1. Introduction
The novel coronavirus disease 2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) is a highly contagious disease which was first detected in Wuhan City, Hubei Province in China (1). The World Health Organization (WHO) China country office reported cases of pneumonia of unknown etiology in December 2019 (2). An analysis of all cases reported until January 2020 revealed that it was a viral disease that was not seen in humans before (3). Although the virus responsible for the disease was designated as 2019 novel coronavirus (2019-nCoV) on January 7, 2020, it was later named as SARS-CoV-2 (COVID-19) due to its similarity to SARS-CoV (4).
The incubation period for COVID-19 is considered to extend to 14 days, with a median time of 5 - 6 days from exposure to symptoms. Severe acute respiratory syndrome coronavirus-2 shares many similarities with SARS-CoV and Middle East respiratory syndrome (MERS)-CoV. Severe acute respiratory syndrome coronavirus-2 and MERS-CoV outbreaks have caused significant morbidity and mortality between 2002 and 2003 and in 2012, respectively (4, 5). Severe acute respiratory syndrome and MERS have significantly higher case fatality rates than COVID-19. However, COVID-19 is more infectious. The disease which has affected nearly 213 countries to date has been declared a pandemic by the WHO on March 11, 2020 (6).
Severe acute respiratory syndrome coronavirus-2 Spike Antibody Test aids in identifying individuals with an adaptive immune response to the spike (S) protein of SARS-CoV-2. This assay allows detection of antibodies produced against the virus. Antibodies are immune system-related proteins known as immunoglobulins, which help the immune system recognize and eliminate foreign (non-self) threats to the body. Our immune system also produces some antibodies to protect our body against SARS-CoV-2 (COVID-19). In general, antiviral immunoglobulin M (IgM) antibodies appear about 10-15 days after the onset of symptoms and antiviral immunoglobulin G (IgG) antibodies become detectable 10 - 20 days after infection in patients. Quantitative measurement of the IgG antibody against the S protein of the virus is more valuable in assessing an individual’s immune response. The antibody testing is performed on blood samples and an anti-SARS-CoV-2 IgG Antibody value ≥ 50 AU/mL is considered positive (7, 8).
Studies have shown that while the age group most commonly affected by this infection is the 30 to 79 age group, the mortality rate is much higher in individuals over 80 years of age. Limited information is available on the disease course in pregnant women and the risks to the fetus and neonate; however, it is known that pregnant women constitute a vulnerable population who can be more severely affected by all viral infections compared to non-pregnant women (9). The Centers for Disease Control and Prevention (CDC) report states that women diagnosed with COVID-19 during pregnancy should be isolated under appropriate conditions and supported by local healthcare teams in order to reduce the possibility of transmission (10). In this case report, we present the results obtained from the cord blood of a baby born from a mother diagnosed with COVID-19 before getting pregnant who did not receive any vaccination during pregnancy and subsequently recovered. Before collecting study data, the mother was informed about the purpose of the study and both oral and written informed consent was obtained from the mother on 01.07.2021.
2. Case Presentation
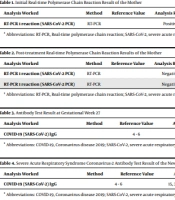
The mother is 29 years old and works as a medical doctor. She tested positive for COVID-19 on 24.07.2020 before getting pregnant and experienced symptoms of severe arthralgia, headaches and fever during the 10-day quarantine period in which she received treatment (Table 1). At the end of 10-day treatment period following the COVID-19 (+) result, the mother tested negative on a polymerase chain reaction (PCR) test on 03.08.2020. She tested negative again on a repeat PCR test on 04.08.2020 and the result was confirmed (Table 2).
| Analysis Worked | Method | Reference Value | Analysis Result | Material | Result Date/Time |
|---|---|---|---|---|---|
| RT-PCR 1 reaction (SARS-CoV-2 PCR) | RT-PCR | Positive | Combined nose and throat swab | 24.07.2020; 14:27 pm approved |
Abbreviations: RT-PCR, real-time polymerase chain reaction; SARS-CoV-2, severe acute respiratory syndrome coronavirus-2.
| Analysis Worked | Method | Reference Value | Analysis Result | Material | Result Date/Time |
|---|---|---|---|---|---|
| RT-PCR 1 reaction (SARS-CoV-2 PCR) | RT-PCR | Negative | Combined nose and throat swab | 03.08.2020; 14:53 pm approved | |
| RT-PCR 1 reaction (SARS-CoV-2 PCR) | RT-PCR | Negative | Combined nose and throat swab | 04.08.2020; 16:38 pm approved |
Abbreviations: RT-PCR, real-time polymerase chain reaction; SARS-CoV-2, severe acute respiratory syndrome coronavirus-2.
The mother reported that she had her last menstrual period on 26.09.2020 and became aware of her pregnancy at the end of September 2020. On 31.03.2021 (at gestational week 27), she underwent a follow-up antibody test which showed an IgG value of 50 AU/mL (Table 3). It was learnt that the mother did not develop any illness throughout her pregnancy but experienced “severe” COVID-19 symptoms before getting pregnant.
| Analysis Worked | Method | Reference Value | Analysis Result | Material | Result Date/Time |
|---|---|---|---|---|---|
| COVID-19 (SARS-CoV-2) IgG | 4 - 6 | 50 < positive | Blood test | 31.03.2021; 15:00 pm approved |
Abbreviations: COVID-19, coronavirus disease 2019; SARS-CoV-2, severe acute respiratory syndrome coronavirus-2; IgG, immunoglobulin G.
