1. Background
After non-beating cardiac operation, such as prosthetic mitral valve replacement (MVR), ischemic reperfusion injury is a frequently encountered situation (1). The timely diagnosis of presence or absence of reperfusion injury after cardiac operation is critical for the patient’s outcome. Besides electrocardiogram (ECG) as a routine means of monitoring, transesophageal echocardiography (TEE) has also been increasingly used to monitor regional myocardial function and myocardial ischemia during cardiac operation (2-4). Previous studies have shown that cyclic variation in myocardial gray level detected by simple videodensitometric analysis can be served as a marker of viability (5). In addition, strain by Doppler is also a sensitive means for detecting myocardial ischemia during coronary bypass operation of the beating heart, also capable of correctly localizing the ischemia (3). However, in our experience, regional strains and strain rates often have a poor repeatability in the spontaneously weak beating heart during the reperfusion process of non-beating cardiac operation. They often reduce, disorder, reverse or disappear especially when cardiac chambers are not completely filled, which make them difficult to identify. In contrast, regional grayscale intensity, velocity, and displacement can be continuously identified, and a good repeatability can be obtained.
2. Objectives
In this study, we investigated whether these parameters can effectively reflect the situation of ST-segment elevation ischemic reperfusion injury (STEIRI) in patients after cardiac operation and which has a higher performance of discrimination between patients with and without STEIRI.
3. Patients and Methods
3.1. Study Population
Our study was approved by the local human research ethics committee and free informed consent was obtained from all the participants. The study population consisted of 53 patients who had undergone the non-beating cardiac operation because of prosthetic mitral valve replacement (MVR): 26 patients with STEIRI (17 females, 9 females; mean age: 56.7±10.5 years, range: 46-67 years) and 27 patients without STEIRI (16 females, 11 males; mean age: 55.4±10.7 years, range: 45-66 years). The diagnosis of STEIRI was based on STEIRI lasting ≥30 minutes, ST-segment elevation ≥2 mm (0.2mV) in at least two contiguous electrocardiography leads (6, 7). The control group consisted of 30 normal participants (17 females, 13 males; mean age: 52.8 ±9.7 years, range: 43-62 years). All the participants had a sinus rhythm, and a normal systolic function of the left and right ventricles. The participants with coronary artery disease, hypertension, congenital heart disease, cardiomyopathy, and diabetes mellitus were excluded from the study.
3.2. Multiplane TEE
TEE examinations were performed in all patients from the spontaneously weak beating to the completely stronger beating in the process of reperfusion after non-beating surgery (about 2 hours after reperfusion).The echocardiographic data were acquired using a commercially available ultrasonic system: Vivid E9 ultrasound machine (GE Healthcare, Horten, Norway) equipped with 6Tc multiplane transducer. Two experienced operators who were blinded to all clinical data and previous readings performed all acquisitions. Data sets were stored digitally for off-line analysis. All values for each parameter were obtained by averaging measurements from five successive cardiac cycles.
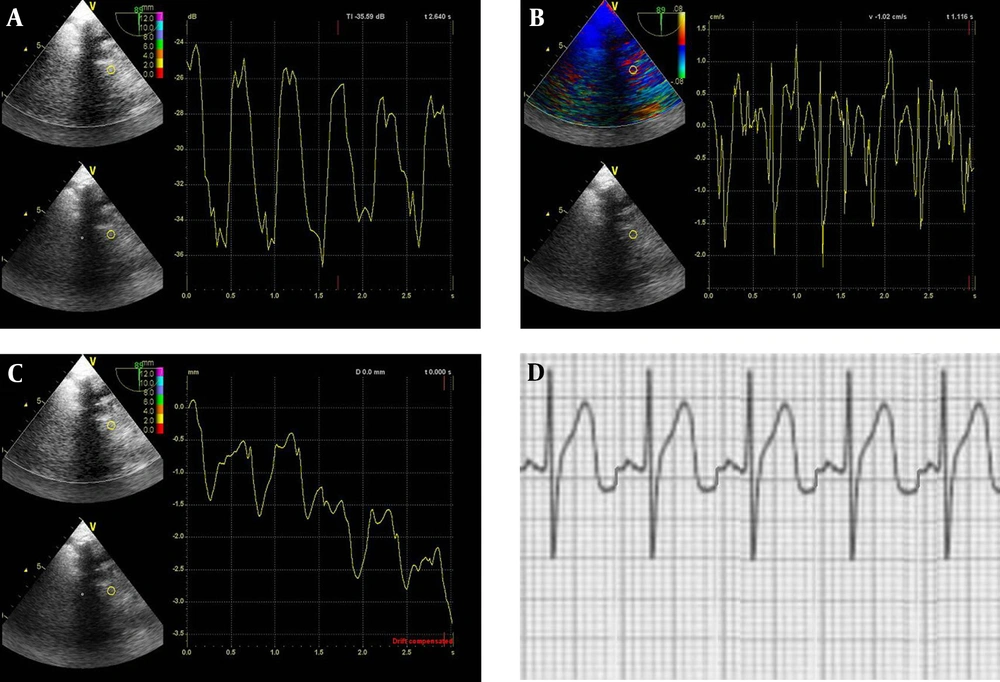
