1. Introduction
Diagnostic approaches in cases of pediatric gastrointestinal (GI) bleeding and anemia depend on the patient’s age and individual characteristics. Various etiologies, including anal fissure, intussusception, Meckel’s diverticulum, infection, Henoch-Schonlein purpura, and juvenile polyps, are responsible for GI bleeding in pre-school aged pediatric patients. However, vascular malformation is a rare cause of GI bleeding in pediatric patients (1, 2).
Although several studies have been published regarding vascular malformation of the GI tract (1-4), there are few reports on imaging findings, especially ultrasound (US) and computed tomography (CT) findings of long-segmental vascular malformation of the small bowel. Here, we report a case of vascular malformation manifesting as long-segmental and circumferential bowel wall thickening in a young child who presented with GI bleeding accompanied with anemia.
2. Case Presentation
A three-year-old girl came to our children’s hospital with anemia. She had experienced two episodes of melena after upper respiratory infections, was diagnosed with anemia about 6 months prior to presenting to our hospital, and had recovered after being given iron supplements. She presented to our hospital with no specific abdominal pain, diarrhea, fever, weight loss, or appearance of being ill. Her abdomen was soft and flat with no tenderness.
Her height and weight were in the 50th percentile for her age. An initial complete blood count suggested mild anemia with a hemoglobin of 9.7 g/dL (normal range; 10.2 - 12.7 g/dL), mean corpuscular volume (MCV) of 70.1 fl (72.3 - 85.0), and mean corpuscular hemoglobin concentration (MCHC) of 31.3 g/dL (31.8 - 34.6 g/dL). A biochemical screen revealed no abnormality. A 99mTc-pertechnetate scan showed no ectopic gastric mucosa and gastroduodenal endoscopy was negative for bleeding in the upper GI tract.
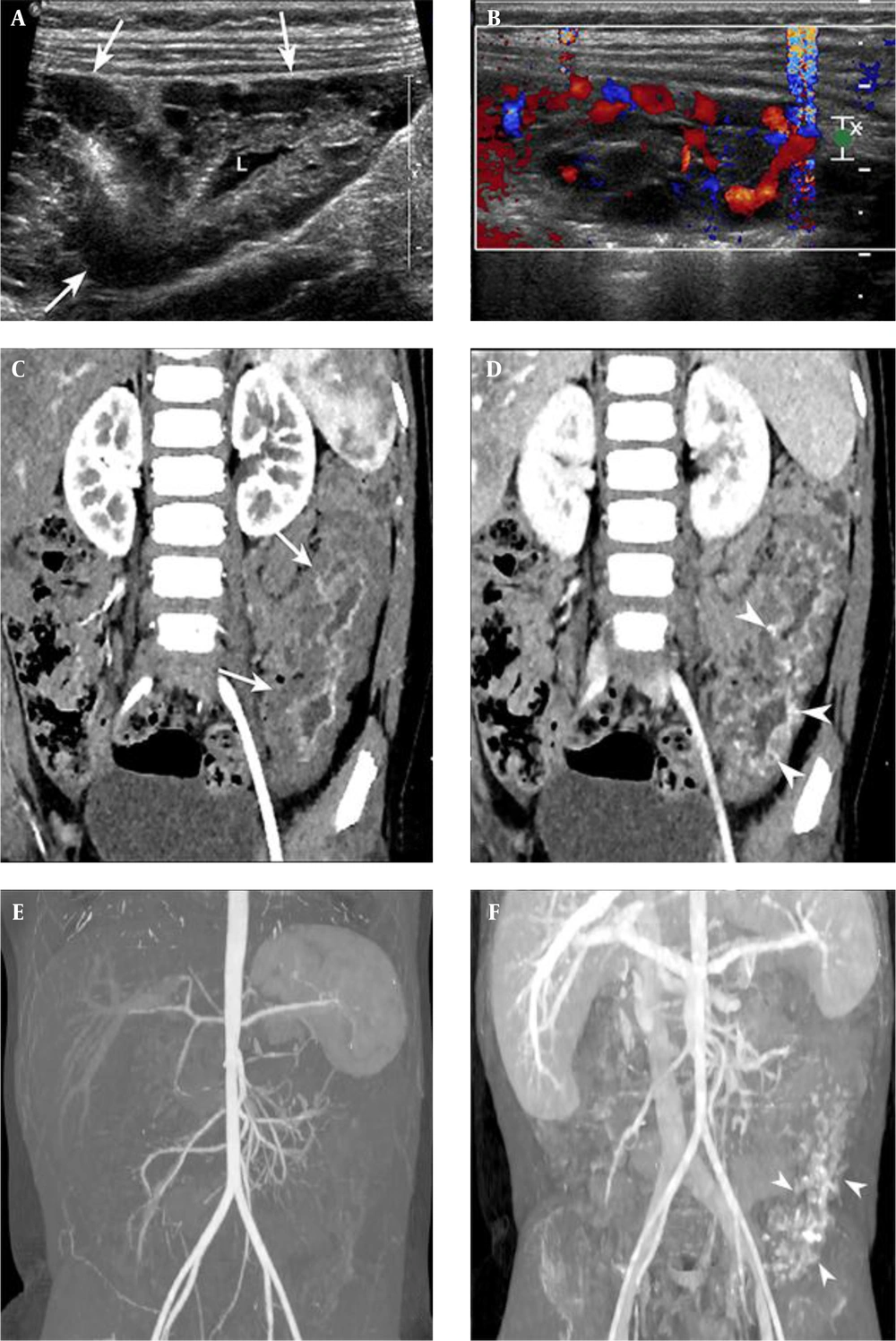
