1. Background
Transjugular intrahepatic portosystemic shunt (TIPS) has been known to be effective in treating complications of portal hypertension such as gastroesophageal variceal bleeding and refractory ascites (1-3). However, with the development of endoscopic techniques and the introduction of less invasive alternative procedures such as balloon-occluded retrograde transvenous obliterans (BRTO) and plug-assisted retrograde transvenous obliterans (PARTO), the number of cases of TIPS has decreased (4-7). Therefore, maintenance of skill is somewhat difficult (8).
Furthermore, it is agreed upon that puncture from the hepatic vein to the portal vein to create a shunt is the most challenging step in the procedure (2, 8-10). Various techniques seeking to enhance the feasibility of portal vein access have been described in the literature in the past decades. For instance, procedures such as wedge hepatic portography with CO2 injection, intravenous ultrasound (IVUS), direct portography via portal vein puncture and ultrasound (US), computed tomography (CT), and magnetic resonance imaging (MRI)-guided puncture have been introduced. But in many cases, some of these techniques are not available, owing to various factors as well as potential risks of complications (11, 12).
Previously, Matsui et al. introduced hepatic artery targeting wire with fluoroscopy to facilitate portal vein puncture (10). Additionally, Yamagami et al. reported that this technique exhibited a high technical success rate and a low complication rate (8). To our knowledge, however, there have been no studies comparing this hepatic artery guiding technique with the simple blind puncture technique.
2. Objectives
In this study, we evaluated the effectiveness and feasibility of the hepatic artery guiding technique in the TIPS procedure.
3. Patients and Methods
3.1. Patients
This retrospective study was conducted in two hospitals, including one tertiary and one secondary institution. The institutional review boards of each institution approved this study, and the requirement for informed consent was waived. From January 2009 to December 2018, data from consecutive patients who underwent TIPS procedures were collected. In total, 41 patients (36 men, five women; mean age, 57.9 years; range, 33 - 77 years) underwent the TIPS procedure during the specified study period. Electronic medical records from each patient were obtained for demographic characteristics and indications of TIPS.
3.2. Procedures
All TIPS procedures were performed in routine method in the angiography suite by four trained interventional radiologists with 2 - 20 years of experience in interventional radiology. Sterile preparation of the right neck was performed, and the right internal jugular vein was punctured under ultrasound guidance. Dedicated ring transjugular intrahepatic access sets (Cook, USA, Bloomington, IN) were used in all patients to catheterize the appropriate hepatic vein. Hepatic venography was taken via a 5Fr catheter to visualize the hepatic venous anatomy. A TIPS sheath was then introduced to stabilize the needle for puncture. After puncturing from the proximal hepatic vein with a 16-gauge Colapinto needle to target the portal vein, direct portography was obtained via a 5Fr catheter at either the superior mesenteric vein or the splenic vein. Balloon dilatation for the TIPS shunt tract was performed, and the appropriate partially covered stent (Niti-S, Taewoong Medical, Gimpo, Korea) was then deployed from the portal vein to the hepatic vein nearby the inferior vena cava. Pressures at the hepatic vein, portal vein, and shunt were recorded in each procedure for a pressure gradient. In patients who had a lingering high-pressure gradient between the portal vein and the TIPS shunt, additional balloon dilatation was performed. In patients who had residual vascular flow to the varices, variceal embolization was performed.