Firstly, esophageal intubation was made in the dorsal decubitus position after general anesthesia with continuous electrocardiographic monitoring, and the two-chamber view (left atrial and left ventricular view) was obtained. Secondly, the tissue velocity imaging (TVI) function was activated and the TVI of five cardiac cycles in this view was stored at a frame rate of 100 frames per second for subsequent analysis. In this process, gains were adjusted at the minimal optimal level to minimize noise, and the filter settings were kept low (50 Hz). Finally, a region of interest (6×6mm) was placed at the lateral side of the mitral annulus for measurements of grayscale intensity (TI), velocity and displacement (D) using videodensitometric analysis, TVI and tissue tracking (TT), respectively. The maximal grayscale intensity in the cardiac cycle [TI (max)], the minimal grayscale intensity in the cardiac cycle [TI (min)], the systolic velocity (Vs), the early diastolic velocity (Ve), the late diastolic velocity (Va) and the peak displacement in the cardiac cycle(D) were obtained (Figure 1 and 2). The difference of the maximal and minimal grayscale intensity in the cardiac cycle [TI (max-min)] and the cyclic variation index of TI [TI (CVI, %) = TI (max - min)/TI (max) × 100] were calculated. All values for each parameter were obtained by averaging measurements from five successive cardiac cycles.
Measurement of the myocardial grayscale intensity, velocity and displacement at the lateral side of the mitral annulus in cases without STEIRI. A. The maximal and minimal grayscale intensity in the cardiac cycle determined by grayscale intensity curve using videodensitometric analysis; B. The peak systolic velocity, the peak early diastolic velocity and the peak late diastolic velocity using tissue velocity imaging; C. The peak displacement determined by displacement curve using tissue tracking; D. Normal ECG
Measurement of the myocardial grayscale intensity, velocity and displacement at the lateral side of the mitral annulus in cases with STEIRI. A. The maximal and minimal grayscale intensity in the cardiac cycle determined by grayscale intensity curve using videodensitometric analysis; B. The peak systolic velocity, the peak early diastolic velocity and the peak late diastolic velocity using tissue velocity imaging; C. The peak displacement determined by displacement curve using tissue tracking; D. Representative ECG of reperfused ST-segment elevations
3.3.Reproducibility
Intraobserver variability was assessed in 15 subjects by repeating the measurements on two occasions (5 days apart) under the same basal conditions. To test the interobserver variability, the measurements were performed on the same subject by a second blinded observer. Variability was calculated as the mean percentage error, derived as the difference between the two sets of measurements, and divided by the mean observations.
3.4.Statistical Analysis
Data were expressed as the mean ± SD. The differences between the two groups were tested using an unpaired two tailed t test. A receiver operating characteristic curve (ROC) analysis was used to evaluate the performance of discrimination between patients with and without STEIRI. Discriminant analysis was conducted in the traditional manner. A P-value lower than 0.05 was considered statistically significant. Statistical analysis was performed with SPSS version 13 software for Windows (SPSS Inc, Chicago, IL).
4. Results
4.1. Echocardiographic Parameters
As shown in Table 1, TI (max-min), TI (CVI), Vs, Ve, Va and D in patients with STEIRI were all significantly smaller than those of normal participants and patients without STEIRI. There were no significant differences among the patients with or without STEIRI and normal participants according to TI (max) and TI (min). In addition, compared with normal participants, the patients without STEIRI had significantly smaller TI (max-min), TI (CVI), Ve, Va and D except Vs.