US revealed a 10 cm long segmental and circumferential bowel wall thickening at the left lower quadrant of the abdomen. Numerous cystic locules that did not exceed 1 cm in diameter were identified in the thickened bowel wall (Figure 1A). Several locules showed internal echogenic debris, but the majority contained clear anechoic fluid. The echogenic mucosal lining of the involved bowel segment was well preserved. There was no phlebolith apparent on a 2D grayscale image. On a color Doppler study, the cystic locules did not reveal internal vascular flow. Pseudo-flow artifact was present when the lesion was compressed by the US probe (Figure 1B).
Ultrasonography of the small bowel of a three-year-old girl presenting with anemia. A, 2D gray-scale image depicting bowel wall thickening with multiple cystic locules (arrows). The bowel lumen (L) was patent; B, Pseudo-flow artifact was provoked when the lesion was compressed by a sonographic probe; C, Oblique coronal reformatted arterial phase computed tomography (CT) image showing segmental bowel wall thickening involving the middle layer of the jejunum (arrows); D, Portovenous phase CT image revealing the multifocal contrast-filling spaces at the thickened bowel wall (arrowheads); E and F, CT angiography with maximum intensity projection (MIP) images showing no engorged mesenteric vessel on arterial and portovenous phases. Note the contrast filling spaces on portovenous phase (arrowheads).
CT angiography was performed to determine the extent and location of the lesion and to evaluate the associated vascular abnormality with a suspicion that there might be a vascular malformation. An arterial phase CT image (Figure 1C) showed circumferential, low attenuation bowel wall thickening involving the jejunum, and its extent was more than 10 cm in length. The enhancement of the inner layer of the bowel wall was intact, but the middle layer did not show any enhancement. In portovenous phase CT image (Figure 1D) (Figure 1D), multifocal punctate and curvilinear areas of enhancements were presented in the middle layer of the thickened bowel wall, which showed low attenuation in the arterial phase. No engorged feeding artery or early draining vein was demonstrated on the maximum intensity projection image on the arterial phase (Figure 1E) but enhancement of the thickened bowel segment was appeared on the portovenous phase of CT image (Figure 1F). The bowel lumen was patent with no finding suggesting obstruction.
