1. Background
Fat necrosis is a common finding in the post-surgical or post-traumatic breast (1, 2). It is a nonsuppurative inflammatory change of adipose tissue. On imaging, fat necrosis shows a wide variety of presentations (3-5). Mammographically, it could manifest as lipid cysts, coarse calcifications, focal asymmetries, microcalcifications, radiolucent or even spiculated masses (6, 7). The most common findings are dystrophic calcifications and radiolucent oil cysts (8). About one third of fat necrosis appears as round or oval radiolucent oil cyst with thin capsule and egg-shell calcifications (9). Especially in case of early calcification of the fibrous rim of an oil cyst or pure calcifications at the site of previous malignancy, discrimination between early fat necrosis and cancer recurrence could be challenging (10, 11). Fat necrosis shows pleomorphic calcifications suspicious for malignancy in 4% of the cases (4, 6, 7).
Clinically, fat necrosis often manifests as a hard, palpable lump, and as it often occurs around the tumour bed this can cause anxiety in patients. Therefore, imaging to exclude recurrent disease is of great importance.
It is known that iatrogenic or non-iatrogenic trauma to the breast can induce the development of fat necrosis (1, 2, 12, 13). The mean time between breast trauma and the clinical manifestation of fat necrosis as a palpable lump is 68.5 weeks (2). In the absence of a history of trauma most fat necrosis lesions are found in the upper inner quadrant of the breast, which is thought to be due to the use of seatbelts (14).
Another known factor to increase the incidence of fat necrosis after breast conserving therapy is radiation therapy (13, 15). The 5-year rate of fat necrosis after breast conserving surgery and irradiation is reported to be up to 40% (16). A higher rate of fat necrosis after brachytherapy compared to whole breast irradiation was observed (16). The applicator size significantly influences the development of fat necrosis after intraoperative radiation (17). The formation of fat necrosis after radiation therapy without breast surgery is still unknown (15). In breast reconstruction, adjuvant chemotherapy has been described as a factor significantly influencing the development of fat necrosis (18).
2. Objectives
Aim of this study was to compare the incidence and mammographic appearance of fat necrosis in the post-surgical breast on digital mammograms with and without adjuvant treatment. This will serve as a comparison study to the upcoming neoadjuvant radiotherapy (neoRT) trial (19), where radiation therapy will be administered prior to surgical therapy.
3. Patients and Methods
3.1. Patient Cohort
This study was registered as a service evaluation at Cambridge University Hospitals NHS Foundation Trust to evaluate if the rate of detected or caused fat necrosis is higher than expected. A retrospective analysis of all consecutive patients with benign findings managed by surgical excision between January 2011 and December 2014 was performed. Forty-seven patients diagnosed with a lesion of uncertain malignant potential on needle biopsy required open surgical biopsy and confirmed to be non-malignant. Eight patients were excluded as no post-surgical mammographic follow-up was available, leaving a total of 39 patients in the study group.
Also, an analysis of all cancer patients diagnosed in year 2013, the mid point of the benign surgical cohort, in the Cambridge screening programme was performed. Primary surgery was performed in 128 patients with screen-detected breast cancers. Four patients were excluded as no mammography follow-up was available. Another 43 patients were excluded as mastectomy was performed. Hence, 81 patients built the study cohort with surgical excision of a malignant finding.
The cohorts were analyzed regarding age at the time of surgery, lesion size and additional adjuvant therapy. Available mammograms as follow-up tests were noted and analyzed regarding signs of fat necrosis.
3.2. Image Analysis
One reader with six years of experience in reading digital mammography (H.P.) performed the image analysis. The reader was aware of the laterality of the finding as well as the pre-therapeutic imaging but was not aware of the treatment additional to surgical excision. It was noted if there was any sign of (developing) fat necrosis on post-surgical imaging, such as lucencies, masses or calcifications. Measurement of the findings was performed. Findings were noted separately for each imaging time point. The different appearances of fat necrosis were noted.
