Sparganosis is a rare parasite infection which is usually found in East Asia (
1). To the best of our knowledge, 41 cases of breast sparganosis have been reported based on the Korean Medical (KM) database and the Korean association of medical journal editors (KAMJE) database, which were searched to include information from Korean journals that are not included in international citation indexes (
3,
4,
6,
8-
12). It is transmitted by drinking larvae-containing water or consuming raw secondary intermediate hosts such as frogs or snakes (
2). Spargana can move to any site of the body such as the abdomen, scrotum, chest wall, lower extremities, brain, and rarely involve the breast (
3,
4). Sparganosis most commonly manifests as a migrating palpable soft tissue nodule without evidence of inflammatory reaction such as warm sensation or painful swelling (
4). Thus, it may be confused with malignancy and accurate preoperative diagnosis can be difficult in many cases.
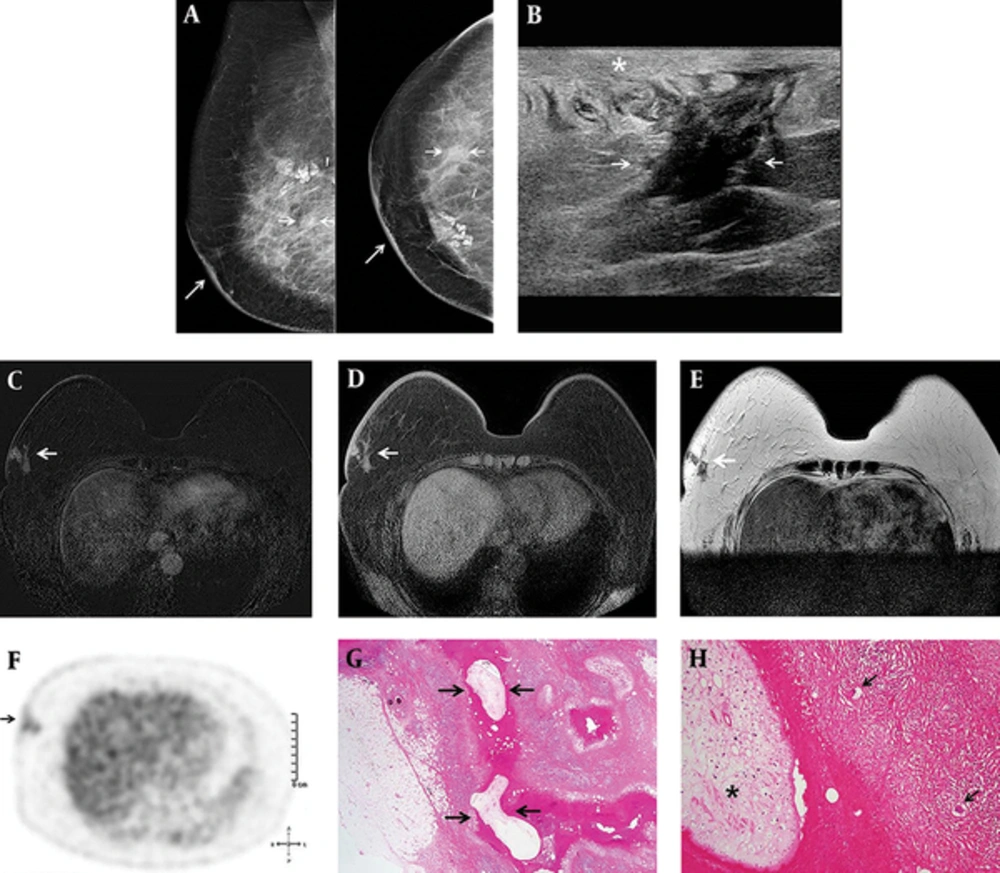
Although breast sparganosis is extremely rare, previously reported cases have described its characteristic radiological findings (
1,
3,
4,
6,
8-
10). The mammographic findings of the breast sparganosis are irregular, lobular or tubular structures or amorphous solid masses (
3,
4,
6,
9,
10). The ultrasonographic findings of breast sparganosis are elongated, folded, tubular hypoechoic structures and serpiginous echogenic lesions, which correlate with pathologic findings (
1,
3,
4,
6,
8-
10). Hypoechoic tubular structures on ultrasonography correlate with the empty tract left by migration of the larvae; whereas, echogenic lesions correspond to living worms or debris (
8). Thus, the diagnosis of breast sparganosis is not difficult if radiologists are familiar with these unique ultrasonographic features. Our case did not represent these typical mammographic and ultrasonographic features of breast sparganosis and mimicked breast malignancy, except for subcutaneous location and thickening of overlying skin that can be helpful for distinguishing breast spargnosis from breast malignancy.