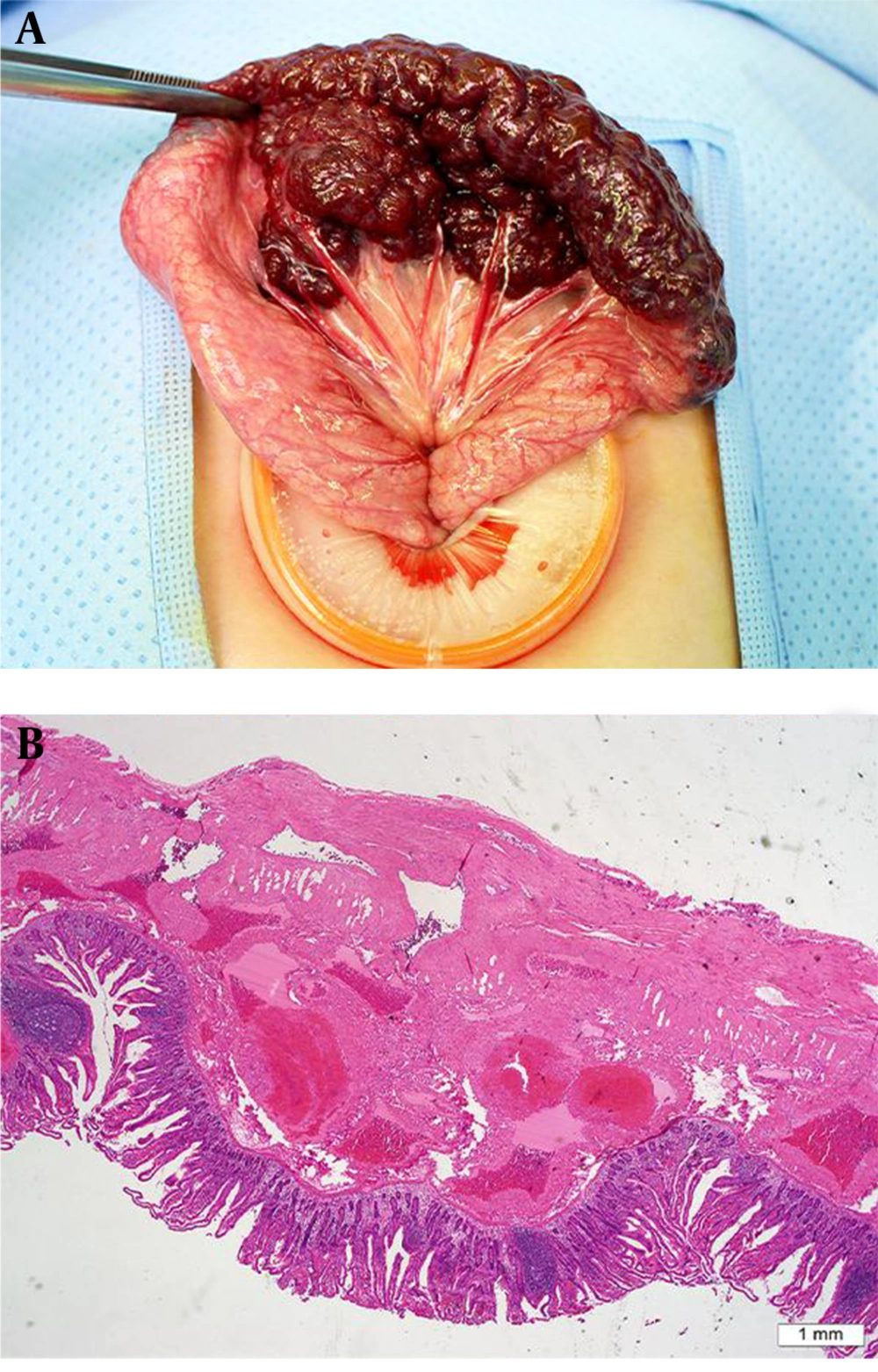
Capsule endoscopy (CE) was performed to evaluate the mucosal lesion, however, the capsule was retained in the stomach for more than 16 hours so that we could not obtain any additional data from CE. Finally, the CE was passed out spontaneously after 30 hours from insertion of it. The patient underwent laparoscopic small bowel resection. Laparoscopic finding showed dark red and purple berrylike lesions covering the small bowel. The mass involved 13 cm of the mid jejunum and the lesion consisted of multiple cystic locules containing old blood (Figure 2A).
Intraoperative photograph and microscopic examination. A, Intraoperative photograph image showing the dark red and purple berry-like vascular malformation encircling the jejunum; B, Hematoxylin and eosin (H&E), 20 × magnified image showing multiple dilated vascular structures of variable sizes and the thickness of the wall affecting the bowel submucosa, muscularis propria, and subserosa.
Microscopic examination revealed variably dilated thick-walled arterial and venous structures involving the submucosa, muscularis propria, and subserosa of the bowel wall. Single or multiple endothelial layers lined the wall of the dilated venous structures (Figure 2B) and these findings were consistent with arteriovenous malformation.
The patient recovered well from the surgery and was discharged uneventfully on postoperative day five. After three months, her general condition was normal and her anemia was fully recovered up to a hemoglobin level of 12 g/dL without medication.