3.3. Statistical Analysis
Statistical analysis was performed using SPSS (SPSS Inc. Released 2007. SPSS for Windows, version 16.0. Chicago, Ill., USA). Results are given as mean ± standard deviation (SD). Student’s t-test was used in case of continuous data, χ2-test in case of categorical data. Logistic regression was performed. Z-transform was used for age and lesion size ([x-mean]/standard deviation). P values ≤ 0.05 were considered statistically significant.
4. Results
4.1. Patient Characteristics
Mean patient age at the time of surgery was 54.6 ± 6.5 years in the benign and 59.8 ± 7.8 years in the malignant group (P = 0.0002).
4.2. Lesion Characteristics
In the benign group, the mean pathological size of the lesion demanding excision was 15.3 mm overall. The mean lesion size was 16.1 mm on mammography, and 17.5 mm on ultrasound. Thirty-two of 39 lesions (82.1%) were visible on mammography; whereas, 15/39 lesions (38.5%) were visible on ultrasound. Of the 32 lesions visible on mammography, 59.4% were calcifications only, followed by lucency and calcifications (34.4%).
In the malignant group, the mean whole pathological tumour size was 16.1 mm. The mean lesion size did not differ between the groups (P = 0.735). The mean diameter of invasive cancer was 12.7 mm and 19.7 mm of pure non-invasive tumor. Of all malignant tumors 74.1% (60/81) were invasive, 21 (35.0%) of them with an extratumoural non-invasive component. Histopathology showed carcinomas of no special type (NST) in 85.0% (51/60), invasive lobular carcinomas (ILC) in 8.3% (5/60), and mucinous and mixed NST/ILC in 3.3% (2/60) each. Invasive cancers were grade 1 in 28.3% (17/60), grade 2 in 46.7% (28/60), grade 3 in 21.7% (13/60), and the grade was unknown in 3.3% (2/60) cases. Pure non-invasive cancers made up 25.9% (21/81), of which 17 were ductal carcinoma in situ (DCIS) (81.0%) and four atypical lobular neoplasias (ALN; 19.0%).
4.3. Surgical Therapy
In the benign group, surgical excision was performed with help of wire marking. The lesions were impalpable in 14 cases (35.9%). Repeated surgery was performed in one case (2.6%) due to suspicion of microinvasion at the margin of atypical ductal hyperplasia (ADH) after the first surgery with no residual disease in the second surgical specimen and therefore, final diagnosis of ADH.
In the malignant group, breast conserving therapy was performed in 18 cases (22.2%) and breast conserving therapy with additional axillary surgery in 63 cases (77.8%). Repeated surgery was performed in 17 cases (21.0%), 15 cases of them (88.2%) to obtain tumor-free margins and two cases (11.8%) to complete surgical axillary therapy.
4.4. Adjuvant Therapy in the Malignant Group
Whole breast radiation therapy was performed in 75 patients (75/81; 92.6%). No breast irradiation was performed in six patients with in situ carcinoma. In 16 patients (16/75; 21.3%), chemotherapy in addition to radiotherapy was performed sequentially. Forty-six patients (56.8%) received adjuvant hormonal treatment in addition to radiotherapy (estrogen receptor modulator or aromatase inhibitor). Hormonal treatment without radiation or chemotherapy was performed in one case (1/46; 2.2%). The combination of breast irradiation and hormonal treatment was applied in 36 cases (78.3%) and in nine cases (19.6%), radiation, chemotherapy and hormonal treatment were performed.
4.5. Mammography Follow-Up
Mean available mammography follow-up was 4.1 years in the benign and 3.6 years in the malignant group. Bilateral mammograms in both views were available four times after surgery in four patients, three times in nine patients, two times in 16 patients and only one follow-up was available in 10 patients in the benign group. Bilateral mammograms in both views were available four times after surgery in 51 patients, three times in 25 patients, two times in two patients and only one follow-up was available in three patients in the malignant group.
