1. Background
The term ‘cephalocele’ refers to extracranial extension of meninges with or without brain tissue. Cephaloceles consist of two types. It is called meningocele when only meninges are involved, and it is called meningoencephalocele if the cerebral tissue protrudes with meninges. Cephaloceles in the anterior cranial base may lead to spontaneous cerebrospinal fluid (CSF) leakage to the sinonasal cavity, called rhinorrhea. CSF leaks are mostly traumatic (accidental or iatrogenic). Only 3% - 4% of CSF leaks are spontaneous and female/ male ratio is 2/1 (1).
Accurate localization of CSF fistulas has an important role in the surgical treatment approach. Presurgical localization increases the chance for proper dural repair and could prevent complications (2). Successive surgical repair of dural tear is also needed to prevent recurrent meningitis (3).
Several imaging techniques are used for detection of proper leakage location. Precise localization of CSF leakage still remains a challenge despite advances in imaging for neuroradiologists. Computed tomography (CT) of paranasal sinuses, intrathecal contrast enhanced CT (CT-cisternography) and heavily T2 weighted magnetic resonance (MR) imaging and intrathecal gadolinium enhanced MR cisternography (Gd-MR cisternography) are mostly used modalities in clinical routine (4). CT has superiority to detect bone defects in the cranium base, but it cannot directly reveal the CSF leakage, which is the major limitation of CT. CT gives false positive results with bone defects without meningeal tear (5). CT cisternography can also show the leak localization, but ionizing radiation and the application of iodine-based contrast material are the major disadvantages of this technique. Intrathecal injected iodinated contrast material cannot disperse in CSF freely causing false negative results. In addition, it has adverse effects including arachnoiditis, epilepsy, and allergic reactions (6, 7).
Gd-MR cisternography is a well-known minimal invasive imaging technique in suspected rhinorrhea. Several reports in the literature described its higher diagnostic sensitivity and specificity in localizing CSF leakage (5, 7, 8).
2. Objectives
This study aimed to present our experience of Gd-MR cisternography in patients with rhinorrhea, and particularly evaluate the frequency of cephaloceles among rhinorrhea patients.
3. Patients and Methods
All Gd-MR cisternography procedures performed between October 2012 and September 2018 were evaluated retrospectively. This study was approved by the local ethics committee. Indications for MR cisternography in our department were rhinorrhea, otorrhea and idiopathic intracranial hypotension, and arachnoid cysts. Patients suffering from persistent or intermittent rhinorrhea and with positive Gd-MR cisternography findings were included in the study. Gadolinium leakage from subarachnoid spaces to paranasal sinuses was accepted as the positive finding for rhinorrhea. Twenty five patients underwent Gd-MR cisternography in the study period, and four patients with suspected rhinorrhea had normal Gd-MR cisternography. A total of 21 patients in whom extravasation from subarachnoid space to paranasal sinuses or nasal cavity was detected, were included. Sixteen of 21 patients also had non-contrast CT examination of paranasal sinuses. Localization of extravasation and causes of CSF leakage were analyzed. Beta2-transferrin test was investigated via database records of our institute.
Patients suffering from suspected rhinorrhea lasting more than 1 month were referred from otorhinolaryngology and/or neurosurgery departments. In these patients, Gd-MR cisternography was carried out in our neuroradiology department after informed consent. Claustrophobia and uncorrectable coagulopathy were accepted as contraindications for the procedure. Before acquisition of MR images, 1 mL gadoterate meglumine (dotarem) was injected intrathecally with a 22 G Chiba needle under sterile conditions at L4 - L5 level. Intrathecal injections were executed with fluoroscopy guidance while patients were in lateral decubitus position by bringing the head and legs closer to the abdomen. Local anesthesia was not administered. After the injection of intrathecal gadolinium, patients were transferred to the MRI scanner in prone and 30 - 40º head down position to increase the transport of contrast material to basal cisterns and sulci in the frontal basal region. All patients were observed for 24 hours after intrathecal injection to assess for adverse effects. MRI protocols included sagittal 3D isotropic T1-weighted turbo field echo sequence (3D isotropic T1W-TFE) with the following imaging parameters: repetition time (TR)/echo time (TE), 8.05 to 8.25 milliseconds/3.65 to 3.75 milliseconds; flip angle, 9º; matrix, 240 × 240; field of view, 220 × 236 × 179; sensitivity encoding factor, 1; slice thickness, 1 mm, and sagittal 3D-Flair with the following imaging parameters TR/TE, 4800/275 milliseconds; flip angle, 90º; matrix, 224 × 224; sensitivity encoding factor, 1; inversion time, 1650 milliseconds; slice thickness, 1 mm. Images were obtained with a clinical 3 Tesla MR imaging system (PhilipsAchieva3T, Best, Netherlands) using 32 channel sensitivity encoding head coil.
