1. Background
The pyramidal lobe (PL) is an anatomic variation of the thyroid gland observed as an incidental finding on ultrasonographic (US) examination. This lobe occurs when the caudal end of the thyroglossal duct differentiates into thyroid tissue (1). Many anatomic and morphological variations of the PL have been described, mostly based on the appearance, position, size, and shape (1). The PL may be susceptible to the same pathologies as the rest of the thyroid tissue; therefore, it is necessary to be informed of this common variation, as it may be a source of residual diseases in patients who previously have had thyroidectomy (2).
The frequency of the PL in adults has been reported to vary from 15 to 75% in cadaveric, surgical, radiological, and scintigraphic series, but to the best of our knowledge, no previous study has demonstrated the prevalence and features of the PL in infants and children.
2. Objectives
The objective of this research was to evaluate the prevalence, anatomic, and morphological features of the PL of pediatric participants on thyroid US.
3. Patients and Methods
3.1. Patients
The Institutional Clinical Research and Ethics Committee approved this study. The descriptive cross-sectional study design was used. The present study analyzed 355 pediatric patients who underwent thyroid ultrasound examination between November 2018 and January 2020 at the University Hospital in Izmir.
The inclusion criteria for this retrospective study were as follows: (1) subjects under 18 years of age; (2) thyroid gland located in the normal position; (3) homogenous echogenicity of the thyroid parenchyma on US; and (4) no previous thyroid surgery. The following exclusion criteria were applied: (1) subjects over 18 years of age; (2) evidence of congenital thyroid disorders; (3) previous thyroidectomy; and (4) poor ultrasound image quality for thyroid evaluation. Patients with congenital absence of the thyroid gland [thyroid hemiagenesis (n = 2) and thyroid agenesis (n = 3)] were excluded from the study. Patients who underwent thyroidectomy previously (n = 3) were excluded from this research as well. Of the 347 patients enrolled, 22 were excluded since their ultrasound image quality was significantly compromised by patient movements.
A total of 325 subjects aged between 0 and 18 years (mean age, 10.95 years ± 4.92) were finally enrolled. The participants were classified into six age groups (Table 1). The age groups were chosen according to the definition in medical subject headings (MeSH) vocabulary (i.e., infants, toddlers, preschool children, school children, and adolescents).
| Groups (%) | Age (mo) | Girls (n) | Boys (n) | Total (n) |
|---|---|---|---|---|
| Group 1 (5.5) | 0 - 12 | 5 | 13 | 18 |
| Group 2 (4) | 13 - 36 | 5 | 8 | 13 |
| Group 3 (10.5) | 37 - 72 | 18 | 16 | 34 |
| Group 4 (17.2) | 73 - 120 | 32 | 24 | 56 |
| Group 5 (33.8) | 121 - 168 | 75 | 35 | 110 |
| Group 6 (28.9) | 169 - 216 | 79 | 15 | 94 |
| Total (100) | 0 - 216 | 214 | 111 | 325 |
Distribution of Patients According to Age and Gender
3.2. Ultrasonography Technique
The thyroid US examinations were performed by a single board-certified radiologist using a linear probe at a frequency of 5 - 12 MHz (Aplio 500, Toshiba Medical System, Japan).
After removing the clothing material from the neck, a sufficient amount of ultrasound gel was applied over the area of the thyroid cartilage, to ensure good contact of the US probe with the skin. Patients were examined from the submental region to the level of the suprasternal notch in a supine position with a pillow under the shoulders to maintain neck extension. Minimal pressure was applied during scanning in order to avoid distortion of the thyroid anatomy.
The thyroid gland was scanned in both the transverse and longitudinal planes. The PL was systematically evaluated as a routine part of the thyroid US examination. On gray-scale images, the PL extended in a cranial direction from the upper margin of the isthmus, with echogenicity similar to the normal thyroid gland (3).
3.3. Analysis
The presence, location, size, volume, morphology of the base, and separation from the thyroid gland were noted for each PL.