4.6. Fat Necrosis
A development of fat necrosis on mammography follow-up was observed in 12.8% (5/39) of the patients in the benign group and in 32.1% (26/81) of the patients in the malignant group (P = 0.024) (Table 1). Logistic regression of all 120 patients showed that the probability of fat necrosis development was significantly higher in case of a malignant compared to a benign lesion (odds ratio [OR]: 3.57; 95% confidence interval [CI]: 1.15 - 11.01; P = 0.027); whereas, patient age and lesion size did not significantly influence fat necrosis development (P = 0.842 and P = 0.300, respectively).
| Lucency only | Lucency and calcifications | Calcifications only | Mass | Total | |
|---|---|---|---|---|---|
| Benign | 4 (80.0) | 1 (20.0) | 5 | ||
| Malignant | 1 (3.8) | 11 (42.3) | 14 (53.8) | 26 | |
| Total | 1 (3.2) | 11 (35.5) | 18 (58.1) | 1 (3.2) | 31 |
Different Appearances of Fat Necrosis on Mammograms in the Benign and Malignant Groupa
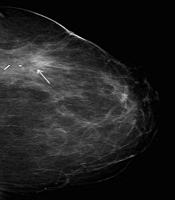
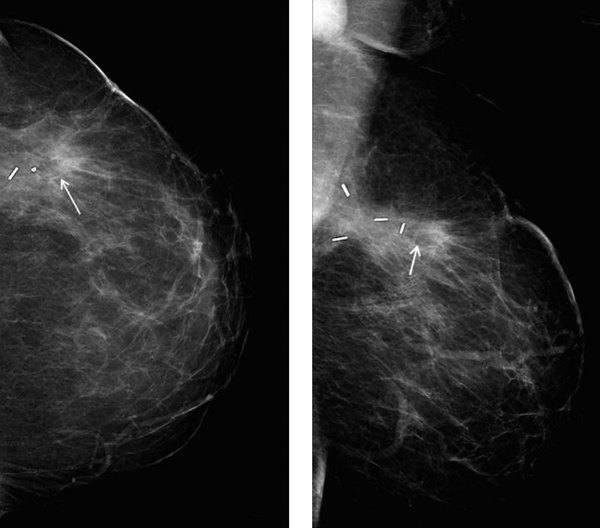
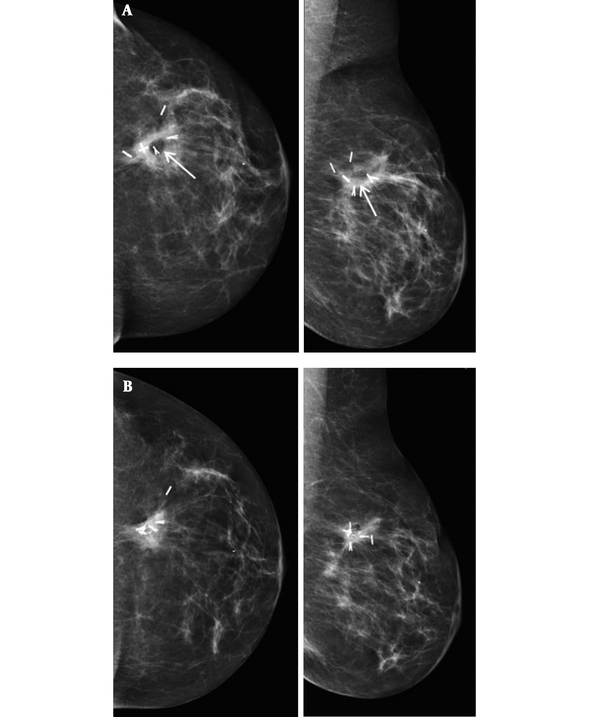
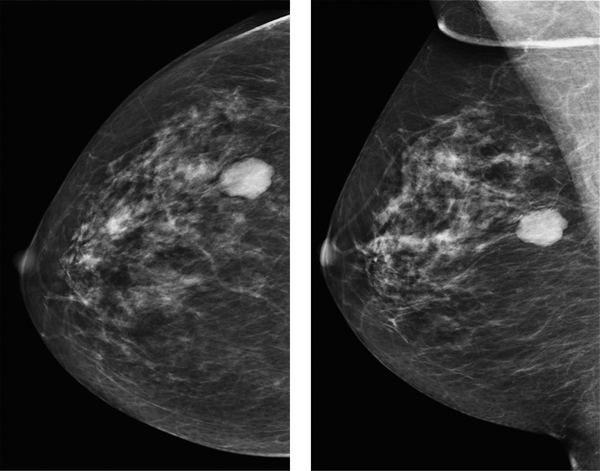
In one patient fat necrosis was detected as lucency only (Figure 1), in 18 as pure calcification, and in 11 patients as lucency with calcifications (Figure 2A and B). In one case a mass on post-surgical mammograms was aspirated and turned out to be an oil cyst (Figure 3). In one case without signs of fat necrosis, DCIS was detected on the second mammography follow-up in the benign group.
