CM has been extensively used in in-vitro studies to mimic the microenvironment of cellular niche and, therefore, dissect the role of secretome on molecular processes (
4). It has been documented that CM derived from various cell types has substantial and multiple effects on target cells’ proliferation (
8), motility (
9), epithelial-mesenchymal transition (
10), gene expression (
11), cell viability (
12), and death (
13). This study, therefore, aimed to investigate the effect of CMs derived from cell lines on each other.
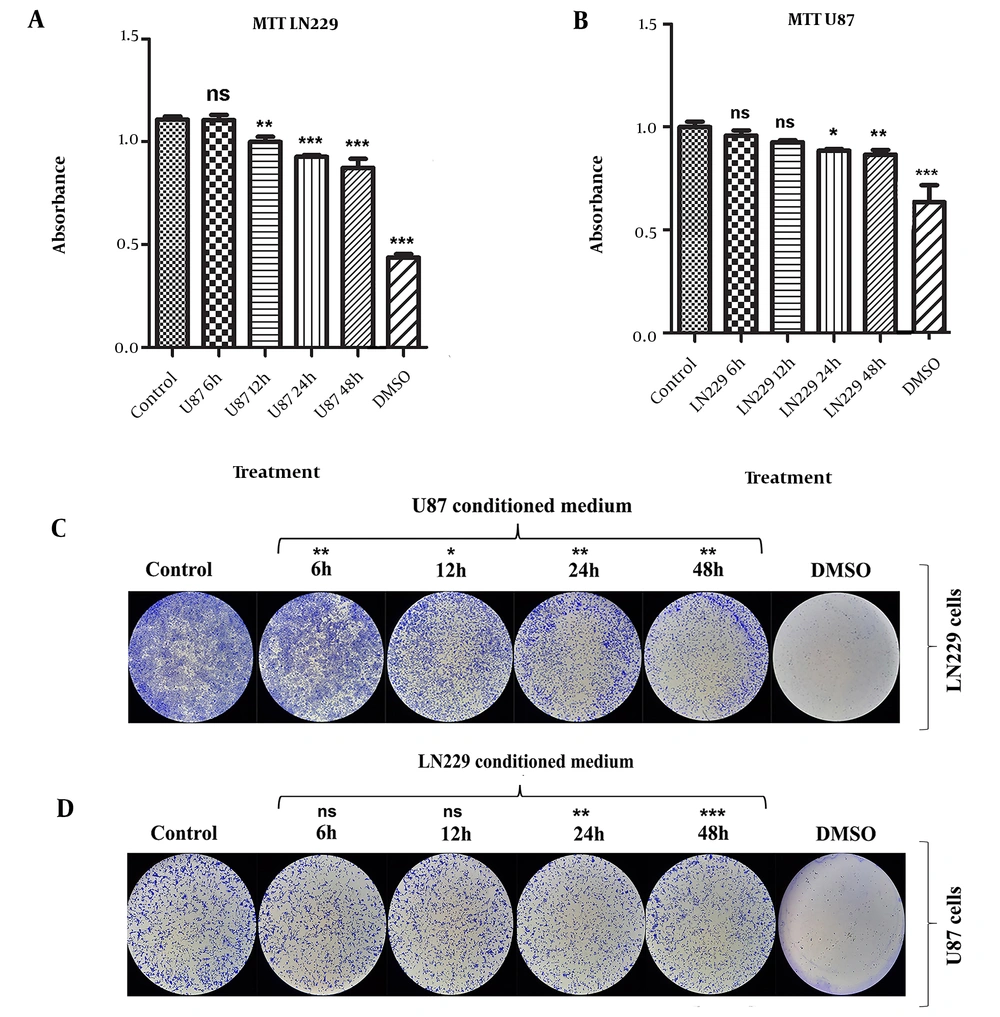
Our study results obtained from MTT and CFA unveiled that GBM CMs have considerably anti-growth effects on each other, particularly for the CMs collected at the 48th h time point. To explore the protein composition of 24th h CMs harvested from GBM cells (HNGC2, U87, and LN229), a proteome analysis was conducted, and proliferation-related proteins such as TIMP1, SERPIN F1, and mTOR were identified commonly as part of secretome content (
13). Intriguingly, CMs of GBM cell lines (U87, U373, and U251) collected after 24 h significantly decreased the proliferation rate of the cells following the reciprocal CM treatment (
14). Therefore, the content of the secretome may have an inhibitory effect on the cell division rate of target cells. Based on the literature, to the best of our knowledge, 48th h CM of GBM cells has not been studied extensively before. It is plausible to suggest that 48th h CM of GBM cells could contain anti-proliferative signals, which should be addressed in future studies.
