1. Background
In recent years, numerous studies have been conducted on brain tumors. The cerebellar tumors are often known as infratentorial or posterior fossa tumors (1). Posterior fossa tumors constitute 75% of all pediatric brain tumors (1-3). Pilocytic astrocytoma, a low grade glial tumor, is the most common pediatric central nervous system (CNS) tumor, accounting for about 35% of all diagnoses (4, 5). Infratentorial ependymoma includes 15% of posterior fossa tumors in children and is more common in males than females (6, 7). Atypical teratoid rhabdoid tumors (ATRTs) are infrequent and highly aggressive CNS tumors that primarily occur in infants less than 3 years of age (8, 9). Medulloblastoma (MB) is a common malignant ectodermal tumor accounting for 7 to 15% of all brain tumors (10, 11). Based on World Health Organization (WHO) classification, medulloblastoma is subdivided into five major groups: classic, anaplastic, large cell, desmoplastic, and extensive nodularity subtypes (12). The mean overall incidence of MB is estimated at approximately six children per million and the survival rate is 50% to 65% for 5-years (10-12). Treatment strategies for this malignant neoplasm include a combination of surgery, chemotherapy, and following radiotherapy (13).
Recently, quantitative techniques, such as computed tomography (CT) scans or magnetic resonance imaging (MRI), is developing for effective volume measuring of a structure with arbitrary shape and size (14). MRI is the best imaging modality in the diagnosis of intracranial tumors due to its high soft tissue contrast and multiplane capability. On the other hand, under a proper sampling design, stereological methods may be applied to estimate geometric parameters of any object (14, 15). For example, the Cavalieri method is used in combination with point counting to obtain an assumption-free estimate of the total volume of the structure of interest (16). The method is delivered by the standard software can be tested and so assessed with Cavalieri methods for brain volume estimation. Liu et al. (2007), Ertl-Wagner et al. (2009) and Gong et al. (2004) evaluated the tumor volume using MRI and stereological methods (16-18).
2. Objectives
In the current study, we evaluated and compared the cerebellum and tumor volume in patients suffering from meduloblastoma tumor, using MRI and Cavalieri stereological method.
3. Patients and Methods
3.1. Patients
The current study was performed between December 2015 and August 2018. The study was carried out on 16 subjects (4 - 7 years old) consisting of two groups of eight people (five female and three male) including patients (with medulloblastoma tumor) and control (five female and three male) with a mean age of 5.7 years. Diagnosis of all patients was based on pathology and imaging findings. Metastatic and vascular tumors were eliminated from the study. In this regard, WHO criteria for pathological diagnosis was used. All the participants’ parents signed the consent forms.
3.2. MRI Procedure
An MRI was performed hours before surgery. An identical MRI was obtained from the patients 2 weeks after surgery. In the pre- and post-operation time, contrast sagittal and axial T1-weighted imaging as well as T2 weighted MR sectioning and fluid-attenuated inversion recovery (FLAIR) sequence imaging were performed for all patients using a 1.5 Tesla MR machine (Philips, Intera, The Netherlands). The following parameters were used for the T1 imaging slices: 2 signal averaging number, repetition time/echo time (TR/TE): 25/5, field of view (FOV): 130 × 160, 0.7 mm slice thickness (ST) with no interval between the sections, and 224 × 224 matrix.
3.3. Cavalieri Principle Applied to MRI Sections
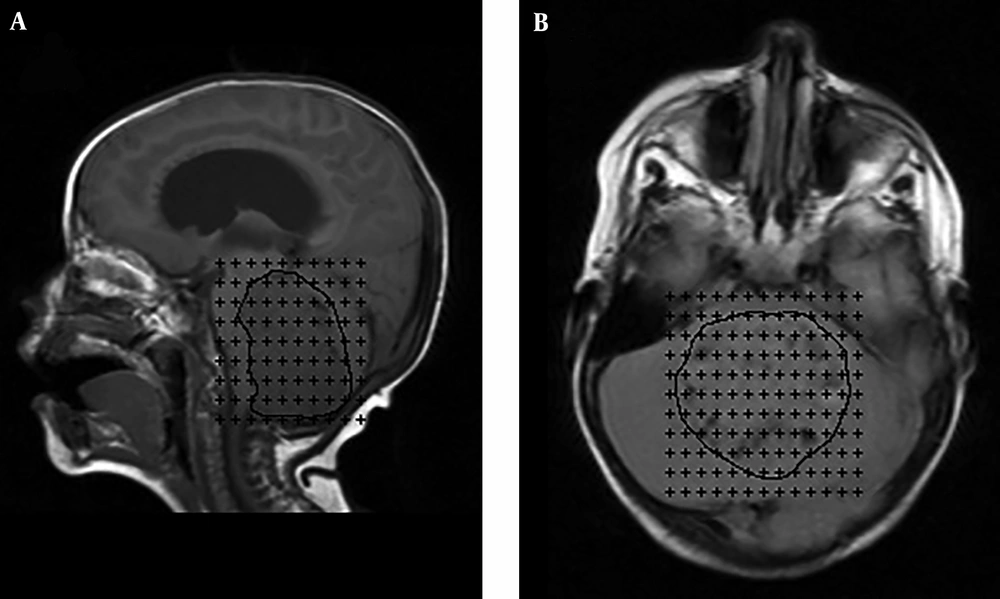
