1. Background
Diabetes is defined as an increase in blood glucose greater than the normal range. It is divided into two types: insulin-dependent (Type I) and non-insulin dependent (Type II). During the past 10 years, the prevalence of diabetes diagnosed in American adults was about 40% and increased from 9.4% in 1990 to 6.9% in 1999. Unfortunately, it is estimated that the incidence will increase 165% by the year 2050. This increase is mainly due to the increased in type-2 diabetes (1).
Diabetes is one of the most common diseases that complicate pregnancy. Increased prevalence of type 2 diabetes in general and in the young population in particular has led to an increase in the number of pregnancies complicated with this disease. The majority of women who are diagnosed with diabetes during pregnancy most likely already had type-2 diabetes, but it was not recognized (1).
Women with diabetes in pregnancy can be divided into two categories: those who were diagnosed with diabetes before pregnancy (overt diabetes) and those who developed diabetes during pregnancy (gestational diabetes).
Intrauterine involvement of the fetus with maternal hyperglycemia leads to increased fetal blood insulin levels and fat cells and eventually causes obesity and insulin resistance in childhood. This issue also leads to impaired glucose tolerance and diabetes in adulthood (1).
Recently, the role of various factors including leptin, which is one of the measurable biomarkers in the umbilical cord that can affect fetal growth rates in pregnant women with diabetes has been studied. Although the short-term impact of these factors on fetal growth has been recognized, its long-term consequences are unknown (2).
Leptin is one of the hormones produced mainly in adipose tissue. Evaluation of its level changes in lipid metabolism seems effective (3). Leptin is a protein composed of 167 amino acids and is secreted from adipose tissue (adipocytes). This hormone adjusts the body fat mass, appetite, and energy expenditure with an impact on the hypothalamus (4).
Leptin is built not only in adipocytes but also in the placenta. A leptin that is produced in the placenta chiefly enters maternal levels (5). About 98% of the placental leptin enters the maternal blood flow, and just about 2% enters the fetal circulation (6). Gross differences between the serum leptin concentration in the mother and in the umbilical cord indicate that leptin is not able to cross the placental barrier (7). The production rate of this hormone in the placenta varies in cases in which it causes a change in the growth of the embryo (preeclampsia and diabetes). One can also measure the amount of this hormone in the umbilical cord to estimate the accumulation rate of fat tissue in the fetal body (8).
Due to the lack of similar studies in Ahvaz, we aimed to determine the relationship between umbilical blood levels of this hormone and growth indices of newborns with normal mothers and mothers with gestational diabetes mellitus (GDM).
2. Objectives
Due to the lack of similar studies in Ahvaz, we aimed to determine the relationship between umbilical blood levels of this hormone and growth indices of newborns with normal mothers and mothers with gestational diabetes mellitus (GDM).
3. Materials and Methods
This analytical, cross-sectional prospective study was carried out with 84 pregnant mothers referring to Ahvaz Imam Khomeini Hospital from April 2013 to March 2014. This study was confirmed by the Ethics Committee of Ahvaz Jundishapur University of Medical Sciences, and all parents signed informed consent forms before entering the study.
Inclusion criteria were gestational age between 34 and 42 weeks, singleton pregnancies, and the first minute Apgar score equal to or greater than 8. Criteria for exclusion were chronic diseases of the mother (thyroid, renal, etc.), congenital sepsis, drug abuse in the mother (alcohol, cigarettes, etc.), chromosomal anomalies, perinatal asphyxia, or major congenital anomalies such as omphalocele.
After vaginal delivery or elective cesarean section and in sterile conditions, 5 mL of venous blood from the umbilical cord was prepared by neonatal resuscitation team personnel (who were present in all shifts and deliveries and all of whom were trained for umbilical blood sampling using a syringe or inserting a catheter) in already prepared tubes. The tubes were then blocked by paraffin and immediately sent to the hospital laboratory. Samples were initially centrifuged in the laboratory, and the serums were then kept at a temperature of −20°C. This process was done similarly for all “case” and “control” samples.