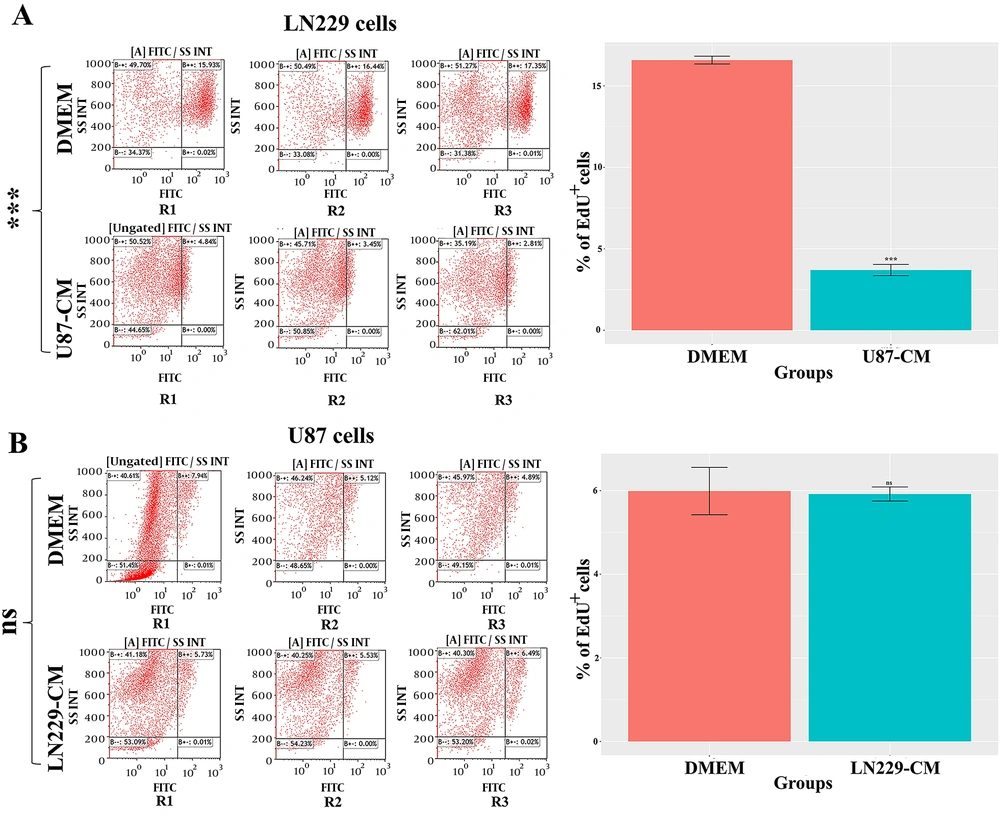
To further evaluate the growth-limiting effect of CMs of GBM cells on each other, the cell division rate was determined by EdU labelling. Cell proliferation results confirmed that U87-CM contained anti-proliferative factors for LN229 cells (
Figure 3A). In contrast, LN229-CM had no significant effect on the proliferation rate of U87 cells (
Figure 3B). As shown in a previous study, CMs harvested from GBM cells exhibited potential inhibitory effects on each other (
14). The given study found that the CM of U251 and U373 cells decreased the proliferation rate of U87. Interestingly, the CM of U87 cells did not significantly affect the proliferation rate of U251 or U373 cells. As mentioned earlier, the results from other studies regarding the diverse impact of GBM CMs on each other were in line with our findings.