Stereological methods, such as Cavalieri method, were used to obtain an unbiased volume estimation of a structure of arbitrary shape and size effectively. According to Cavalieri principle, the MRIs of a section series with a thickness of 0.7 mm were used to estimate the structures and tumor volume. The square grid test system with d = 0.4 cm between test points was superimposed on MRI films (Figure 1). The points hitting the structure sectioned surface area were counted for each section and the volume of the cerebellum and MB tumor were estimated, using the following formula (Equation 1):
Sagittal (A) and axial (B) MR scans with a point-counting grid overlaid on images for the estimation of tumor volume using the Cavalieri technique. The border of the tumor is determined with a black line. Each point (+) is associated with a known area (a/p). (a/p; represents the area of each point on the point counting grid)
where t is the section thickness, a/p represents the area of each point on the point counting grid, ∑P is the total number of the points hitting the section’s cut surface, and ½ Pmax is half of the maximal number of points counted on the largest section of the examined subject (14, 15).
3.4. Statistical Analysis
The volume of the interested structures was compared between the patients and normal subjects using Mann-Whitney U test. The volume in intervention group during pre and post of surgery were compared by Wilcoxon signed-rank test. The volume data are expressed in mean ± standard deviation (SD). All P values < 0.05 were measured as statistically significant. All statistical analysis were performed using GraphPad prism software (version 8) and SPSS software (IBM Corp. Released 2013. IBM SPSS Statistics for Windows, Version 22.0. Armonk, NY: IBM Corp.).
4. Results
The volume of the brain and cerebellum in both axial and sagittal MR planes in the normal and patient groups are shown in Table 1. Cerebellar volume was significantly increased in patients compared to healthy controls. Post-operative cerebellar volume did not show a significant difference with the controls and was very close to the normal values.
| Parameters | Normal group | Patient group | |||
|---|---|---|---|---|---|
| Pre operation | P value | Post operation | P value | ||
| Brain volume, cm3 (axial MR images) | 1350.69 ± 158.49 | 1475.62 ± 177.17 | 0.645 | 1282.52 ± 157.09 | 0.645 |
| Brain volume, cm3 (sagittal MR images) | 1478.41 ± 147.64 | 1471.14 ± 150.17 | 0.936 | 1273.91 ± 170.37 | 0.367 |
| Cerebellar volume, cm3 (axial MR images) | 158.09 ± 20.08 | 236.81 ± 27.82 | 0.037* | 161.74 ± 22.22 | 0.959 |
| Cerebellar volume, cm3 (sagittal MR images) | 149.54 ± 19.90 | 207.64 ± 18.64 | 0.028* | 172.13 ± 20.28 | 0.505 |
The volume of the brain, cerebellum, and MB tumor in both axial and sagittal MR planes in the pre- and post- operation situation in the patient group are shown in Table 2.
| Parameters | Patient group | ||
|---|---|---|---|
| Pre operation | Post operation | P value | |
| Brain volume, cm3 (axial MR images) | 1475.62 ± 177.17 | 1282.52 ± 157.09 | 0.505 |
| Brain volume, cm3 (sagittal MR images) | 1471.14 ± 150.17 | 1273.91 ± 170.37 | 0.328 |
| Cerebellar volume, cm3 (axial MR images) | 236.81 ± 27.82 | 161.74 ± 22.22 | 0.049b |
| Cerebellar volume, cm3 (sagittal MR images) | 207.64 ± 18.64 | 172.13 ± 20.28 | 0.130 |
| Tumor volume, cm3 (axial MR images) | 62.76 ± 18.13 | 25.53 ± 7.16 | 0.046b |
| Tumor volume, cm3 (sagittal MR images) | 63.91 ± 18.71 | 19.51 ± 5.22 | 0.028b |
Volume of the Brain, Cerebellum, and Medulloblastoma Tumor Represented in Both Axial and Sagittal MR Planes in the Pre- and Post- Operation Sessions in the Patient Groupa
The volume of the MB tumor in both axial and sagittal MR planes showed significant decreases in the post- operation time compared to pre-operation value. Furthermore, the volume of the cerebellum in axial MR planes showed significant reduction after surgery (P < 0.049). The result showed that the post-operative cerebellar volume in sagittal MR planes reduced compared with pre-operative volume, but this change was not statistically significant.