After collection, all samples were sent to the reference laboratory in a cold box. There, after standard calibration, the samples were tested using a Labor Diagnostika Nord (LDN, made in Germany) kit using an Eliza method, and the serum leptin level was measured in nanograms per milliliter (ng/mL). The remaining samples were maintained at a temperature of −20°C in the reference laboratory until the completion of the project to ensure the accuracy of the investigation; if necessary, repeated reviews were available.
3.1. Statistical Methods to Analyze the Results
Repeated measures as well as independent groups t-test, paired T, and chi‐square (χ2) tests were used to compare the relationship between variables; in addition, data were analyzed using SPSS version 19.
4. Results
In this study, 84 newborns were evaluated. Of these, 42 (50%) were male, and 42 were female (50%). They were equally divided into two groups: “case” and “control.” Each group included 21 male (50%) and 21 female newborns (50%). Sixty-two newborns (74%) were between 37 and 42 weeks old, and 22 newborns (26%) had a gestational age of 34 - 36 weeks. All newborns with low Apgar scores (≤ 7) were excluded. Among the newborns in these groups, small for gestational age (SGA) and intrauterine growth restricted (IUGR) cases were excluded.
In the “case” group, 10 newborns (24%) weighed more than 4000 grams, and the “control” group contained no newborns weighing more than 4000 grams. Seven (8%) of the total newborns weighed less than 2500 grams. In all the newborns in both groups, the lowest weight was 2100 grams (newborns with gestational age of 34 weeks), and the highest weight was 4650 grams.
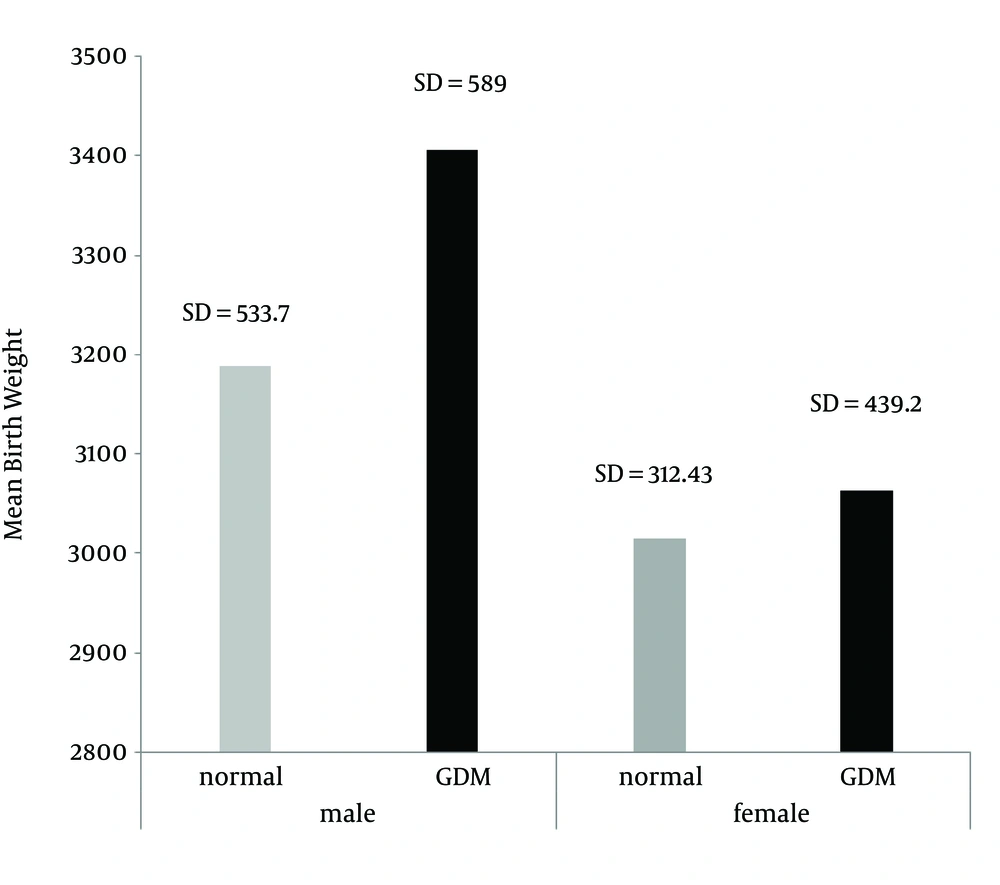
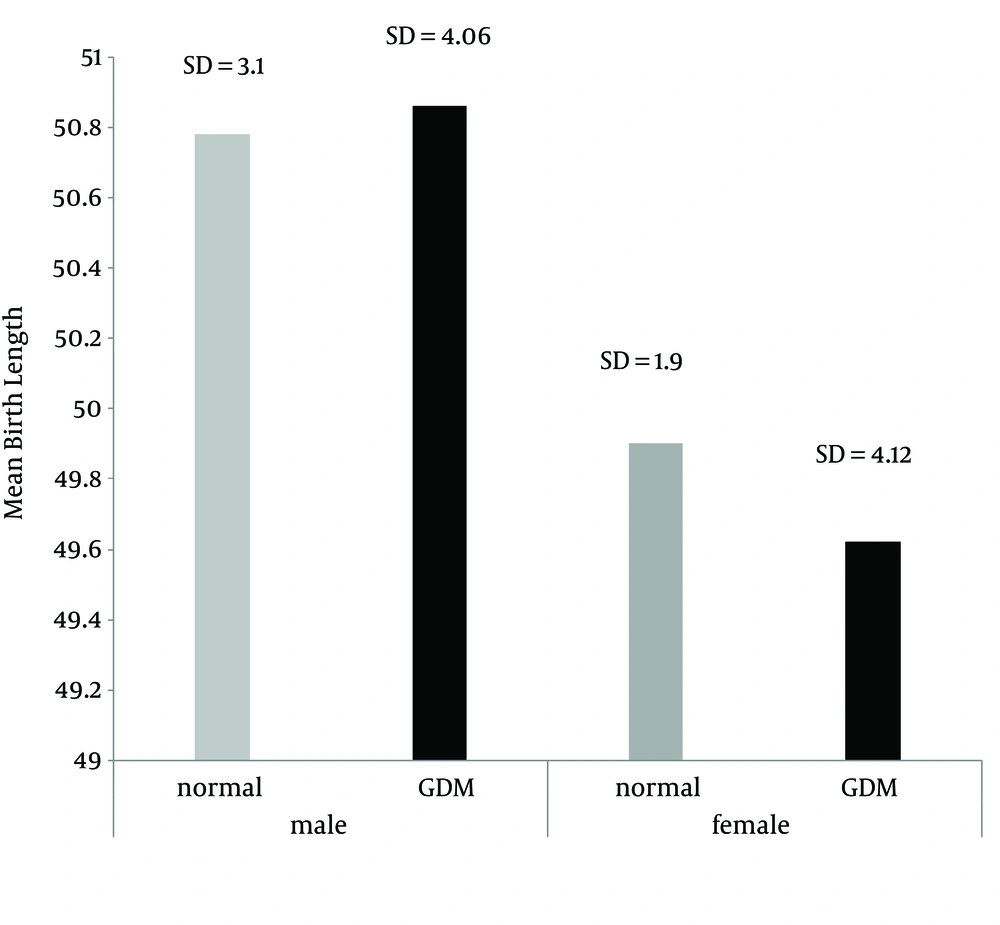
The average weight of infants in the “control” group was 3058.3 grams and in the “case” group was 3498.8 grams (Figure 1). The average heights in the “control” and “case” groups were 50.3 and 50.2 cm, respectively. No significant relationship was seen between the groups (Figure 2).