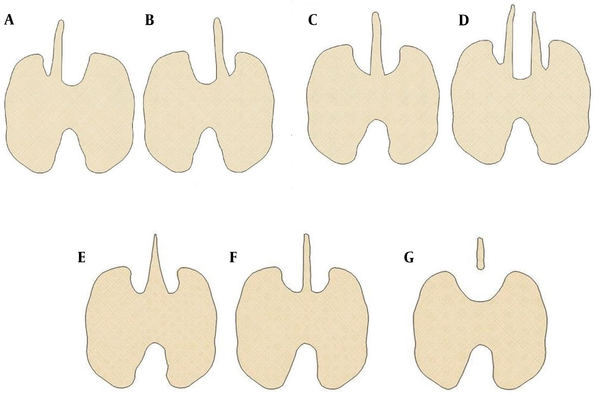
The location of the PL was classified according to the site of its origin as midline of the isthmus, right of the midline, left of the midline, and bilateral (Figure 1A - D).
The graph shows the location and morphology of the base of the pyramidal lobe. (A) right of the midline of the isthmus; (B) left of the midline of the isthmus; (C) midline of the isthmus; (D) double pyramidal lobe; (E) wide base size with narrow apex; (F) thin base size same as the apex size; (G) separation of the pyramidal lobe from the main thyroid gland.
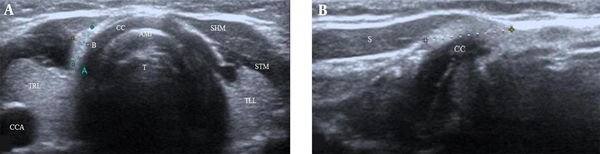
The size assessment was based on the linear dimensions and volumes of the PLs. The PL was examined in both transverse and sagittal scan planes (4, 5). On transverse scans, the maximum width of the PL was obtained. On this image, the maximum transverse (mediolateral) diameter or width of the PL was measured. The maximum anteroposterior (AP) diameter or thickness of the PL was measured perpendicular to the transverse diameter (Figure 2A). The probe was then rotated through 90°, to obtain the view of the PL in the sagittal plane. On this image, the maximum longitudinal diameter or length of the PL was measured (Figure 2B).
(A) Transverse image of the right pyramidal lobe detected on thyroid US in a 6-year-old female patient. The PL shows hyperechogenicity to adjacent muscles and homogeneous parenchymal echogenicity. A- maximum transverse diameter. B- maximum anteroposterior diameter. (B) Sagittal view shows the maximum longitudinal diameter of the PL. TRL-thyroid right lobe. TLL-thyroid left lobe. SHM - sternohyoid muscle. STM - sternothyroid muscle. T - trachea. AMI - air-mucosa interface. CC - cricoid cartilage. CCA - common carotid artery. S - strap muscles.
The volume of the PL was calculated using the following formula:
A correction factor of 0.524 was used to determine the volume of structures with ellipsoid shape.
The morphology of the base of the PL was categorized as follows: wide base size with a narrow apex and thin base size the same as the apex size (5) (Figure 1E and F).
The separation of the PL was described as a lack of continuity between the PL and the main thyroid gland (Figure 1G).
3.4. Statistical Analysis
Continuous data were presented as mean ± standard deviation (SD) or median (interquartile range, IQR), as appropriate.
The distribution of quantitative variables was tested for normality using the Kolmogorov-Smirnov and Skewness-Kurtosis test. Quantitative data with a normal distribution were compared using the one-way ANOVA (followed by Bonferroni and Games-Howell post hoc analyses), while the Mann-Whitney U test, Kruskal-Wallis test, and Spearman's correlation test were employed for the analysis of continuous variables with a non-normal distribution. Chi-square and Fisher's exact tests were used for the analysis of categorical data.
SPSS (version 23.0 Inc., Chicago, IL, USA) was used for statistical evaluation. A P-value < 0.05 was considered significant and P-value < 0.001 was considered very significant.
4. Results
The PL was present in 110 patients (87 of 214 females and 23 of 111 males; age range, 24 - 216 months; mean age ± SD, 11.92 ± 4.56 years), giving an overall prevalence of 34.1%. The PL was more frequent in girls than in boys, and this gender difference showed a strong significant correlation (40.6 vs. 20.7%, P < 0.001, respectively).