Obtained Gd-MR cisternography and also non-enhanced paranasal CT images (scanned before MR imaging) were transferred to a workstation (GE Advantage Workstation; GE Healthcare, Milwaukee, WI) and evaluated by two neuroradiologists with 15 and 5 years of experience with consensus agreement. Leakage of gadolinium-based contrast material on MR imaging and bone defects on CT imaging were analyzed. All statistical analyses were performed with commercially available software (SPSS 16.0 for Windows; SPSS Inc, Chicago, IL). The data were entered into Excel spreadsheets (Microsoft, Redmond, WA).
4. Results
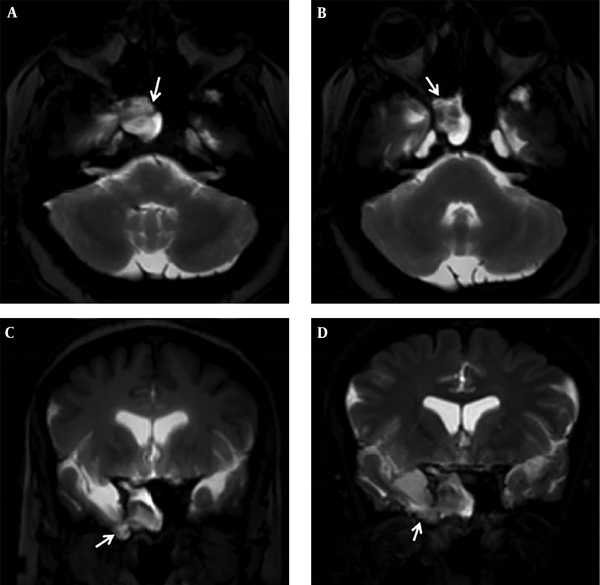
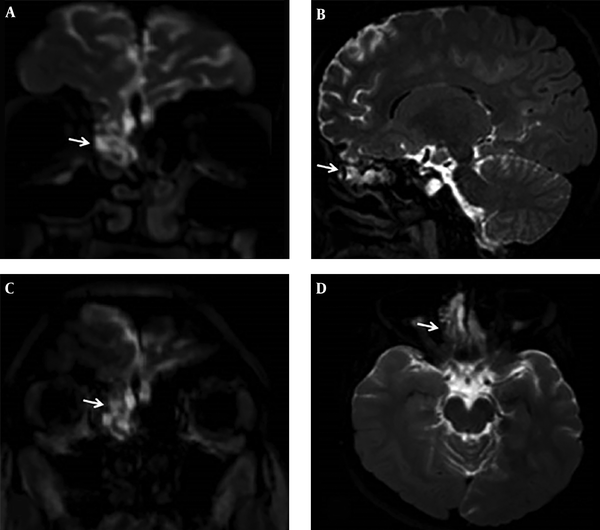
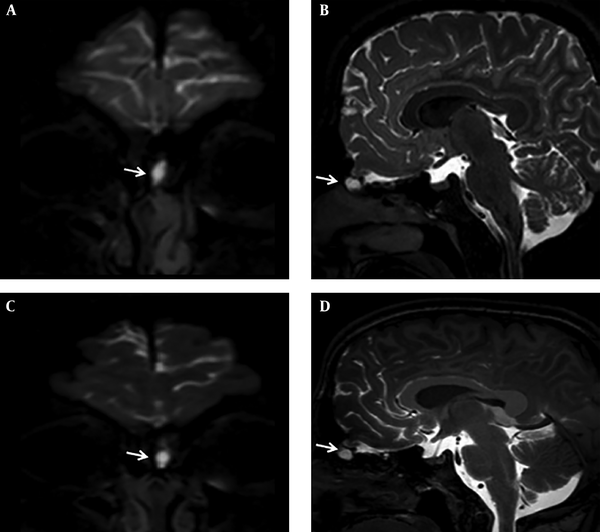
The patient group consisted of 21 patients (15 female and 6 male) between 16 and 59 years of age (mean age: 43.5). Demographic data and CSF leak sites are demonstrated in Table 1. Beta2-transferrin test was positive in 15 patients, negative in two patients and was not performed in four patients. In all 21 patients, gadoterate meglumine entered basal cisterns and sulci in the frontal basal region after intrathecal injection successfully. No complication related to intrathecal gadolinium injection was detected during the first 24 hours after the procedure or during the follow-up period. CSF leakage from subarachnoid space was observed objectively by two neuroradiologists (15 and 5 years experienced) with consensus. Location of leakage was frontal sinus in one patient, sphenoid sinus in 10, and ethmoidal sinus in 10 patients. In the evaluation of CT images, there were bone defects in 12 of 16 patients and the localization of bone defects were consistent with the CSF leakage sites in Gd-MR cisternography. Two patients with bone defects in the frontal region did not show positive Gd-MR cisternography finding for CSF leakage. Ten (47.6%) of the patients had a cranial trauma, five (23.8%) had an endoscopic sinus surgery history, one (4.8%) had a history of rhinorrhea after suppurative meningitis, while five patients (23.8%) were accepted as spontaneous rhinorrhea. Cephalocele was detected in 10 patients (47.6%), of