First (A) and forth (B) post-surgical follow-up mammogram after 0.9 and 4.0 years, respectively. Breast conserving therapy of a 5 mm invasive carcinoma in the left breast was performed. In the area of the post-surgical clips at the 12 o’clock position scarring tissue as well as a lucency in the centre of the clips is visible (A, arrows), which shows calcification later on (B) typical for fat necrosis.
Second post-surgical mammogram of a 61-year-old patient 4 years after surgical excision of a B3 lesion in the right breast. There is a dense mass at the 9 o’clock position in the right breast with radiological benign appearance. The lesion showed cystic appearance on ultrasound and proved to be an oil cyst in aspiration.
One of six patients (16.7%) not treated with radiotherapy developed fat necrosis in the analyzed follow-up. With radiation therapy ± hormonal treatment, 24 of 73 patients (32.9%) developed fat necrosis, whereas four of 16 patients (25.0%) treated with radiation and chemotherapy showed a development of fat necrosis. None of the nine patients treated with irradiation, chemotherapy and hormonal treatment showed fat necrosis development (Table 2). In the malignant group, logistic regression showed that hormonal treatment significantly influenced the development of fat necrosis, in detail lowering its chance as the odds ratio was < 1 (OR: 0.23; 95% CI: 0.06 - 0.86; P = 0.029) (Table 3).
| no RT +/- HT | RT only | RT + HT | RT + CT | RT + CT + HT | Total | P value | |
|---|---|---|---|---|---|---|---|
| Fat necrosis present, invasive | 4 (17.4) | 11 (30.6) | 4 (57.1) | 0 | 19 | ||
| Fat necrosis absent, invasive | 4 (17.4) | 23 (63.9) | 3 (42.9) | 9 (100) | 39 | ||
| Fat necrosis present, non-invasive | 1 (16.7) | 5 (21.7) | 1 (2.8) | 7 | |||
| Fat necrosis absent, non-invasive | 5 (83.3) | 10 (43.5) | 1 (2.8) | 16 | |||
| Total | 6 | 23 | 36 | 7 | 9 | 81 | 0.83 |
Development of Fat Necrosis According to Adjuvant Treatment and Invasive or Non-Invasive Cancer in the Malignant Groupsa
Logistic Regression Analysis of Factors Influencing the Development of Fat Necrosis in the Malignant Group
The frequency of fat necrosis development did not differ significantly between invasive and non-invasive cancers (P = 0.83) (Table 2).
Mean follow-up time until the first sign of fat necrosis was 2.6 years in the benign and 2.0 years in the malignant group (P = 0.187). In the malignant group fat necrosis was first detected after more than 2 years in 10 cases (38.5%) and in two cases (7.7%) after more than 3 years.