The PL originated from the left of the midline of the isthmus in 62 (56.4%) and from the right of the midline in 46 (41.8%) patients. There was 1 (0.9%) patient in whom the PL originated from the midline of the isthmus, and in 1 (0.9%) patient, the PL was located bilaterally. The left-sided PL was more frequent in males than in females, but this gender difference was not significant (65.2 vs. 54.1%, P = 0.735, respectively). The left-sided PL was more common in group 4, group 5, and group 6; however, the differences between the age groups were not statistically significant (P = 0.192). Data related to the prevalence and location of the PL are given in Table 2.
| Items | Group 1 | Group 2 | Group 3 | Group 4 | Group 5 | Group 6 | P-Value b |
|---|---|---|---|---|---|---|---|
| Prevalence of PL; % (n/actual numbers) | 0 (0/110) | 3.6 (4/110) | 10 (11/110) | 18.2 (20/110) | 30.9 (34/110) | 37.3 (41/110) | 0.016 |
| Gender | |||||||
| Female | 0 | 2 (1.8) | 7 (6.4) | 15 (13.6) | 25 (22.7) | 38 (34.5) | 0.060 |
| Male | 0 | 2 (1.8) | 4 (3.6) | 5 (4.5) | 9 (8.2) | 3 (2.7) | |
| Origin in relation to isthmus | |||||||
| Right | 0 | 3 (2.7) | 9 (8.2) | 6 (5.4) | 16 (14.5) | 12 (10.9) | 0.192 |
| Left | 0 | 1 (0.9) | 2 (1.8) | 14 (12.7) | 18 (16.4) | 27 (24.5) | |
| Midline | 0 | 0 | 0 | 0 | 0 | 1(0.9) | |
| Bilateral | 0 | 0 | 0 | 0 | 0 | 1(0.9) |
Distribution of Patients According to Gender, Location, and Prevalence of the Thyroid Pyramidal Lobe Among the Age Groups a
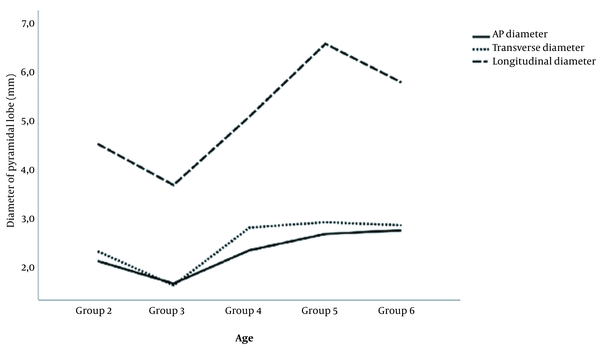
The mean values for the anteroposterior, transverse, and longitudinal diameters of the PL were 2.5 ± 1.4 (range, 0.4 - 6.6), 2.7 ± 1.3 (range, 0.6 - 8.0), and 5.6 ± 2.5 (range, 2.0 - 14.6) mm, respectively. Based on the mean values, the PL was 0.4 mm thicker, 0.3 mm wider, and 0.7 mm longer in female compared to male patients; however, these changes were not significant (2.6 vs. 2.2, P = 0.254; 2.8 vs. 2.5 P = 0.434; 5.8 vs. 5.1, P = 0.406, respectively). There were statistically significant differences in the width and length of the PL among the age groups, but no significant difference in the thickness of the PL was detected (P = 0.007, P = 0.006, and P = 0.141, respectively). Figure 3 shows the relationship between the three dimensions of the PL according to each age group.
The median (IQR) volume of the PL was 15.11 (7.25 - 39.36) mm3. Although statistically not significant, the median volume of the PL was higher in girls compared to boys [16.86 mm3 (6.78 - 41.88) vs. 10.43 mm3 (7.55 - 19.62), P = 0.238]. There was a significant positive correlation between the volume of the PL and the age groups (P = 0.015). Data for the size and the volume of the PL according to age and gender are summarized in Table 3.