A male baby weighing 4170 g was born on 03.07.2021 at gestational week 41. A standard umbilical cord blood sample was collected from the newborn immediately after delivery under aseptical conditions to learn his blood type and to check his SARS-CoV-2 IgG level. Laboratory workup for the newborn’s cord blood demonstrated a SARS-CoV-2 IgG level of 15.32 AU/mL (Table 4). The baby underwent detailed health checks and was discharged as a completely healthy baby.
| Analysis Worked | Method | Reference Value | Analysis Result | Material | Result Date/Time |
|---|---|---|---|---|---|
| COVID-19 (SARS-CoV-2) IgG | 4 - 6 | 15, 32 positive | Blood test | 03.07.2021; 11:27 am approved |
Abbreviations: COVID-19, coronavirus disease 2019; SARS-CoV-2, severe acute respiratory syndrome coronavirus-2; IgG, immunoglobulin G.
5. Discussion
In this case report, it was demonstrated that antibodies against COVID-19 that developed in the mother before pregnancy passed to the fetus in utero and were detected in the newborn cord blood. Studies conducted during the pandemic have focused on mothers who were diagnosed with COVID-19 during pregnancy, prophylactic administration of COVID-19 vaccines to pregnant women, and COVID-19-related symptoms and antibody formation at birth in newborns following vertical transmission.
In a case report by Dong et al. published a 29-year-old woman at gestational week 34 was reported to present with complaints of fever and nasal congestion on 28.01.2020 and RT-PCR was positive for SARS-CoV-2 (11). The newborn showed no symptoms and at 2 hours after birth, the infant’s blood sample showed a SARS-CoV-2 IgG level of 140.32 AU/mL and an IgM level of 45.83 AU/mL. On 07.03.2020, the baby’s SARS-CoV-2 IgG level was 69.94 AU/mL and IgM level was 11.75 AU/mL, and she was discharged on 18.03.2020. That is a valuable case report showing exposure of the fetus to maternal COVID-19 infection for 23 days by vertical transmission during the intrauterine period (11).
In a 2020 study, Khan et al. reported their follow-up of 3 pregnant women who were COVID-19 positive at second and third trimesters (12). All 3 pregnant women delivered via vaginal route and were healthy, with no adverse outcomes observed in the postpartum period. PCR test results were negative for all newborns (12).
In one study, Zeng et al. evaluated six COVID-19 positive pregnant women and reported that none of the infants born to these mothers showed SARS-CoV-2 positivity on RT-PCR testing of throat swab and blood samples (13). However, all newborns had detectable antibodies against the virus in their serum samples (13).
Consistently, in a study by Mo et al. including six pregnant women, 3 of whom diagnosed with COVID-19 in the second trimester and 3 in the third trimester, all of their newborn infants were reported to have antibodies at birth (14). Among infants born to mothers infected in the 3rd trimester, 2 had detectable IgG and one had detectable IgM levels, whereas only IgG was found in infants born to mothers infected in the 2nd trimester (14).
The vertical transmission potential of COVID-19 in pregnant women was discussed in a study by Chen et al. published in the Lancet (15). Emergency preventive measures were implemented in 9 pregnant women with COVID-19 who developed pneumonia. Neonatal nasopharyngeal swab, amniotic fluid, cord blood and breast milk samples were collected and SARS-CoV-2 was diagnosed in 2 newborns (15).
In a case report by Paul and Chad, a pregnant healthcare worker with no history of COVID-19 was vaccinated with an mRNA COVID-19 vaccine (Moderna) at gestational week 36 (16). In line with hospital protocol, she underwent COVID-19 testing at the time of admission to the labor and delivery ward which was negative. At 3 weeks after the first dose of Moderna, a healthy girl was born via normal, spontaneous vaginal delivery. A standard cord blood sample (0.5 mL) was collected into a red-top tube immediately after the birth to determine newborn blood type under aseptic conditions. IgG antibodies to SARS-CoV-2 were detected in the cord blood at a level of 1.31 U/mL (16).
There are several studies in the literature showing significant differences among antibody levels in infants born to SARS-CoV-2 positive mothers or mothers who were vaccinated during pregnancy (11-16). However, no study was identified in both national and international literature reporting on the antibody level of the infants born to mothers who were SARS-CoV-2 positive before pregnancy.
3.1. Conclusions
Pregnant women and newborns should be evaluated and followed attentively since they are considered to be at-risk, prioritized populations in the COVID-19 outbreak. However, the case reported here shows us that the effects of a maternal COVID-19 infection occurring before pregnancy may be seen in the newborn. For this reason, consideration of the transmission routes of COVID-19 infection and transfer of antibodies to the newborn both before and during pregnancy will allow us to take measures to eliminate potential health risks to the mother and the baby.
Here, we presented the first known case of an infant with detectable SARS-CoV-2 IgG antibodies in the cord blood immediately after birth born to a symptomatic mother who was diagnosed with COVID-19 before getting pregnant and subsequently recovered with pharmacological treatment. We believe that this case report provides valuable data to the literature by showing the persistent nature of antibodies against SARS-CoV-2.