which seven were meningocele and three were meningoencephalocele. One of these cephaloceles was spontaneous, while five were traumatic, three were secondary to endoscopic surgery and one meningocele was developed secondary to infectious meningitis. In two traumatic meningoencephaloceles, one patient had meningoencephalocele via a defect in the lateral wall of the sphenoid sinus (Figure 1), and the other one was through ethmoidal air cells (Figure 2). In one patient with a history of recurrent meningitis, CSF leakage was detected through the cribriform plate (Figure 3). Eight of 10 cephaloceles were detected in endoscopic surgery for dural repair, while two of the cephaloceles refused operation. Sixteen of 21 patients underwent endoscopic closure of the dural tear. On the other hand, five patients refused endoscopic surgery and they are still on follow-up.
Intrathecal gadolinium enhanced MR cisternography examination in a 52-year-old female patient with post-traumatic persistent rhinorrhea. Axial and coronal three-dimensional T1 Weighted (3D-T1W) (A, C), and axial and coronal three dimensional fluid attenuated inversion recovery (FLAIR) (B, D) images showing hyperintense cerebrospinal fluid and accompanying temporal lobe hernia in sphenoid sinus, concordant with meningoencephalocele.
Intrathecal gadolinium enhanced MR cisternography examination in a 16-year-old female patient with post-traumatic persistent rhinorrhea. Coronal and sagittal three dimensional fluid attenuated inversion recovery (FLAIR) (A, B) images, and coronal and axial three dimensional T1 weighted (3D-T1W) (C, D) images showing hyperintense cerebrospinal fluid and accompanying frontal lobe hernia into ethmoid air cells.
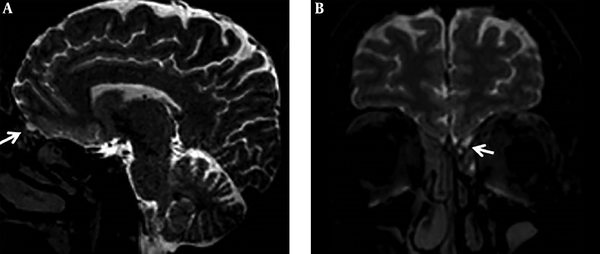
Intrathecal gadolinium enhanced MR cisternography examination in a 22-year-old male patient with persistent rhinorrhea. Coronal and sagittal three dimensional fluid attenuated inversion recovery (FLAIR) (A, B) images, and coronal and sagittal three dimensional T1 weighted (3D-T1W) (C, D) images showing a cystic pouch filled with gadolinium based contrast material, accepted as meningocele.