3. Discussion
Our case showed a circumferential and long-segmental vascular malformation involving the small bowel that led to GI bleeding. Vascular anomalies arising from the GI tract causing bleeding are rare in pediatric patients (1, 2). CE is the only diagnostic modality capable of directly visualizing the bleeding focus to evaluate patients with small bowel bleeding. However, capsule retention is a risk of CE, especially in young children. Therefore, diagnosis of small bowel bleeding in children depends largely on imaging modalities. Our patient underwent CE, however, no consecutive image was obtained due to retention of the capsule in the stomach.
In the literature review, several studies have shown cases of vascular malformation involving the small bowel similar to our patient in the imaging, operative, and histologic findings, but the use of terminology is variable, including arteriovenous malformation, vascular malformation, and hemangioma (4-6). The international society for the study of vascular anomalies (ISSVA) classification system divides vascular anomalies into vasoproliferative or vascular neoplasms and vascular malformations (7). Vascular malformations can be subcategorized as capillary, lymphatic, and venous malformation (VM) for slow-flow lesions, and arteriovenous malformations and arteriovenous fistula for high-flow lesions according to the most affected vascular structures and these entities can manifest as either a single lesion or a combined lesion (1, 7, 8). The ISSVA further subcategorized VM as follows; familial VM cutaneous-mucosal, blue rubber bleb nevus (Bean) syndrome VM, glomuvenous malformation, cerebral cavernous malformation, and others (8). It can also associated with Klippel-Trenaunay syndrome, and Proteus syndrome but our patient did not have any sign or symptoms of these syndromes.
Imaging findings of vascular malformations of the GI tract reported previously have focused on CT, magnetic resonance imaging (MRI), and angiography (1, 7, 9). In our patient, CT images showed a long-segmental bowel wall thickening consisted of numerous small cystic channels those was demonstrated on US. Although blood flow was not detected on the color Doppler study, delayed enhancement of the thickened bowel wall was appeared on portovenous phase with no discernible engorged artery or vein on both color Doppler and CT angiography.
Typical CT findings of AVM include highly enhancing lesions with engorged feeding and draining vessels. On MRI scans, AVM may reveal multiple signal-void structures and anomalous vessels. In VM, CT scans may reveal bowel wall thickening of low attenuation with or without phlebolith and MRI may show bowel wall thickening of high signal intensity on T2-weighted images and variable enhancement on post-contrast T1-weighted images. Nuclear scintigraphy can demonstrate blood pooling within the venous channel in the case of VM or bleeding (1). Although the histopathologic diagnosis of our patient was arteriovenous malformation, however, the imaging findings were similar to those of VM rather than arteriovenous malformation. Histopathologic findings revealed that the majority of the vascular abnormalities was involving the venous channels with marked dilatation whereas only few arterioles were mildly dilated. We hypothesized that predominant involvement of the venous channels could lead to the VM-like imaging findings in our patient.
There were few reports about sonographic findings of vascular malformations of GI tracts. However, US was valuable for detecting the location and cystic character of the bowel wall thickening in our patient. Unlike in adult patients, US can be useful for evaluating bowel disease in pediatric patients due to small body and less abdominal fat tissue. US is effective to detect cystic lesions such as duplication cysts, mesenteric cysts, lymphovenous malformations, and bowel wall thickening due to Henoch-Schonlein purpura, infectious enterocolitis, and inflammatory bowel disease. Moreover, US with Doppler study can provide detailed information regarding the vascular flow to help differentiate high-flow from low-flow vascular malformations. A pseudo-flow artifact may be present when cystic or vascular structures are compressed by an US transducer due to motion of the fluid. Although a pseudo-flow artifact can mimic vascular flow, provocation of the pseudo-flow artifact is useful to differentiate cystic lesions such as vascular malformations from hypoechoic solid lesions.
After confirming the extent and location of the lesion, surgery was performed due to persistent anemia and to prevent recurrent melena. Intraoperative sclerotherapy or embolization could be ineffective due to the disease duration (2). Surgical resection may be a curative treatment option when the lesion can be completely removed. Fortunately, in our patient, laparoscopic exploration allowed clear identification of the border of the affected segment; successful and complete resection was done, and pathologic study confirmed a clear resection margin.
In summary, although vascular malformation in GI tract is rare in children, it should be considered when GI bleeding occurs in pediatric patients.