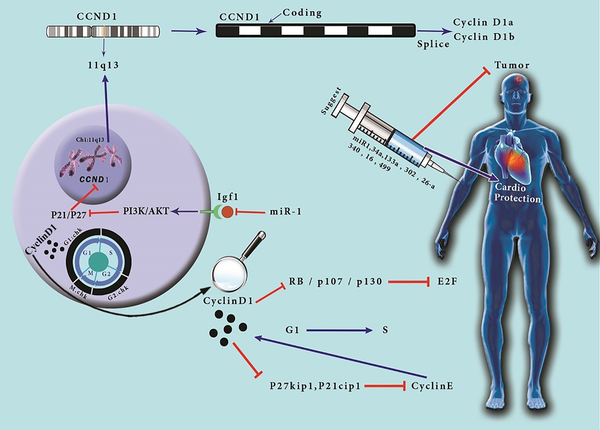
1. Context
CCND1 gene is located at chromosome 11, band q13, and encodes cyclin D1 (1). This gene has five coding exomes, that cyclin D1a and cyclin D1b transcripts are derived (2). Cyclin D1 is an essential protein acting as a mitogenic sensor (1). This protein is an allosteric activator of CDK4/6, which has an important role in cell cycle regulation, not only via phosphorylation of pRB, p107, and p130, which suppress E2F transcription factors activity in their hypophosphorylated forms and G1 phase proceeding to S phase of the cell cycle but also through separating cell cycle inhibitors p27Kip1 and p21Cip1 from cyclin E-CDK2 (3). Cyclin D1 overexpression and hyperphosphorylation of the mentioned proteins are common in cancers, but the former is not sufficient to proceed oncogenic transformation (1). Recent studies have suggested that cyclin D1 controls the G1 phase of cell cycle progression via both CDK-dependent and CDK-independent activities of cyclin D1 (4). There are remarkable advances in oncology leading to longer survival in patients. However, the mean cardiotoxicity associated with cancer therapies elevates the need for cardio-oncology to monitor the following cardiac events (5). A cohort study conducted by World Health Organization’s (WHO) drug monitoring database reported several cases of myositis and mortality in checkpoint inhibitor and anti-PD-1 users as well as major adverse cardiac events (MACEs) (6). The main reason for focusing on cardio-oncology is diagnosing the cardiac events at the earliest time and minimizing cardiovascular disease (CVD) in cancer patients and survivors (7) (Figure 1). In this manuscript, we discuss the role of cyclin D1 in oncology and cardio-oncology, and we challenge the prognostic and therapeutic response values of this protein to find out if it can be used as a useful marker. We also discuss the agents and microRNAs that can be used as a potential therapeutic approach via regulating cyclin D1 expression. We believe that the role of cyclin D1 in oncology and cardio-oncology should be highlighted together because the clinical significance of cyclin D1 is defined not only in several cancers such as breast cancer, melanoma, and glioblastoma but also in cardiomyocyte regeneration and cardiac hypertrophic growth. However, it has an oncogenic role which can have a protective effect on cardiomyocytes, so when it comes to its role in cancer development, we should notice its protective role on cardiomyocytes during chemotherapy; thus, we should work on the effective balance of cyclin D1 in cancer patients.
CCND1 gene is an allosteric activator of CDK4/6, which has an important role in cell cycle regulation, not only via phosphorylation of pRB, p107, and p130, which inhibit the transcriptional activity of E2F transcription factors in their hypophosphorylated forms and G1 phase proceeding to S phase of the cell cycle but also through separating cell cycle inhibitors p27Kip1 and p21Cip1 from cyclin E-CDK2. The clinical significance of cyclin D1 is defined not only in several cancers such as breast cancer, melanoma, and glioblastoma but also in cardiomyocyte regeneration and cardiac hypertrophic growth. According to our extensive research, we found that cyclin D1 overexpression is positively correlated with tumor progression, treatment response, cardiotoxicity, and poor prognosis and has an important role in cardiac hypertrophy, making it a promising marker in monitoring cardiomyocyte treatment responses. Cyclin D1 can induce cardioprotection via hypertrophic growth of cardiomyocytes through a novel CDK-independent mechanism, so it can be a favorable biomarker in future cardiology and cardio-oncology. We noticed that overexpression of some miRs such as miR-1, miR 34a, miR-133a, miR-302, miR- 26a, miR-340, miR-16 and miR-499 can be a favorable factor in suppressing cancer progression and increasing cardiomyocyte regeneration.