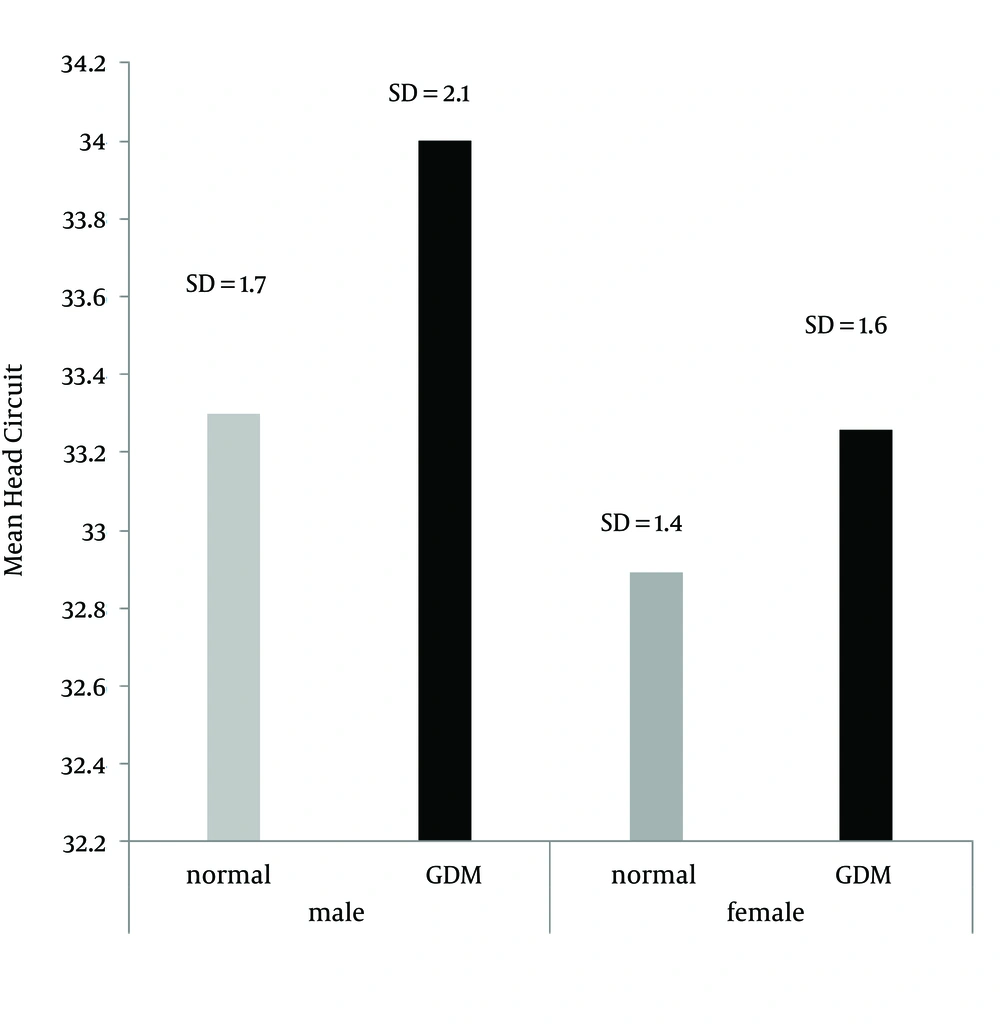
The maximum and minimum head circumferences were 37 and 29 cm. Twenty-five newborns (29%) had a head circumference smaller than 33 cm (Figure 3). The mean weight, height, and head circumference of boys and girls showed no significant statistical difference in either group.
The average serum leptin levels in the “control” group were 12.05 and 14.17 ng/mL in the male and female gender, respectively (P = 0.5). The same results were obtained in the “case” group; the average serum leptin levels in the “case” group were 100.88 and 91.39 ng/mL in the male and female gender, respectively (P = 0.7) (Table 1).
| The Average Serum Level of Leptin (Ng/ML) | Male | Female |
|---|---|---|
| Control group | 12.05 | 17.14 |
| Case group | 100.88 | 91.31 |
The Average Serum Leptin Levels in the “Case” Group Compared With the “Control” Group by Gender Distribution
The mean serum leptin levels in the “case” and “control” groups were 13.01 ng/mL and 96.13 ng/mL, respectively. As seen in Table 2, a strong significant correlation exists between leptin levels in the umbilical cord blood of infants in the “case” group compared with those in the “control” group (P < 0.001).
| Variable | Number (Average ± S.D.) | P Value | |
|---|---|---|---|
| Case | Control | ||
| Serum leptin level | 42 (96.13 ± 90.03) | 42 (13.01 ± 11.28) | < 0.001 |
| Weight at birth | 42 (3498.8 ± 558.3) | 42 (3068.3 ± 416.83) | < 0.001 |
| Height at birth | 42 (50.2 ± 4.09) | 42 (50.3 ± 2.69) | 0.8 |
| Head circumference at birth | 42 (33.6 ± 1.94) | 42 (33.1 ± 1.63) | 0.2 |
A Brief Look at the Values Obtained in This Study
5. Discussion
As was stated, a significant difference exists between the blood leptin level in the umbilical cords of newborns with mothers who have gestational diabetes compared to those with normal mothers (P < 0.001) (96.13 vs. 13. 01 ng/mL).