| Parameter | Normal Participants | Patients Without STEIRI | Patients With STEIRI |
|---|---|---|---|
| TI (max,dB) | -37.147±4.41 | -34.66±5.13 | -32.92±3.59 |
| TI (min,dB) | -20.49 ± 1.59 | -21.03 ± 1.34 | -23.75 ± 2.32 |
| TI (max-min,dB) | 16.66 ± 0.49 | 13.62 ± 6.11 a | 9.18 ± 2.09a,b |
| TI (CVI, %) | 44.85 ± 6.35 | 39.27 ± 5.35 a | 28.59 ± 4.52b,c |
| Vs (cm/s) | -6.16 ± 1.91 | -4.93 ± 1.38 | -2.74 ± 1.36 b,c |
| Ve (cm/s) | 6.13 ± 1.78 | 1.73 ± 0.61 c | 1.24 ± 0.59 b,c |
| Va (cm/s) | 6.30 ± 2.13 | 2.82 ± 1. 31 c | 1.34 ± 0. 73 b,d |
| D (mm) | -12.33 ± 0.51 | -4.53 ± 3.25 c | -1.81 ± 1.43b,d |
The Myocardial Grayscale Intensity, Velocity And Displacement aAt tThe Lateral Side oOf tThe Mitral Annulus Among Normal Participants aAnd Patients WWith oOr Without STt-Segment Elevation Ischemic Reperfusion Injury (STEIRI) Wwithin 2 Hhours Aafter Rreperfusion
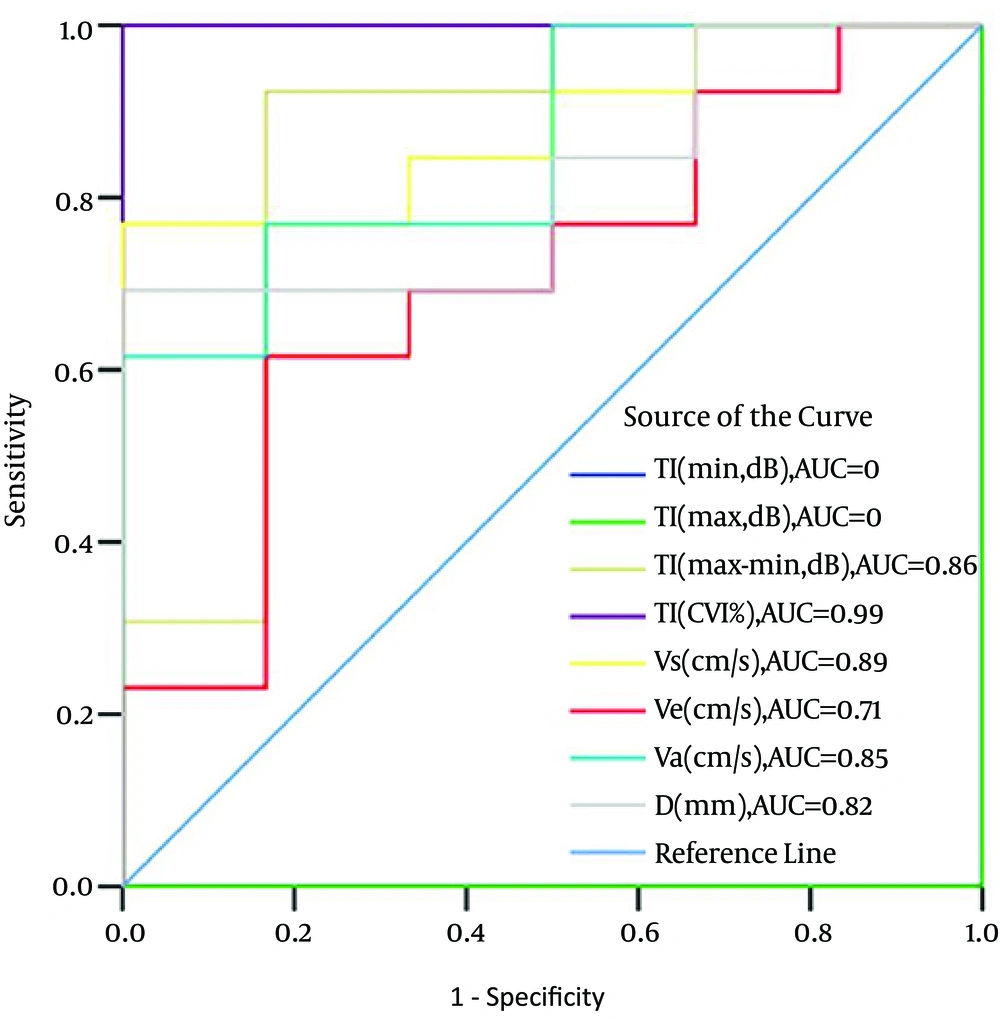
4.2. ROC Analysis
As all known, the area under the ROC (AUC) is positively correlated with the discrimination performance. The performance of the myocardial grayscale intensity, velocity and displacement at the lateral side of the mitral annulus in discrimination between patients with STEIRI and without it are shown in Figure 3. The AUCs for TI (max) and TI (min) in distinguishing patients with STEIRI or without it were 0, which meaned TI (max) and TI (min) had no performance in distinguishing patients with STEIRI or without it, while they were 0.86 for TI (max-min), 0.99 for TI (CVI), 0.89 for Vs, 0.71 for Ve, 0.85 for Va and 0.82 for D. The AUC for TI (CVI) was maximum, which was significantly higher than those of other parameters (P < 0.05).