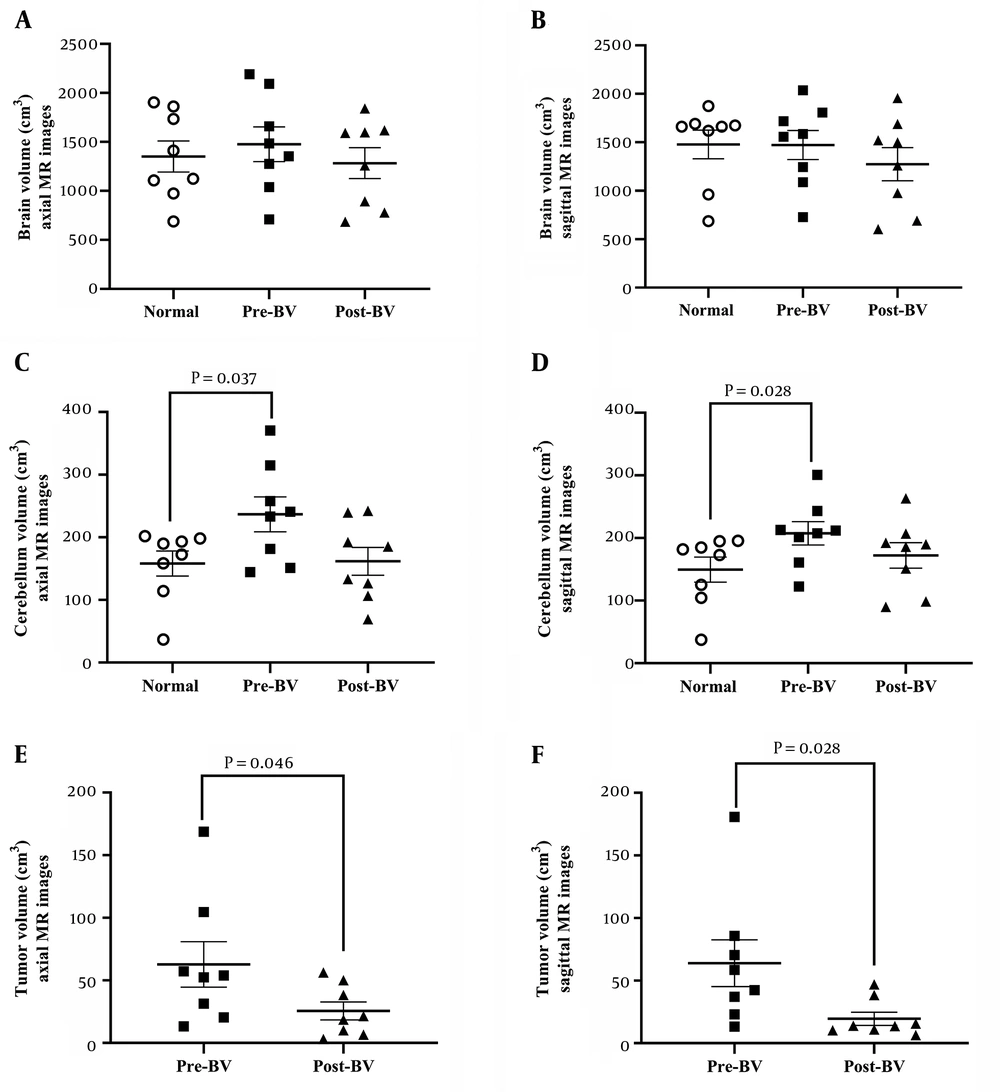
The data analysis of imaging is presented in Figure 2. The post-operative volume of the MB tumor was statistically reduced by more than 90% in both axial (P = 0.046) and sagittal (P = 0.028) planes in comparison to the pre-operative volume. The pre-operative volume of the cerebellum in axial/sagittal planes increased by 38% in patients compared to the normal subjects.
The volumes of the brain, cerebellum, and medulloblastoma tumor are represented in both axial and sagittal MR planes. Dot plots show the brain volume in the axial and sagittal planes (A and B), cerebellar volume in the axial and sagittal planes (C and D), and tumor volume in axial and sagittal planes (E and F) in the experimental groups. Each dot represents one subject. (BV: brain volume, CV: cerebellar volume, and TV: tumor volume).
5. Discussion
Primary tumor volume is an important prognostic factor that affects survival compared to the stages of the tumor (19). Some studies reported that calculation of the tumor volume with pre-catchment imaging methods may be useful to assess the surgical treatment options, and response of the tumor to the treatments such as radiation therapy, affects the anatomical structure of the brain (20, 21). Tumor volume is also used to determine tumor location, relationship with adjacent structures and its directions (22).