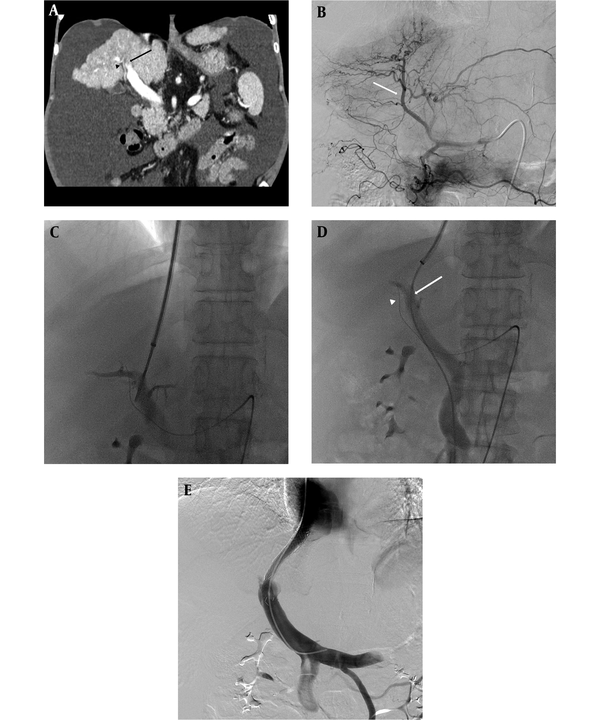
In the hepatic artery guiding technique group, angiography was performed before puncturing the right internal jugular vein. With celiac and/or superior mesenteric angiography, indirect portography was obtained as well as hepatic artery angiography to delineate the portal vein and hepatic artery anatomy. Determination of the precise anatomy of the hepatic vein, portal vein, and hepatic artery on pre-procedural CT scans was done. The closest hepatic artery adjacent to the targeted portal vein was negotiated with a 0.035-inch guide wire or with a coaxial micro guide wire system. As in many cases, right portal vein traverse posterior-superior aspect of the right hepatic artery, so right hepatic artery was commonly negotiated with the wire. Next, a 16-gauge Colapinto needle was used to puncture in the posterior-superior direction of the targeted wire. If the puncture failed, the C-arm angle was rotated to adjust the puncture direction for the target portal venous system. After these steps, the operators proceeded with the TIPS procedure as in the previously described technique (Figure 1).
Hepatic artery guiding technique. A, Pre-procedural CT scan showing a large amount of ascites. The right hepatic artery (arrow head) traverses the inferior aspect of the main and right portal vein (arrow). B, Celiac angiography taken before transjugular intrahepatic portosystemic shunt (TIPS) procedure for hepatic artery guiding. Here, the right hepatic artery (arrow) was used as the reference. C, Right portal vein punctured with a 16-gauge Colapinto needle. To avoid hepatic artery injury, the puncture was attempted at the superior aspect of the wire. D, Portography obtained via a 5Fr catheter at the superior mesenteric vein. Here, puncture time was 9 min. The position of the wire (arrow head) in the hepatic artery and the catheter (arrow) in the portal vein. E, Final portography showing successful TIPS without complications. The pressure gradient between the portal vein and the inferior vena cava was roughly 10 mmHg.
No additional means were implemented to delineate the portal vein in the simple blind puncture group. In the hepatic artery guiding technique group, different movements of the C-arm angle were attempted to aid in targeting portal vein puncture. In both technique groups, the procedures were carried out conscientiously and meticulously in order to avoid complications and decrease the radiation dose of each patient. Decision of each type of technique was decided by each operators’ preference and no other techniques other than hepatic artery guiding technique was attempted in all patients.
3.3. Data Evaluation
Detailed information from the procedures was obtained via the picture archival communication system (PACS). The selected hepatic vein and punctured portal vein were recorded. Exact time of initial angiography and/or initial hepatic venography, initial portography after puncturing the portal vein, and the last portography after TIPS creation were recorded via the PACS system. In some patients who were available for the total procedural radiation dose, each individual radiation dose was recorded as the radiation quantity (mGy) and dose area product (μGym2). Any additional procedures following TIPS creation were recorded.
Technical success was defined as successful creation of TIPS between the hepatic vein and the intrahepatic branch of the portal vein. Total procedure time was defined as the time interval between initial hepatic venography (simple blind puncture) or initial angiography (hepatic artery guiding technique) and last portography after successful TIPS creation. Puncture time was defined as the time interval between the last hepatic venography and initial portography. Radiation dose was defined as the sum of each radiation dose through the course of the procedure. According to the quality improvement guidelines established by the Society of Interventional Radiology, a minor complication is defined as no therapy required or the administration of nominal therapy including overnight observation. A major complication is defined as major therapy required, with unplanned, increased levels of care, prolonged hospitalization, permanent adverse outcomes, or death (13).