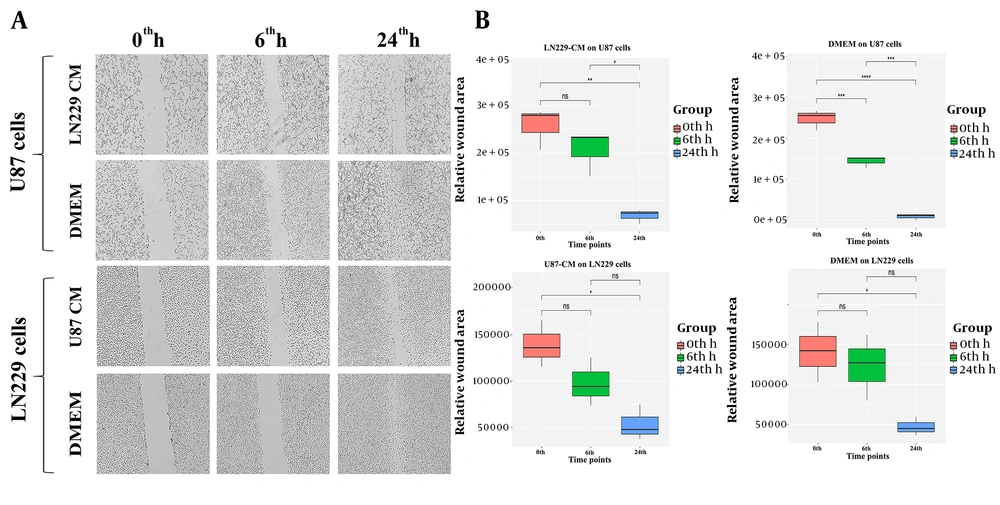
A significant anti-migratory effect of LN229-CM on U87 cells was observed (
Figure 2A -
B); however, U87-CM did not significantly affect the LN229 migration ability. In a recent study, CM collected from the B16F10 mouse melanoma cancer cells reduced macrophage motility (
15). On the other hand, there are some contrary reports on the migratory effect of CM. It has been reported that CMs derived from malignant breast cancer cell lines MDA-MB-231 and MDA-MB-453 increase the migration capacity of non-tumorigenic MCF10A cells (
16). The ambiguous effect of CM on the migration capacity might be attributed to the CM origin and target cells. More studies are required to unveil the content of LN229 and U87 CMs as well as to extend our current understanding of the contribution of CMs to cell migration capacity.
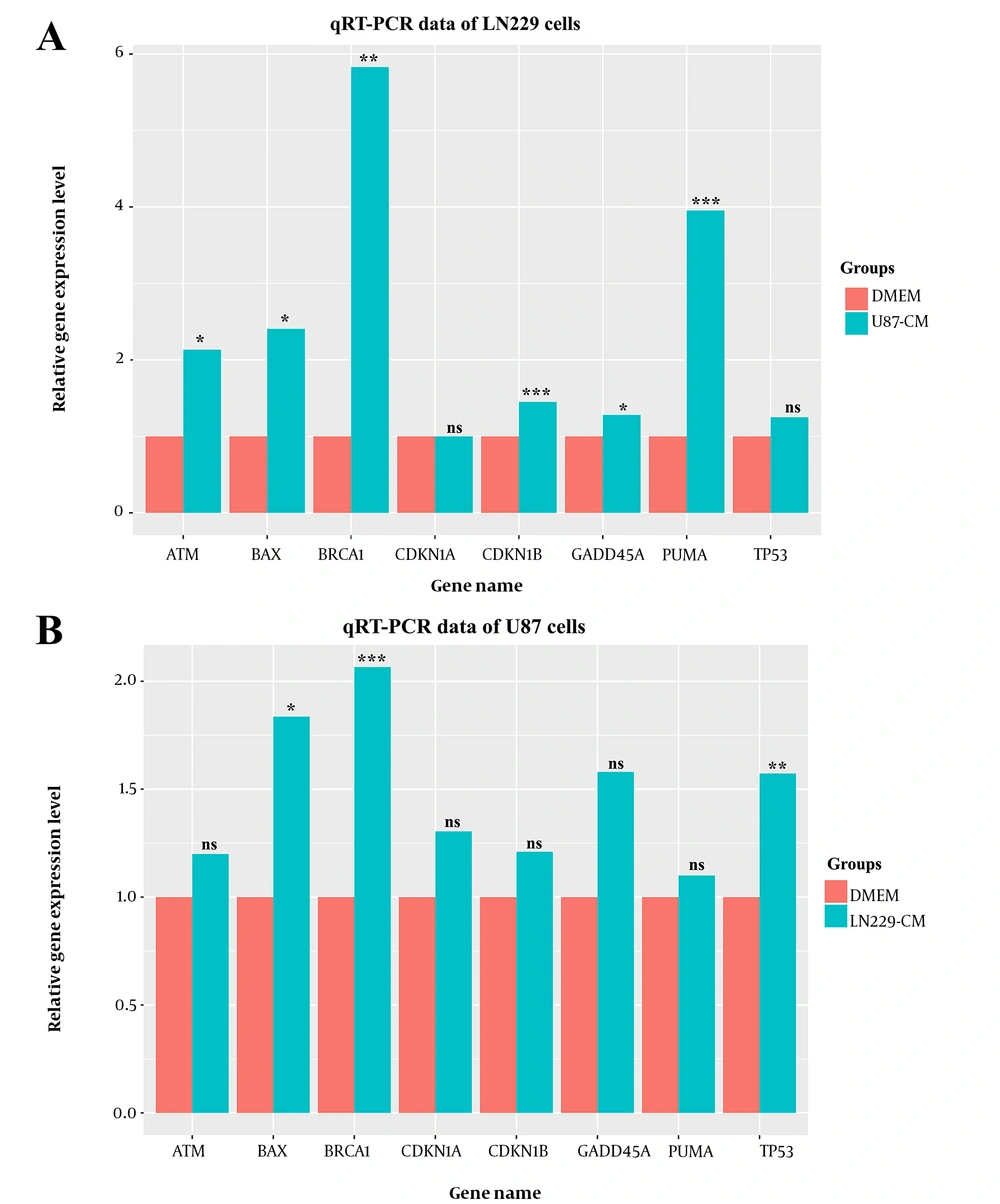
To get more insights into the anti-proliferative activity of CMs, the expression level of several anti-proliferative genes was evaluated in our study. Upon CM treatment, most of the anti-proliferative genes’ (GADD45A, CDKN1B, BAX, ATM, BRCA1, and PUMA) expression level was significantly upregulated in CM-treated LN229 cells compared to the control group. Increased anti-proliferative activity of genes in U87-CM treated LN229 cells provided plausible explanations for the decreased viability (
Figure 1) and proliferation rate (
Figure 3A). The diminished proliferation rate following CM treatment may have been associated with the inhibition of cell cycle progression by the increased GADD45A and CDKN1B levels. Moreover, upregulated pro-apoptotic factors may have accounted for the increased rate of cell death, which may have further decreased the number of alive cells following the CM treatment.
In contrast, a similar expression level for most anti-proliferative genes was detected in CM-treated and non-treated U87 cells. Moreover, no significant differences were observed in GADD45A, CDKN1A, CDKN1B, ATM, and PUMA levels. BAX, BRCA1, and TP53 were significantly upregulated in CM-treated cells which may have highlighted the anti-proliferative content of harvested CM. Altogether, activation of anti-proliferative gene expression upon CM treatment indicated a negative regulatory function of CMs on viability. Decreased viability upon CM treatment might have been associated with an elevated apoptosis and cell cycle arrest rate.
Several factors limited this study and its conclusions. Gene expression profile upon CM treatment may have been investigated using high throughput sequencing technologies which would allow the comprehensive elucidation of altered pathways. Moreover, cell lines at different glioma stages were likely more informative in revealing the CM effect on cancer development and aggressiveness.
5.1. Conclusions
In this study, CMs of GBM cell lines were used on each other to investigate the CMs’ effect at the cellular and molecular levels. It was demonstrated that GBM CMs collected at 48th h decreased the viability and modulated the gene expression of each other. Furthermore, the migration capacity of CM-treated U87 cells was remarkably reduced. Activation of anti-proliferative gene expression programs underlined the necessity of further studies to dissect the molecular content of the secretome.