1.1. The Importance of Cyclin D1 in Cardiotoxicity
Cardio-oncology represents a new platform for the toxic effects of cancer treatments on the cardiac system. Recent advances have greatly improved the diagnosis of chemotherapy side effects on the cardiac system, including cardiovascular toxicity, QT prolongation, arrhythmia, myocardial ischemia, stroke, hypertension, thromboembolism, left ventricular dysfunction, and heart failure which might happen during or after chemotherapy (8). The first unexpected report was on trastuzumab (a monoclonal antibody that targets HER2 receptor) which developed cardiomyopathy (9). Awareness of cardiotoxicity during or after cancer treatments and optimization of cardiovascular (CV) risk factors are essential in increasing patient's survival (10). Finding early detectable biomarkers of cardiotoxicity is an important point in this respect (11). The myocardial performance index (MPI), which is a sensitive parameter in evaluating cardiac dysfunction, and serum biomarkers such as cTnT and NT-pro-BNP are useful parameters in the early diagnosis of chemotherapy-induced cardiotoxicity (12). Anthracyclines such as doxorubicin (DOX) induce severe cardiotoxic side effects in cancers, so it is important to prevent the negative side effects of DOX and to find early biomarkers of chemotherapy-induced cardiotoxicity. MicroRNAs (miRs) such as miR-34a, miR-34b, miR-187, miR-199a, miR-199b, and miR-424 have been reported to be associated with cardiomyocyte function and cardiotoxicity, and this finding supports the potential of these miRs as biomarkers for early detection of chemotherapy-induced cardiotoxicity (13).
Cyclin D1 is low in the adult heart, and its expression level might be associated with cardiac hypertrophy. Therefore, cyclin D1 expression in the adult heart might induce mechanisms distinct from those that drive proliferation during cardiac development, which might be valuable in inducing hypertrophic responses in the adult heart in response to cardiac injuries (14). Mitogenic stimulation increases hypertrophic growth and cyclin D1 expression in differentiated cardiomyocytes. Studies showed that cyclin D1 is placed in the nucleus of fetal cardiomyocytes. A variant of cyclin D1 (D1NLS) which is directly associated with the nuclear localization signals (NLSs), increases cell division in adult cardiomyocytes in the knock-in study. Thus, differentiated cardiomyocytes have the potential to reenter the cell cycle via cyclin D1 (15). The transgenic mice, which overexpress cyclin D1 in the heart, have shown that cyclin D1 overexpression presents the cardiomyocyte DNA synthesis in the mice (16). Several ex vivo and in vitro studies on cardiomyocytes suggested that the expression of cyclin D1 decreased in left ventricle hypertrophy and that cyclin D1 expression played an important role in cardiac hypertrophy (17). Due to their important roles in the regeneration of cardiomyocytes, cyclins, mainly cyclin D1, improves cardiomyocyte regeneration (18). Therefore, it seems that cyclin D1 can be used as a marker to estimate the effect of regenerative treatments in chemotherapy-induced cardiotoxicity.
Differentiated cardiomyocytes have low growth potential; however, several studies support the important roles of cyclin D1 and CDK4 as inducers of the cell cycle in cultured neonatal rat cardiomyocyte hypertrophy. A study on cyclin D1 confirmed that cyclin D1 and CDK4 overexpression led to hypertrophic growth of cardiomyocytes and that CDK inhibitors such as p16 did not suppress the hypertrophic growth of cardiomyocytes. Research has suggested that cyclin D1 and CDK4 play an important role in the hypertrophic growth of cardiomyocytes via a new mechanism (19). Overexpression of cyclin D1 by adenoviral gene transfer has been reported to induce cardiomyocyte hypertrophy (20). Since cardiomyocyte proliferation decreases after birth, its regenerative growth is limited and is not sufficient to affect repair mechanisms. A study on canine models of ischemic cardiomyopathy using echocardiographic recordings and myocardial sampling for measurement of gene expression of cyclins suggested that cyclins played several roles in hypertrophic responses in cardiomyocytes (21). A study on mouse heart showed that the injection of cardiotoxic agents such as DOX induced damage and increased cyclin D expression at a single injection, which might be related to DXO-mediated damage in the adult heart. However, in DOX-treated rat neonatal, the expression of cyclin D decreased. The differences in the gene expression profile in DOX-treated adult compared to neonatal cardiomyocytes have shown that several changes during the development may change the cardiomyocyte responses to the damages (22). Another study on developed Flag- and haemagglutinin-tagged cyclin D1 knock-in mouse suggested that low levels of cyclin D1 decreased the Notch1 transcript in cyclin D1-null (Ccnd1-/-) retinas. The mentioned study showed the transcriptional function of cyclin D1 in mouse development (23). Research on hearts of neonatal mice and adult zebrafish has shown that cyclin D1 expression increases just in differentiated cardiomyocytes of adult mice. Analysis of cell cycle in this study also indicates that > 40% of the examined cardiomyocytes restart the cell cycle via cyclin D1, and this finding supports the important role of cyclin D1 in cardiomyocyte regeneration and treatment (24).