This finding is consistent with the results of other studies, including that conducted by Ornoy et al. in Canada in 2012. In this study for assessment of the value of different biomarkers on prognosis of fetal problems in diabetic mothers, Ornoy observed that the blood leptin level in the umbilical cords of newborns whose mothers had diabetes is higher and can be valuable in estimating fetal growth (2).
Lindsay et al. in England in 2004 showed that diabetes in the mother is associated with higher newborn weight and an increase in the levels of insulin and leptin (9). Likewise, Lepercq’s study in France in 2007 (8) and Silva’s study in Sri Lanka in 2008 (10) concluded that leptin level increases during pregnancy. Atavi’s study in Saudi Arabia in 2004 investigated a total of 38 pregnant mothers in the first, second, and third trimesters. The results of these study showed that leptin levels clearly increase in pregnancy and are reduced after delivery (11).
Based on statistical calculations, the sample size in each “case” and “control” group was estimated to be equal to 42 neonates. Since one of the objectives of the study was to investigate the relationship between cord blood level and neonate gender, we attempted to select those samples equally in both groups; in this way, the effect of unequal samples altering the results of study was resolved. In both groups, 21 female and 21 male newborns were studied. In the present study, no significant relationship existed between the serum leptin levels in the umbilical cord and newborns’ gender, consistent with the results of previous studies, including the study performed by Moradi et al. in 2006 in Iran (3). In that study, no significant differences were found between male and female newborns in two groups in terms of weight, height, and amount of leptin in the mothers as well as body mass index.
Additionally, a significant relationship was found between serum leptin levels in the umbilical cord of newborns with mothers who have gestational diabetes and weight in this group. This means that, in the group of infants with mothers who have gestational diabetes, the leptin level changes in the newborns’ umbilical cord were coordinated with weight. No such relationship existed in the infants with mothers without gestational diabetes mellitus. These results correlate with the results of other studies.
In Mexico in 2008, Velahorta showed that the level of leptin in large for gestational age (LGA) neonates is higher than in appropriate for gestational age (AGA) neonates, and hyperleptinemia may have a role in resistance to insulin in these two groups (12).
In addition, in a study in France in 2007, Lepercq observed that leptin levels in pregnancy increases, and this increase might have placental origin. The increased secretion of placental leptin in diseases such as preeclampsia and diabetes may be intensified. Similarly, the level of leptin in the umbilical cord can be used as an estimation of infants’ body fat tissue in the future (8).
Lindsay et al. in a study in Glasgow, UK (2004) observed that diabetes in the mother is associated with higher infant weight and an increase in insulin and leptin levels (9). In a study in Canada (2012) to assess the value of different biomarkers in prognosis of fetal problems in diabetic mothers, Ornoy perceived that the measurement of the leptin level in umbilical cord blood can be used in estimating fetal growth in diabetic mothers (2).
In their study in Toronto in 2012, Retnakaran concluded that the amount of fat tissue and blood leptin levels in non-pregnant mothers with diabetes are the strongest metabolic determining factors to predict LGA babies (13).
Karamizadeh, in a study in 2008 in Iran, found that there is no correlation between sex or length and cord venous serum leptin level and IGF-1, but birth weight was correlated with leptin in cord blood (CB) samples. Additionally, following classification into SGA and AGA groups, statistical calculation showed correlation between IGF-1 and leptin in cord blood samples. In all newborns, CB IGF-1 had a correlation with CB leptin (14).
Since the main source of leptin in the fetus, as an adult, is fat mass (3), we can summarize that the infant weight gain can be estimated using blood leptin levels in the umbilical cord. In this study, a significant difference was found in the serum level of leptin between the “case” and “control” groups; furthermore, a significant relationship existed between serum leptin levels in the “case” group and neonatal weight. Therefore, we can conclude that, by measuring the serum leptin level in the umbilical cord, we can obtain an estimation of fetal and infant weight and, if necessary, prevent possible complications.