| Pyramidal Lobe | Group 2 | Group 3 | Group 4 | Group 5 | Group 6 | P-Values b |
|---|---|---|---|---|---|---|
| AP diameter (mm) | ||||||
| F | 2.25 ± 0.71 | 1.31 ± 0.86 | 2.67 ± 1.45 | 2.54 ± 1.56 | 2.88 ± 1.39 | 0.067 c |
| M | 2.00 ± 0.14 | 2.28 ± 0.90 | 1.38 ± 0.41 | 3.06 ± 1.27 | 1.10 ± 0.46 | 0.011 d |
| T | 2.12 ± 0.17 | 1.66 ± 0.96 | 2.34 ± 1.39 | 2.68 ± 1.48 | 2.75 ± 1.42 | 0.141 |
| Transverse diameter (mm) | ||||||
| F | 2.60 ± 0.23 | 1.51 ± 0.50 | 2.80 ± 1.02 | 3.02 ± 1.36 | 2.90 ± 1.59 | 0.040 c |
| M | 2.05 ± 0.71 | 1.83 ± 0.56 | 2.84 ± 0.95 | 2.66 ± 0.85 | 2.30 ± 1.01 | 0.261 d |
| T | 2.32 ± 0.36 | 1.63 ± 0.51 | 2.81 ± 0.98 | 2.92 ± 1.25 | 2.86 ± 1.56 | 0.007 |
| Longitudinal diameter (mm) | ||||||
| F | 4.30 ± 0.28 | 3.30 ± 1.36 | 5.34 ± 2.22 | 6.70 ± 2.89 | 5.91 ± 2.68 | 0.016 c |
| M | 4.75 ± 0.35 | 4.35 ± 1.24 | 4.32 ± 1.29 | 6.24 ± 2.46 | 4.17 ± 0.91 | 0.519 d |
| T | 4.52 ± 0.37 | 3.69 ± 1.36 | 5.01 ± 2.05 | 6.58 ± 2.76 | 5.78 ± 2.63 | 0.006 |
| Volume (mm3) | ||||||
| F | 13.24 (11 - 17) | 1.58 (1 - 9) | 13.67 (7 - 41) | 21.10 (9 - 43) | 23.05 (7 - 43) | 0.035 c |
| M | 10.16 (10 - 13) | 11.24 (3 - 18) | 7.55 (4 - 18) | 15.99 (11 - 50) | 8.43 (1 - 17 ) | 0.133 d |
| T | 10.87 (10 - 14) | 2.05 (1 - 15) | 13.66 (6 - 37) | 20.42 (10 - 44) | 19.83 (6 - 40) | 0.015 |
The Mean Diameters and Median Volume of the Pyramidal Lobe According to Age Groups and Genders, and the Statistical Significance Among the Age Groups a
The PL with a wide base and narrow apex was present in 63.6% (70/110) of the patients, and PL with a thin base size the same as the apex size was found in 36.4% (40/110). The wide-based PL was more frequent in females than in males, but this gender difference was not significant (84.3 vs. 15.7%; P = 0.202, respectively). The thin-based PL was more frequent in girls than in boys, and this gender difference showed a significant correlation (70 vs. 30%, P = 0.003, respectively).
Complete separation of the PL from the thyroid gland was not observed in the study population.
5. Discussion
The PL -also called Morgagni's or Lalouette’s pyramid- occurs when the caudal portion of the thyroglossal duct differentiates into thyroid tissue (1, 2). All the pathologies that may be seen in the normal thyroid gland are also seen in the PL. Therefore, knowledge of anatomy of this variant is of significance to the thyroid surgeons, as incomplete removal of the thyroid tissue prevents the completeness of thyroidectomy and may cause recurrent thyroid diseases (2, 3). US is a safe, low-cost, easy-to-implement, first choice for evaluating the thyroid gland and variants in infants and children (6). The PL is located in the pretracheal region between the isthmus of the thyroid gland and the hyoid bone. The echotexture of the PL is homogeneous, bright, and similar to the parenchymal echogenicity of the normal thyroid gland (3, 5).