1.2. The Role of Cyclin D1 in Therapeutic Response Prediction
Monitoring of minimal residual disease (MRD) is of critical importance for the management of metastasis in several cancers, and numerous studies suggested that assessing the evaluated CCND1 gene can be used as a novel MRD marker of metastasis in neuroblastoma (NB) (25).
MRD is a criterion for the disease outcome in mantle cell lymphoma (MCL). Several studies used cyclin D1 as a marker for quantitative MRD monitoring by assessing cyclin D1 mRNA. Since cyclin D1 monitoring reveals quantitative changes in relapse, successful treatment response, or disease remission, cyclin D1 detection can be suggested as a hopeful method for quantitative MRD monitoring in MCL cases (26). The level of cyclin D1 and underlying chromosomal aberration can be useful as molecular markers for MRD detention. The potential role of cyclin D1 in the pathogenesis of malignancies developed the idea of generating a monoclonal antibody specifically detecting cyclin D1 protein in different mechanisms. Bartkova et al, for instance, used the DCS‐6 antibody for breast carcinomas and concluded that this antibody had the potential to be used as a tool for diagnostic or prognostic evaluation (27). Non-small cell lung cancer (NSCLC), accounting for 85% of lung cancers, has several priorities for treatments such as chemotherapy, radiotherapy, and surgery, and the choice of treatment is determined by tumor-node-metastasis (TNM) stage. A study on cyclin D1 expression in NSCLC demonstrated that cyclin D1 expression could be an independent prognostic factor for determining the stage in NSCLC (28). More recent studies have suggested that cyclin D1 overexpression can be an indicator to predict complete or partial response to neoadjuvant chemotherapy with cisplatin: 5-FU, and a cyclin D1 negative profile in tumors may be resistant to this approach (29). A study in the genomic field has shown that cyclin D1 (CCND1) A870G polymorphism may be a predictive molecular marker in estimating the clinical outcome of colorectal cancer with received cetuximab. According to the mentioned study, this polymorphism is a good biomarker for estimating survival in metastatic colorectal cancer treated with TROG cetuximab (30). Cyclin D1 overexpression in breast cancer has been found to be associated with an impaired tamoxifen response, and it may increase the risk of disease recurrence after tamoxifen treatment. Therefore, cyclin D1 protein expression and CCND1 amplification can be used as indicators for tamoxifen responses (31).
1.3. The Role of Cyclin D1 in Treatment Approaches
1.3.1. The Challenge of Cyclin D1 in Cancer Treatment
The important role of cyclin D1-CDK4/6 in cell cycle progression makes it an interesting target for cancer treatment (32). Cyclin D1 does not have any enzymatic activity and makes it a mysterious therapeutic target. However, CDK4/CDK6 can be important targets in therapeutic approaches, and CDK inhibitors such as abemaciclib are generated based on this idea (33).
Studies on abemaciclib, support the efficacy of this medicine alone or in combination with fulvestrant in women with hormone receptor (HR)–positive, human epidermal growth factor receptor 2 (HER2)–negative advanced breast cancer. Hopefully, this combination was efficient as a basic therapy and it increases the survival in the patients (33). Metformin is a very useful antidiabetic agent to regulate glucose level, and recent studies suggest a new potential for metformin in decreasing cancer risk (34). Several studies have supported the effect of metformin on prostate cancer via decreasing cyclin D1 protein level through AMP kinase pathway on the energy sensor signaling pathway (35). Knockdown of LC3 and Beclin-1 (autophagy genes) has been shown to inhibit cyclin D1 expression, contributing to tumor growth and progression. Therefore, targeting autophagy genes can be a useful therapeutic approach in breast cancer treatment (36). Depsipeptide (FR901228) is a novel cyclic peptide inhibitor of histone deacetylase, which directly inhibits CDK activity and decreases cyclin D1 within 6 to 12 hours after treatment, and growth arrest happens in the early G1 phase. It can be regarded as a potential agent in cancer treatment (37). MicroRNAs (miRs) such as miR-17-5p/miR-20a miRNA cluster are inversely associated with increased cyclin D1 in breast cancer, and miR-17/20 inhibits breast cancer cell via decreasing cyclin D1 translation (38).