3.4. Statistical Analysis
Comparison between the time interval of total procedure and puncture time in the simple blind puncture and hepatic artery guiding technique groups was performed using the Mann-Whitney U test. Comparison between radiation doses in the simple blind puncture and hepatic artery guiding technique groups was also done using the Mann-Whitney U test. Results were presented as mean ± standard deviation. In the hepatic artery guiding technique group, mean puncture times were compared among operators using the Kruskal-Wallis test. Statistical significance was set at P < 0.05. All statistical analyses were performed using the Statistical Package for Social Science for Windows (SPSS 21.0 version; IBM Corp, Armonk, NY, USA).
4. Results
Among 41 patients, 11 patients (26.8%) underwent TIPS procedure by simple blind puncture, and 30 patients (73.2%) underwent TIPS procedure by hepatic artery guiding technique. Technical success was achieved in all patients. The most common indication for TIPS was refractory ascites (n = 23, 56.1%) followed by variceal bleeding (n = 14, 34.1%). In most of the patients, TIPS creation was performed between the right hepatic vein (n = 40, 97.6%) and the right portal vein (n = 34, 82.9%). After TIPS creation, four patients underwent additional procedures for variceal embolization (left gastric vein embolization [n = 2] and posterior gastric vein embolization [n = 1]) and portal vein thrombus aspiration (n = 1). No significant complications were observed in either group. Demographic characteristics of the patients and procedure details are listed in Table 1.
| Simple blind (n = 11) | Hepatic artery guiding (n = 30) | Total | |
|---|---|---|---|
| Age (years)a | 58.4 (48 - 74) | 57.8 (33 - 77) | 57.9 (33 - 77) |
| Sex (female to male ratio) | 2:9 | 3:27 | 5:36 |
| Indication | |||
| Refractory ascites | 8 | 15 | 23 |
| Variceal bleeding | 2 | 12 | 14 |
| Refractory hydrothorax | 1 | 1 | 2 |
| Ascites + variceal bleeding | 0 | 2 | 2 |
| Hepatic vein | |||
| Right | 11 | 29 | 40 |
| Middle | 0 | 1 | 1 |
| Portal vein | |||
| Right | 11 | 23 | 34 |
| Left | 0 | 5 | 5 |
| Main | 0 | 2 | 2 |
| Thrombosed portal vein | 0 | 2 | 2 |
| Additional stenting | 3 | 2 | 5 |
| Post-balloon PTA | 2 | 8 | 10 |
| Additional procedure | 0 | 4 (3 variceal embolizations; 1 portal vein thrombus aspiration) | 4 |
| Complication | 0 | 0 | 0 |
Demographic Features and Procedure Details
The mean total procedure time was 86.92 ± 26.55 min. Differences in procedure times between the hepatic artery guiding technique group (93.2 ± 23.94 min) and the simple blind puncture group (94.3 ± 36.32 min) were not statistically significant (P = 0.757). The mean puncture time for all patients was 27.12 ± 18.65 min. When comparing the two groups, mean puncture time was significantly shorter in the hepatic artery guiding technique group (26.67 ± 11.46 min) than in the simple blind puncture group (38.50 ± 29.69 min) (P = 0.045) (Table 2). In the hepatic artery guiding technique group (n = 30), three operators performed TIPS using this technique. Mean puncture times for the operators were 19.7 (n = 17), 32.8 (n = 5), and 24.3 (n = 8) min. There were no statistical differences in mean puncture times among the operators (P = 0.13).