Overall, the mean size of fat necrosis was 9.04 mm in the malignant group. Lucencies with calcifications measured 18.6 mm on average, pure calcifications 4.1 mm. One case was a 12 mm measuring lucency only. In the benign group, fat necrosis calcifications had a mean diameter of 2.7 mm, the mass measured 21 mm and there was one case with scattered fat necrosis calcifications in the periareolar scar tissue.
5. Discussion
Our study showed that significantly more cases of fat necrosis are observed in the operated breast when additional adjuvant treatment was performed compared to surgery alone (32.1% vs. 12.8%). The chance of fat necrosis development was 3.5 times higher in case of a malignant lesion that was operated. In prior studies, radiation therapy was quoted as a factor significantly influencing the development of fat necrosis (13, 15, 16). In our patient collective radiation did not significantly influence fat necrosis development. Chemotherapy alone has been reported as a factor influencing the development of fat necrosis in breast reconstruction after mastectomy (1). As none of the patients in our study collective received chemotherapy without irradiation of the breast, this could only be analyzed in logistic regression. The only factor significantly influencing the development of fat necrosis was hormonal treatment. In case of hormonal treatment, the chance of fat necrosis development was significantly lower.
The appearance of fat necrosis ranges from incidental benign findings to imaging features highly suggestive of malignancy (20, 21). The majority of fat necroses presented as pure calcifications in our study (59.4%), followed by lucencies with calcifications (34.4%). This is in accordance with literature pointing out dystrophic calcifications and radiolucent oil cysts as the most common mammographic findings (8). If fat necrosis becomes manifest as calcifications only the probability that it is mistakenly scored as suspicious for recurrent malignancy is high. In case of a lucency in addition to the calcification, the fat necrosis can be verified on ultrasound (21). The mean follow-up time until fat necrosis manifested on the mammogram was 2.6 years in the benign and 2.0 years in the malignant group. As 46.2% of the fat necrosis after surgery of breast cancer were initially detected more than two years after the surgery, it is reasonable to have annual follow-up of at least 5 years to be able to detect those changes. Even on MRI a delayed development of enhancement in fat necrosis has been reported (22). If accessible, MRI has an excellent negative predictive value in case of uncertain mammographic lesions without enhancement, but if there is enhancement in a probable fat necrosis, biopsy has to be performed to exclude malignancy (22).
Dependent on adjuvant treatment the development of fat necrosis was observed in 17% of the patients not treated with radiotherapy, in 33% of the patients treated with radiation therapy ± hormonal treatment and in 25% of the patients treated with radiation and chemotherapy in our study. The reported rate of fat necrosis after breast conserving therapy and irradiation is up to 40% and both radiation and chemotherapy have been shown to increase the risk of development of fat necrosis (16, 18). However, in our series, the effect of adjuvant chemotherapy did not add to the effect of radiotherapy. As fat necrosis often occurs around the tumour bed, it will be highly interesting to compare our results with the incidence, location, morphology and time of appearance of fat necrosis after neoadjuvant radiotherapy. As part of the neoRT trial, surgical excision of the malignant findings will be performed after neoadjuvant radiotherapy, so part of the radiated tissue will not stay in the breast. Perhaps this will end in a lower rate of fat necrosis, and the time of appearance after surgery will differ.
There are limitations in our study. First, due to the small case numbers overall and the small case numbers of histopathological cancer types other than no special type (NST), no analysis of a possible impact of tumor histopathology on the development of fat necrosis could be performed. Second, the presence of fat necrosis was only rated radiographically as ultrasound follow-up is not performed routinely in Cambridge or the UK.
In conclusion, this study shows the supposable impact of adjuvant therapy to the development of fat necrosis after surgery in a series of cancer patients with adjuvant radiotherapy compared to patients with surgery of benign findings. This has to be compared to the incidence, morphology, and time span of the potential development of fat necrosis after neoadjuvant radiotherapy with and without chemotherapy and hormonal treatment as part of the radiated tissue will be removed during surgery.