1.3.2. The Challenge of Cyclin D1 in Cardiotoxicity Treatment
Cardiomyocytes have limited capacity for cell division and numeral changes, so they are not naturally replaced following ischemic injuries; thus, the point is that the hypertrophic growth confirms the cardiac reaction to the events such as myocardial infarction (39). Therefore, we can say that hypertrophic reaction is a green card in the cellular response to harmful situations such as stroke, ischemia, and even cardiotoxicity. Recent studies have worked on reactivating cardiomyocyte mitotic activity by focusing on stem cell biology, which can be used for therapeutic purposes to replace injured cardiomyocytes and myocardium. However, stem cell-derived cardiomyocytes are also differentiated, and their mitotic activities are limited, so studying the pathways and the molecules that induce cell cycle can be very useful following cardiac injuries (40). Cyclin D1 and its partner CDK4 are essential molecules in cell cycle induction and increase the cell cycle reentry via Rb regulatory pathway, which is required for cell cycle progression in cardiomyocytes (41). Left ventricle hypertrophy is induced by several stimuli such as chemotherapy, and it can lead to cardiomyopathy and heart failure. Several signaling pathways associated with cyclin D play an important role in this process. Cyclin D1/CDK4 and cyclin D1/CDK6 pathways are potential therapeutic targets for heart failure and hypertrophy treatment (42). Since cardiomyocyte proliferation decreases remarkably after birth, regenerative growth in the postnatal heart via cardiomyocytes is limited, and it is not sufficient to repair cardiac injuries. Based on the fact that cyclin D expression can increase the cell cycle activity, the myocardial repair following injury is improved (43). Several indicators of new cardiomyogenesis in post-mitotic cardiomyocytes, such as cardiomyocyte-specific expression of fluorescence ubiquitination-based cell cycle indicators (FUCCIs), can be used as a detector in cardiac injuries. The FUCCI transgenic (FUCCI-Tg) mouse is a useful study in evaluating cell cycle progression in adult myocardial tissue (41). However, several studies indicated that cyclin D1 can also inhibit the differentiation of cardiomyocytes through CDK4-dependent phosphorylation of GATA4 and recent studies on mice suggested the regenerative potential in embryonic and neonatal life, but this does not reprogram the cell cycle in adult cardiomyocytes (44).
2. MicroRNAs: A Way to Regulate Cyclin D1 and a New Therapeutic Approach
MicroRNAs (miRs) are non-coding RNA molecules with the potential for silencing coding genes at the posttranscriptional level (45). MiRs can target various genes, and a single protein-coding gene could be targeted by several potential miRs. However, few miR/mRNA interactions are experimentally used (46). Numerous experiments on miRNA/CCND1 interactions have shown the importance of several miRNAs such as miR-193b in CCND1 regulation in melanoma development, and the prognostic value of this miR in melanoma has been reported (47). Cardiac miRs such as miR-1, miR133a, miR-208a/b, and miR-499 play an important role in cardiac developmental programs and can be valuable diagnostic biomarkers for the early diagnosis of acute myocardial infarction (AMI) (48). Studies in the field of cardiotoxicity support the therapeutic potential of cardiac miRs in chemotherapy-induced cardiotoxicity (49). Studies on miR-34a have suggested that miRNA plays a critical role in cardiac repair and regeneration post-myocardial infarction (MI) in neonates, and it can have a cardiac repair function (50). Studies on cancer have shown that miR-34a can inhibit prostate cancer metastasis in mice (51). MiR-34a is a transcriptional target of p53 that is down-regulated in many cancers such as glioblastoma. Several studies have indicated that miR-34a expression is down-regulated in glioblastoma, and miR-34a plays an important role in cell cycle arrest and inhibits c-Met and Notch and probably cyclin D1, making it a strong therapeutic target for future studies (52).
3. Conclusions
A growing body of evidence suggests that controlling cardiac risk factors may also improve cancer incidence and outcomes. Additional collaboration between oncologists and cardiology-oncologists should occur as part of medical intervention in cancer patients. The importance of cell cycle in proliferating cells is well known, but little is known about the role of cell cycle regulatory proteins in cardiac myocytes, which are completely differentiated cells.
The clinical significance of cyclin D1 is defined not only in several cancers such as breast cancer, melanoma, and glioblastoma but also in cardiomyocyte regeneration and cardiac hypertrophic growth. Therefore, it is necessary to validate and apply new factors in cancer screening and cardiac intervention. Cardiomyocyte regeneration towards the development of therapeutic targets for cancer patients is the final goal of oncologists, which will be facilitated by using standardized preclinical models to study cardiomyocyte regeneration and hypertrophy. In this study, we highlighted the importance of a molecular biomarker called cyclin D1 in oncology and cardio-oncology. According to our extensive research, we found that cyclin D1 overexpression is positively correlated with tumor progression, treatment response, cardiotoxicity, and poor prognosis and has an important role in cardiac hypertrophy, making it a promising marker in monitoring cardiomyocyte treatment responses. Cyclin D1 can induce cardioprotection via hypertrophic growth of cardiomyocytes through a novel CDK-independent mechanism, so it can be a favorable biomarker in future cardiology and cardio-oncology. We noticed that overexpression of some miRs, such as miR-1, can be a favorable factor in suppressing cancer progression and increasing cardiomyocyte regeneration. Therefore, overexpression of these highlighted miRs can be a very good therapeutic target in cancer treatment because they fight cancer cells and interestingly protect cardiomyocytes and cardiac system. We recommend future studies to evaluate these miRs in cancer treatment.