancreatic carcinoma are extremely infrequent. To the best of our knowledge, there have been only a few cases of skeletal muscle metastases from pancreatic carcinoma reported in the English literature. Previously reported involved muscles are the gluteal muscle, psoas, rectus abdominis, pectoralis major, quadriceps femoris, and biceps (Table 1) (11-13). Metastasis to the triceps surae has not been mentioned. Patients with lung and thyroid cancer, melanoma and lymphoma may present with skeletal muscle metastasis, but pancreatic carcinoma is a particularly unusual primary malignancy for this finding (14). Although skeletal muscle comprises nearly 50% of the total body weight, and has an abundant blood supply, metastasis to the skeletal muscle is an infrequent phenomenon (14, 15). The mechanism of skeletal muscle metastasis in patients with pancreatic cancer is not well known, but it may be related to turbulent blood flow, high tissue pressure, contractility, accumulation of lactic acid, pH environment, and oxygenation (9, 16). Diagnosis is confirmed mostly by needle aspiration. However, before pathological diagnosis, radiological assessment of the mass is necessary and often provides useful information with respect to the overall assessment and treatment of this lesion. In comparison with the MR image, plain film and CT are of little value regarding the characterization of the lesions. MRI can be used not only for diagnosis of metastatic tumors, sarcomas, hematomas, and abscesses, but also for delineating the extent of the involved muscles from adjacent muscles on multiplanar sections (17, 18) and accurately determining invasion to the bone and vascular structures. Besides, MR appearance is also helpful in planning the treatment strategy. In particular, contrast-enhanced MRI helps in evaluating the blood supply of the tumor in designing the excision plan of the lesion. In a word, although MR imaging is not a specific imaging tool for skeletal muscle metastasis (19), it is an essential imaging modality for the diagnosis and treatment of soft tissue tumors. As demonstrated by this case, biopsy with histology and immunohistochemistry should be performed for the final diagnosis of the mass and to differentiate primary tumors from metastases. Our CT and MRI findings favored a primary soft tissue tumor. A tissue diagnosis is especially useful in the case of nonspecific imaging findings or atypical clinical features. Skeletal muscle metastases from pancreatic cancer and other malignant tumors do occur and should be considered in cancer patients with soft tissue abnormalities and pain. Biopsy with accompanying histology and immunohistochemistry play a vital role in identifying primary tumor from metastatic disease.