The total radiation dose was available in 25 patients, including 10 from the simple blind puncture group and 15 from the hepatic artery guiding technique group (Table 2). Between the two groups, hepatic artery guiding technique group showed high radiation dose in both the radiation quantity and the dose area product. However, there was no statistical significance (P = 0.086 and P = 0.912).
| Total (range) | Simple blind puncture (range) | Hepatic artery guiding (range) | P value | |
|---|---|---|---|---|
| Total procedure time (minutes) | 86.92 ± 26.55 (38 - 145) | 94.3 ± 36.32 (51 - 145) | 93.2 ± 23.94 (53 - 130) | 0.757 |
| Puncture time (minutes) | 27.12 ± 18.65 (1 - 117) | 38.50 ± 29.69 (8 - 117) | 26.67 ± 11.46 (9 - 50) | 0.045 |
| Radiation quantityb (mGy) | 2273.26 ± 1444.92 (227 - 5450) | 2384.20 ± 1755.56 (227 - 5450) | 2199.30 ± 1258.24 (617 - 4693) | 0.912 |
| Dose area productb (μGym2) | 44783.68 ± 25418.00 (5034 - 104150) | 33487.60 ± 21795.65 (5034 - 73212) | 52314.40 ± 25500.22 (20017 - 104150) | 0.086 |
Procedure Time and Radiation Dosea
5. Discussion
In the present study, a technical success rate of 100% was achieved with both the simple blind puncture and the hepatic artery guiding technique, and no complications occurred in either group. With the assistance of the hepatic artery guiding technique, the mean puncture time was significantly shorter than the mean puncture time in the simple blind puncture group (P = 0.045). Furthermore, there were no significant differences in the total procedure time, despite the additional time required for angiography and guidewire placement. However, since this study is intended to evaluate the ease of puncture, we believe that evaluation of the puncture time itself is of importance.
The most frequently used technique to visualize the portal vein is wedge hepatic venography by CO2 or iodinate contrast (14, 15). However, portal vein images can sometimes be quite poor due to technical and physiological factors (15). There is also the potential risk of parenchymal laceration, air locking, and air embolism (16). Intravenous ultrasound has been attempted with good results in portal venous access (12). However, IVUS is still an unfamiliar device for radiologists, and it is somewhat unavailable for use by radiologists in Korea. Although direct portal vein approach can provide good image quality of the portal venous system, it is time consuming and suffers from potential several complications (17, 18). Furthermore, these procedures also require additional time to be carried out.
Previously, Matsui et al. introduced a hepatic artery targeting wire technique, having a high technical success rate (10). As the hepatic artery traverses close to portal venous system and the right hepatic artery runs anterior to the right portal vein, keeping the guide wire in the hepatic artery and puncturing posteriorly and superiorly toward the wire in aiming at the portal vein could make the procedure easier, while avoiding damage to the hepatic artery (8, 10).
Among the three operators who performed the hepatic artery guiding technique in the present study, two had less than 5 years of experience in interventional radiology. Although there were differences in levels of experience, mean puncture times of TIPS were not significantly different. Furthermore, TIPS is currently a rarely performed procedure, with less than five procedures performed per year in participating hospitals. This suggests that portal vein access with hepatic artery guiding procedure could be successfully performed with high reproducibility despite lack of experience with the operator.
Data on radiation doses from the procedure were available in 25 patients. Although there was no statistical significance, the hepatic artery guiding technique group had a higher radiation dose than the simple blind puncture group. Compared with previous reports describing ultrasound guided portal vein puncture, the hepatic artery guiding group in our study exhibited high radiation doses (129 ± 117 Gycm2 vs. 523.14 ± 255.00 Gycm2) (19). This is due to the fact that in the hepatic artery guiding group, more than one angiography including celiac and/or superior mesenteric artery angiography must be taken. Thus, with this technique, effort should be taken to reduce radiation dose.
There were several limitations in this study. First, as this study was designed in retrospective manner and is subject to selection bias. All accurate procedure times were difficult to obtain, and information on fluoroscopic times or radiation doses were not required in some patients. Also there may have outcome measurement error to occur. Thus, further study with randomized clinical trial is needed. Second, there may be limitations in confirming statistical significance due to the small number of patients used. Third, for an accurate evaluation of the technical feasibility, the present procedure should be compared to the various other portal vein puncture techniques. However, TIPS procedure is rarely performed in our institutes, and all operators performed simple puncture or hepatic artery guide puncture techniques. And last, decision of each technique was by operators’ preference which affect selection bias.
In conclusion, hepatic artery guiding technique could increase the feasibility of portal vein puncture in TIPS without a significant increase in radiation dose.