4.3. Discriminant Analysis
Based on the results mentioned above, discriminant analysis of TI (CVI) for predicting a situation of STEIRI was conducted. The best cutoff value was a TI (CVI) of less than 34.45% (sensitivity, 94.74%; specificity, 97.05%; positive predictive value, 94.74%; negative predictive value, 97.05% and accuracy, 96.22%; P<0.001).
4.4. Reproducibility
Intraobserver and interobserver variability for TI (max) and TI (min) ranged from 2.2% to 2.9%. Intraobserver and interobserver variability for Vs were 2.5% ± 0.9% and 2.9% ± 1.2%, respectively. Intraobserver and interobserver variability for Ve were 1.7% ± 1.1% and 1.8% ± 1.4%, respectively. Intraobserver and interobserver variability for Va were 2.5% ± 1.9% and 2.1% ± 2.4%, respectively. Intraobserver and interobserver variability for D were 1.5% ± 0.7% and 1.7% ± 0.6%, respectively.
5. Discussion
The results presented here indicate that myocardial grayscale intensity, velocity and displacement can effectively reflect the situation of STEIRI in patients after cardiac operation, and TI(CVI) has a higher performance in discrimination between patients with and without STEIRI.
It is important to monitor myocardial function and detect myocardial ischemia during cardiac operation. With the advances in digital signal processing, the clinical used equipment have allowed myocardial tissue characterization (MTC) to integrate other technical parameters, such as motion and deformation, velocity, displacement and strain, for better assessment of myocardial structure and function(8). Usually, MTC may provide two types of information: one is static and consists of the absolute myocardial echo intensity that reflects the ultrastructural myocardial changes in different diseases; the other is dynamic and is related to the variations of echo intensity during the cardiac cycle that is linked to the intrinsic myocardial contractility(9-12). For the first one, there are currently no uniform standards for discrimination of different diseases because of many factors, such as the different instruments used, the different attenuation effects, the different references chosen, and the different sites. In this study, our findings confirm this conclusion. For example, there were no significant differences between the patients with STEIRI and normal participants according to TI (max) and TI (min). For the second one, it seems to be feasible to identify the characteristics of various diseases, due to its relative independence, objectivity and fewer factors. Our results showed that TI (max-min), TI (CVI) in patients with STEIRI were significantly smaller than those of patients without STEIRI, which indicated a different myocardial characteristics between patients with STEIRI and those without it. In other words, TI (max-min) and TI (CVI) can be used to distinguish patients with STEIRI from those without it.
TVI and its implementation, including tissue tracking and strain rate imaging, based on post-processing conversion of regional velocities in local myocardial displacement, deformation rate (strain rate) and percent deformation (strain), have been applied to stress echocardiography for quantitative evaluation of regional myocardial function and detection of ischemia and viability (13, 14). Our results showed that Vs, Ve, Va and D in patients with STEIRI were all significantly smaller than those of patients without STEIRI. In other words, Vs, Ve, Va and D can also be used to distinguish patients with STEIRI from those without.
Then, to find which parameter has the highest performance in discriminating between patients with and without STEIRI, we made a ROC analysis. Our results showed that the AUC for TI(CVI) was maximum, which was significantly higher than those of other parameters. This indicates that TI(CVI) has the highest performance in discriminating between patients with and without STEIRI. The best cutoff value for this variable was TI (CVI) of less than 34.45%, with a higher sensitivity, specificity and accuracy.
In our study, we chose the lateral MV annulus as the region of interest because this position is correlated with the function of the left ventricle, it was easy to identify and was less vulnerable to the impact of adjacent tissues in the process of reperfusion after non-beating surgery especially when cardiac chambers are not completely filled, while other myocardial segments did not have these advantages.
Our study had limitations. First, the grayscale intensity of echocardiographic images is dependent on the pre- and post-processing of raw radiofrequency signals, and it is difficult to obtain the raw radiofrequency signals. Second, a region of interest was not always accurately placed at the lateral side of the mitral annulus, because when the initial spontaneous myocardial motility occurred, cardiac chambers were not yet completely filled that resulted in blurred endocardial borders. Third, the number of participants was limited. New data need to be collected in subsequent studies. Fourth, we did not make a comparison between grayscale intensity and strain rate for the detection of myocardial ischemia. Even so, grayscale intensity, being simple, rapid, and reliable in the assessment of STEIRI, has potential for clinical implications.
In this study, we assessed and compared the performance of TEE acquisition of regional grayscale intensity, velocity, and displacement at the lateral side of the mitral annulus after non-beating cardiac operation in discrimination between ST-segment elevation ischemic reperfusion injury and without it. We demonstrate that the cyclic variation index of grayscale intensity has a higher performance in discrimination between patients with and without ST-segment elevation ischemic reperfusion injury. Although some limitations have been mentioned above, this method still holds considerable clinical promise for the early detection of ischemic reperfusion injury, especially when the representative ECG is not obtained.