| Patient No. | Age | Gender | Etiology | Outcome | Cephalocele | Leakage site |
|---|---|---|---|---|---|---|
| 1 | 52 | F | Trauma | Operated and cured | Meningoencephalocele | Lateral sphenoid |
| 2 | 48 | M | Trauma | Operated and cured | - | Cribriform plate |
| 3 | 16 | F | Trauma | Operated and cured | Meningoencephalocele | Cribriform plate |
| 4 | 22 | M | Meningitis | Operated and cured | Meningocele | Cribriform plate |
| 5 | 41 | F | Iatrogenic | Operated and cured | Meningocele | Cribriform plate |
| 6 | 32 | F | Iatrogenic | Operated and cured | - | Cribriform plate |
| 7 | 43 | F | Trauma | Operated and cured | - | Lateral sphenoid |
| 8 | 54 | F | Spontaneous | Operated and cured | - | Cribriform plate |
| 9 | 46 | M | Trauma | Follow up | - | Midline sphenoid |
| 10 | 53 | F | Trauma | Operated and cured | Meningocele | Midline sphenoid |
| 11 | 59 | F | Spontaneous | Operated and cured | Meningocele | Cribriform plate |
| 12 | 37 | M | Iatrogenic | Follow up | Meningocele | Cribriform plate |
| 13 | 28 | F | Trauma | Follow up | - | Frontal |
| 14 | 56 | F | Trauma | Follow up | Meningocele | Midline sphenoid |
| 15 | 33 | F | Iatrogenic | Operated and cured | - | Cribriform plate |
| 16 | 45 | F | Spontaneous | Operated and cured | - | Cribriform plate |
| 17 | 55 | F | Trauma | Operated and cured | - | midline sphenoid |
| 18 | 57 | F | Spontaneous | Follow up | - | Lateral sphenoid |
| 19 | 41 | F | Spontaneous | Operated and cured | - | Midline sphenoid |
| 20 | 53 | M | Iatrogenic | Operated and cured | Meningoencephalocele | Lateral sphenoid |
| 21 | 43 | M | Trauma | Operated and cured | Meningocele | Midline sphenoid |
Demographic Data of Patients and Localization of Cerebrospinal Fluid Leakages
5. Discussion
Cephalocele (congenital or acquired), is defined as protruding of the meninges with or without cerebral parenchyma (1). Cephaloceles protruding through the sinonasal cavity can present with rhinorrhea, nasal obstruction, and recurrent meningitis. Therefore, surgical repair of the dura is mandatory for treatment. Radiological detection of the dural tear and the location of CSF leakage is important, because it increases the chance of successful surgical approach (2). Several imaging methods have been evolved for diagnosis of CSF leakage location. Frequently, un-enhanced CT of paranasal sinuses is the first radiological technique, requested by clinicians in patients with suspected rhinorrhea. CT has advantages and disadvantages in the diagnosis of CSF leakage location. It is easily accessible in many times, fast and cheap regarding to MR imaging. Bony defects in the anterior cranial fossa are best revealed with CT. But this is an indirect sign, and CT images do not show CSF leak directly. Bony defect and CSF leak location does not overlap in all patients. However, in patients with multiple bone defects, it is difficult to guess where the leakage is (9). Because of these limitations, CT imaging is commonly combined with other modalities in many institutions. We use CT imaging in combination with Gd-MR cisternography in our center (7, 8, 10).
CT cisternography is a widely used technique worldwide. Nevertheless, CT cisternography has also disadvantages including low sensitivity in low flow fistulas, ionizing radiation exposure and adverse effects of intrathecal administration of the iodinated contrast material. Iodinated contrast materials do not spread diffusely in CSF, decreasing the sensitivity of CT cisternography. A study conducted by Goel et al. reported 38% sensitivity of CT cisternography, while three dimensional constructive interference in steady state (3D-CISS) had 75% and intrathecal gadolinium enhanced MR cisternography had 89% sensitivity (4). Diffuse infiltration of gadolinium in CSF gives rise to high sensitivity of Gd-MR cisternography.
MR imaging without contrast enhancement is another radiologic technique used in patients with rhinorrhea. Heavily T2 weighted 3D-constructive interference in steady state (CISS) is the sequence used for unenhanced MR cisternography. However, 3D-CISS images have a high false negative and false positive results. In the study performed by Hegarty et al., there were 40% false positive results with unenhanced MR-cisternography method. Particularly, when there is an accompanying paranasal sinus inflammation, false positive results may occur. As a result of its higher rates of false positive and negative results, we do not use 3D-CISS in our routine clinical practice. Even so, its sensitivity is higher than CT cisternography (9, 11).