Volumetric measurement of the tumor area in magnetic resonance imaging (MRI) is clinically important for diagnosis and treatment assessment in patients with brain tumors.
Moreover, stereology is used to calculate unbiased properties of structures existing in three dimensions (3D) from two-dimensional (2D) medical images. An unbiased estimate of organ volume can be obtained using the Cavalieri principle, considered a stereological method (23).
In fact, this method has the following advantages for the researcher: (1) The structure under study needs no preconditioning since the Cavalieri method is a design-based method but not a model based approach; (2) The actual properties of the structure, such as section thickness, are taken into consideration; and (3) The sampling or estimating system can be easily modified to obtain an appropriate coefficient of variation. Furthermore, the reliability and efficiency of the Cavalieri method for volume determination have been confirmed frequently (24).
Applying the Cavalieri principle in studies aimed at obtaining quantitative data on irregularly shaped three-dimensional objects suggests benefits such as consequential quantitative data, application of strict sampling procedures, easily reproducible data, and a well-established theoretical background, making the reliability of the data easy for examination (25-27). Therefore, this method accompanied with MR imaging data can be potentially applied as a simple, reliable and quantitative method for diagnosis and follow-up of the treatment options in patients with brain tumors.
There has been growing recent interest in quantitative techniques for measuring volume using CT scans or MRI. Some proposed geometric methods for assessing the volumetric properties of radiological images based on direct geometric dimensions such as length, diameter, or the largest diameter measurements (28, 29). In such methods, it has been presumed that the structure under investigation has an ellipsoidal, spherical, or known geometric shape. However, the assumption of such smooth geometric properties in biological tissues is not always correct (23). In addition, the other techniques such as computer-based 3D volumetric reconstructions, and planimetry methods, are also used for volume estimations (30-33). The planimetric methodologies give more precise and accurate results compared to the above-mentioned geometric techniques (28, 30, 31).
However, several studies showed that there are also assumptions in the planimetric methods resulting in volume measurement over-estimation and some degree of systematic bias (28, 29, 34, 35). In addition, the computerized reconstructions generally require quite expensive equipment and trained personnel for their routine application, and these features make these applications unsuitable for most clinics or research centers (36, 37).
Point counting techniques represent a more reliable and efficient approach than a planimetric methods (38-42); therefore, results obtained through point counting will be a direct and assumption-free estimate of the total volume of the interest structure, and hence very valuable in monitoring brain tumors.
In fact, the volume of any structure may be considered using histological sections, CT or MR images through Cavalieri method (22). Other advantages of this method are cost and time efficiency (43).
Results showed that the tumor volumes in axial/sagittal sections after surgery were significantly lower than those before surgery. In pediatric population, the spread of residual surgical resection has been shown to influence the survival outcomes. Excision of more than 90% of the tumor is correlated with improved survival rate in children older than three years (44). In our study, in four patients, more than 90% of the tumor was removed, suggesting an increase in survival rate. The results of the present study also showed differences in the volume of the cerebellum between the control and patient groups before surgery. According to this result, the cerebellum volume in axial and sagittal MRI images in patients with medulloblastoma tumor were significantly higher compared to that of the control group. This agreement with structural MR scanning studies revealed that medulloblastoma tumor can cause bilateral olivary hypertrophy in the human cerebellum which is known as hypertrophic olivary degeneration (HOD) (45).
A common problem in the stereological method is an over-projection or under-projection effect, an artifact that results from section thickness. This problem also exists when MRI or CT scans are used for volume estimations (23, 24, 46). There are several solutions for management of the under-projection or over-projection effects of radiological imaging (47, 48). A previous study showed that the approach formulized as Equation 1 eliminates possible over-estimation due to over-projection (24). Therefore, we used this method in the present study.
Some brain tumors such as MB do not show a smooth demarcation with brain on MRI or CT scans. Therefore, it is almost impossible to obtain reliable quantitative data on tumor size using the diameter or length of the tumor on MRI. However, the surgeon needs the exact volume of the tumor for comparison of the pre-operative and post-operative scans (28).
During the operation, the whole tumor mass cannot be resected totally to avoid excision of the pathological structures with the surrounding healthy tissues. Thus, assessment of the exact tumor volume not only provides important data for the assessment of the size of the structures to be excised during the operation but also allows treatment planning and post-operative follow up (28, 34, 49).
In addition, Cavalieri method using automated software is currently available and could be performed easily and rapidly. Stereological measurement can therefore provide additional useful data to supplement MR measurements, especially in borderline and controversial cases.
It could be concluded that the combination of MRI and the Cavalieri principle could estimate tumor volume that may be useful in evaluating the efficiency of surgical treatment and prognostication of tumor regression rate. The presented method does not require any change in routine procedures and can be performed on any complete set of MRI scans. The method can be efficiently used without any need for additional equipment and expert personnel that is required for routine MRI.