The prevalence of the PL in adults has been reported to vary from 15 to 75% in cadaveric, surgical, radiological, and scintigraphic series (7-10). The reasons for this wide range may include differences in diagnostic tools, selection bias, or racial and geographical factors. The largest postmortem study, carried out by Harjeet et al. (8), showed the presence of the PL in 28.9% of 570 cadavers. Another study conducted by Sturniolo et al. (9) detected the PL during thyroid surgery in 25.5% of 1405 adult patients. Using scintigraphy, Cengiz et al. (10) showed the presence of the PL in 18% of 866 patients. Using US, Mortensen et al. (5) reported the presence of the PL in 21% of 416 patients. A recent US study by Akudu et al. (11) clearly revealed no incidence of the PL among 321 adult patients, which could probably be attributed to racial factors. Moreover, in the study by Kim et al. (3), the prevalence of the PL on US and at surgery was 56.8 and 59.8%, respectively. Two studies showed that thyroid US could identify the PL more accurately than computed tomography (12, 13). The present study is the first to report the prevalence of the PLs in 325 pediatric participants. In our research, the PL was found in 34.1% of the study population, which is in agreement with much of the published literature. The present higher prevalence of the PL in adolescents compared to infants might be due to the easier ultrasound identification of the PL in older children.
Previous studies have suggested that PL is more frequent among females (3, 4, 10, 12-16), and some other studies have reported that it is more commonly seen among males (2, 8). However, in the present study, the PL was more frequent in girls than in boys, and this gender difference showed a strong significant correlation (P < 0.001).
Published data showed that PL is most commonly seen on the left side of the midline of the isthmus (3, 4, 8-15, 17). The reason for this probably lies in the fact that the thyroglossal duct is usually developed in the left caudal direction (1). In the current study, the PL was most frequently noted on the left side of the median plane (56.4%). Moreover, in one patient, the PL was located bilaterally. This finding conforms to the previous work reported by Mortensen et al. (5), who also found one patient with two PLs on thyroid US. The bilaterality rate of the PL in sonographic studies was lower than those in previous reports obtained on cadavers, ranging from 3.3 to 10% (18-20). The low bilaterality rate of the PL on US might be explained by the limitation in sonographic detection for thin PLs.
The present study is the first to evaluate the size and the volume of the PL in infants and children. The mean values for the thickness, width, and length were 2.5 ± 1.4, 2.7 ± 1.3, and 5.6 ± 2.5 mm, respectively. The median volume of the PL was 15.11 mm3. According to Kim et al. (4), the mean thickness, width, and length of the PL on thyroid US were 2.0 ± 0.9, 6.1 ± 2.2, and 22.7 ± 10.8 mm, respectively. The same study revealed that the mean volume was 168.9 ± 188.2 mm3. However, the age range in this work was 19 - 86 years and cannot be considered appropriate for pediatric patients. According to Milojevic et al. (16) and Gürleyik et al. (20), the PLs in females were longer than in males. In the present study, the PL in girls was found to be longer than in boys; however, these differences are not significant (P ≥ 0.223). This study demonstrated that the age of children is positively correlated with the size and volume of the PL.
The morphological features of the PL have been analyzed in several previous studies. Mortensen et al. (5) postulated that 28% of patients had a PL with a wide base and narrow apex and 32% had a PL with a base size the same as the top size. The same study revealed that 20% of the PLs showed complete separation from the thyroid gland. In two recent studies using CT, the PL was separated in 9.2 and 12.5% of the cases (13, 14). To the best of our knowledge, no data are available on the morphological features of the PL in infants and children. The present study reported that 63.6% of patients had a PL with a wide base and narrow apex and 36.4% had a PL with a thin base size the same as the apex size. Moreover, no complete separation of the PL was detected. Deviations from the normal embryological development of the thyroid gland, or ethnic and geographical factors might be the reason for the differences between present findings and previous data.
The present study had limitations that should be considered. This was a single-center, retrospective study. The number of subjects in each age group was relatively small. Future studies should focus on larger group sizes in order to further validate the results of the present study. There is a lack of studies to confirm the results of this study. Therefore, it is difficult to generalize the findings of this study to the whole pediatric population.
In conclusion, the present study provides first evidence for the anatomic and morphological features of the PL in the pediatric population. The present study showed, for the first time, that the age of children is positively correlated with the size and volume of the PL. The prevalence and location of the PL in the pediatric population are similar to those reported in the adult population. The high prevalence and variations in the location and size of the PL may impact the completeness of the total thyroidectomy. Therefore, it is important for the radiologists to report the presence of the PL on thyroid US.