Gd-MR cisternography is a minimally invasive diagnostic method with intrathecal gadolinium injection. First studies of Gd-MR cisternography were on animal models (12, 13). Subsequently, various human studies reported highest sensitivity, specificity, and accuracy ratios with Gd-MR cisternography in the detection of CSF leakage in patients with rhinorrhea (4, 7, 14, 15). In many institutes, Gd-MR cisternography is applied to detect CSF leakages in patients suspected with rhinorrhea and otorrhea. However, there is no consensus on the amount and type of intrathecal gadolinium-based contrast material. According to a review report analyzing 33 articles about Gd-MR cisternography (16), Gadopentate dimeglumin (Magnevist) with the range of 0.2 - 1 mL was used in 29 studies, three studies used gadodiamide (Omniscan) with the range of 1 - 2 mL and gadobutrol (Gadovist) (1 mL) was used in one study. We used 1 mL gadoterate meglumine (Dotarem) to reveal CSF leakage. To our knowledge, in the literature, there is only one report that evaluated intrathecal injection of gadoterate meglumine. According to this report by Ozturk et al., there was no measurable signal intensity changes in dentate nucleus and globus pallidus after a single dose injection of gadoterate meglumine (17). Similar with this report, we also did not encounter any major adverse effects related to intrathecal gadoterate meglumine injection.
CSF fistulas are commonly a result of head trauma. 80% of CSF leaks were post-traumatic, while 17% were secondary to endoscopic surgical interventions. Only 3% - 4% were spontaneous (18). Etiologies of rhinorrhea in our patient group were similar to the literature. Cephaloceles of anterior cranial base commonly cause rhinorrhea when dural tear occurs. In case of a cephalocele, as a cause of rhinorrhea, patients may present with nasal obstruction. Endoscopic examination may reveal smooth polypoid mass in the nasal cavity. In this manner, radiological evaluation of rhinorrhea exhibits more importance. Because biopsy or removal of the polypoid mass in the nasal cavity is contraindicated, it may contain meninges and brain parenchyma (19). None of the patients in the study group presented with nasal obstruction findings. There were only two meningoencephaloceles in our patients. In one patient, frontal lobe protruded through the ethmoid air cells and in one patient, the right temporal lobe protruded through the sphenoid sinus. The other five cephaloceles were small meningoceles and all presented with watery nasal discharge (Figure 4).
In the literature, several studies reported cribriform plate as the most common location of CSF leakage in spontaneous rhinorrhea patients (20-23). The other possible sites for spontaneous CSF fistulas were craniopharyngeal canal, sella and sphenooccipital synchondrosis. Arachnoid granulations near the ethmoid and sphenoid sinus were seen responsible for meningocele development (23). In our series, three of five spontaneous CSF leakages were in the cribriform plate, one midline sphenoid and one was in the lateral sphenoid location. Selcuk et al. reported ethmoid sinuses as the common location of CSF leakage in their study with Gd-MR cisternography in rhinorrhea patients. Locations of CSF leakage in our series including 21 rhinorrhea patients were compatible with the literature (9).
This study had several limitations. Its retrospective design and limited number of patient groups was the first limitation. Second, five of 21 patients did not undergo endoscopic diagnosis. Third, MRI protocol in this study did not include precontrast images. To our knowledge, there is only one report evaluating Gd-MR cisternography with gadoterate meglumine, and this is the first study evaluating CSF leaks with Gd-MR cisternography using intrathecal gadoterate meglumine (17).
We conclude that MR cisternography with intrathecal administration of gadoterate meglumine is an effective, minimal invasive and safe diagnostic method inducing admissible adverse effects for evaluating CSF leakage in patients with suspected rhinorrhea. Multiplanar imaging capability of Gd-MR cisternography without ionizing radiation exposure provides high sensitivity and specificity in detecting CSF leakage. Cephaloceles must be kept in mind as a cause in non-traumatic rhinorrhea. As a supplement to CT imaging, Gd-MR cisternography enables detection of CSF leakage location and also meningeal and cerebral parenchyma protrusion.