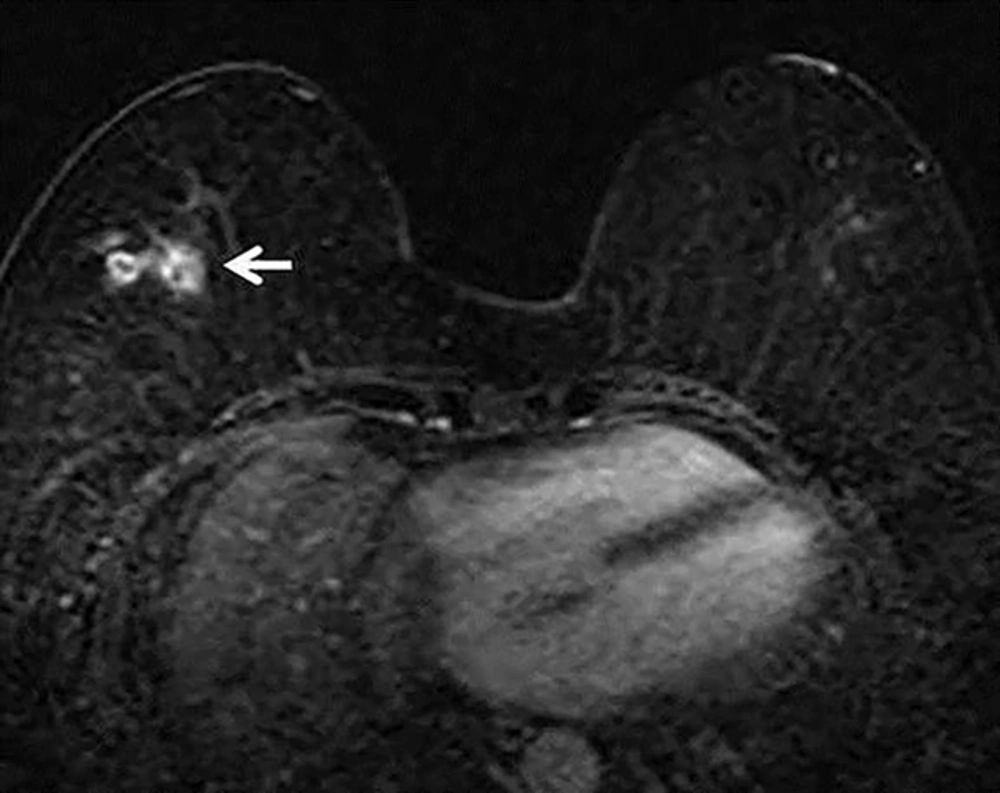
To the best of our knowledge, only two case reports describing DCE-MRI findings of the breast sparganosis have been reported (
11,
12). In these previous reports, the MRI appearance of breast sparganosis includes multiple round and elongated tubular-shaped lesions showing rim enhancement with persistent kinetic patterns in the subcutaneous fat on post-contrast T1 weighted images. We also observed similar MRI findings of breast sparganosis in our case. Although the internal characteristic of rim enhancement on DCE-MRI has a 79% - 92% positive predictive value for breast cancer (
13), benign conditions such as an inflamed cyst, fat necrosis or abscess can also cause rim enhancement. In this case, inflammation of the surrounding subcutaneous fat tissue induced by migrating worms generated various enhancing patterns on DCE-MRI. Only two case reports have described PET/CT findings of breast sparganosis. Ha et al. (
11) reported a case of breast sparganosis incidentally detected by PET/CT with a focal FDG uptake and a standardized uptake values (SUV) max of 1.2, which was not as high as malignancy or active inflammation. However, a case of axillary sparganosis reported by Lee et al. (
7) showed a hypermetabolic mass with a SUVmax of 7.2. Our case showed mildly increased FDG uptake with a SUVmax of 2.1. Ha et al. (
11), Lee et al. (
7), and our case showed different degrees of FDG uptake on PET/CT. The degree of 18FDG uptake on PET/CT seems to have a broad spectrum according to parasite type, infection site, host immune status, and inflammatory response.
Although the radiological findings of breast sparganosis have been established, diagnosis can be confusing, particularly in cases of newly diagnosed breast cancer or patients who have undergone treatment for breast cancer (
7,
12). Lee et al. (
12) reported a case of breast sparganosis in a patient with coexisting ipsilateral breast cancer, which mimicked a multicentric malignancy. Recently, Lee et al. (
7) also reported a case of multiple axillary lymph node sparganosis that presented as a newly developed axillary mass on follow-up MRI and PET/CT during neoadjuvant chemotherapy. In this case, the main breast cancer lesion exhibited a partial response to chemotherapy, but newly detected axillary lymph node sparganosis on MRI and PET/CT was mistaken for a new, axillary lymph node metastasis of the breast cancer, resulting in a treatment change from chemotherapy to surgery. Correlation with clinical history, the presence of inflammatory symptoms, lesion location, and US and MRI features can be helpful for differential diagnosis. In particular, we think that a subcutaneous location and migration of the lesion on follow-up imaging studies are key distinguishing features of sparganosis in the treated breast.
When sparganosis is suspected, complete surgical removal is the treatment of choice rather than percutaneous biopsy procedures, including core needle biopsy or vacuum-assisted biopsy (
4). Percutaneous biopsy procedures may result in worm fragmentation, preventing complete surgical removal and possibly promoting recurrence.
In conclusion, breast sparganosis is a rare parasite infection that may mimic malignancy both clinically and radiologically. Therefore, diagnosis can be difficult, particularly in breast cancer patients. It should be included in the differential diagnosis for incidental breast masses in patients with a history of BCS. A subcutaneous, migrating breast mass with characteristic US and MRI features is distinctive in differentiating breast sparganosis from breast malignancy.